Abstract
Hereditary xerocytosis (HX), also known as dehydrated stomatocytosis (DHSt) is a dominantly inherited genetic disorder exhibiting red cell membrane dehydration caused by the loss of the monovalent cation K+ and water. Variants in mechanosensitive Piezo ionic channels of the PIEZO1 gene are the primary cause of HX. We have utilized high throughput and highly precise next-generation sequencing (NGS) to make a diagnosis and examine the genotype-phenotype relationship in inflexible HX cases. Seven unrelated patients with unexplained hemolytic anemia were scrutinized with a panel probing 8000 genes related to congenital anemia. Targeted next-generation sequencing identified 8 missense variants in the PIEZO1 gene in 7 unrelated Indian patients. Three of the 8 variants are novel (c.1795G > C, c.2915G > A, c.7372 T > C) and the remaining five (c.4082A > G, c.6829C > A, c.7374C > G, c.7381G > A, c.7483_7488dup) are previously reported. The variants have been validated by Sanger sequencing. One patient with autosomal dominant mutation (c.7372 T > C) is associated with iron refractory iron deficiency anemia. Of the 7 patients, one has HX in combination with a novel homozygous variant (c.994G > A) in the PKLR gene causing PK deficiency resulting in severe clinical manifestations with phenotypic variability. In silico prediction using bioinformatics tools were used to study the possible damaging effects of the novel variants. Structural-functional analysis of the novel variants was investigated by molecular modeling software (PyMOL and Swiss PDB). These results encompass the heterogeneous behavior of mechano-sensitive Piezo1 protein observed in HX patients in India. Moreover, NGS imparted a subtle, economical, and quick tool for understanding the genetic cause of undiagnosed cases of congenital hemolytic anemia. NGS grants a potential technology integrating clinical history together with molecular report profiting in such patients and their families.



Similar content being viewed by others
References
Coste B, Mathur J, Schmidt M, Earley TJ, Ranade S, Petrus MJ, Dubin AE, Patapoutian A (2010) Piezo1 and Piezo2 are essential components of distinct mechanically activated cation channels. Science 330:55–60
Coste B, Xiao B, Santos JS, Syeda R, Grandl J, Spencer KS, Kim SE, Schmidt M, Mathur J, Dubin AE, Montal M, Patapoutian A (2012) Piezo proteins are pore-forming subunits of mechanically activated channels. Nature 483:176–181
Bagriantse SN, Gracheva EO, Gallagher PG (2014) Piezo proteins: regulators of mechanosensation and other cellular processes. J Biol Chem 289(46):31673–31681. https://doi.org/10.1074/jbc.R114.612697
Syeda R, Florendo MN, Cox CD, Kefauver J, Santos JS, Martinac D, Patapoutian A (2016) Piezo1 channels are inherently mechanosensitive. Cell Rep 17:1739–1746
Cahalan SM, Lukacs V, Ranade SS, Chien S, Bandell M, Patapoutian A (2015) Piezo1 links mechanical forces to red blood cell volume. Elife 4:e07370. https://doi.org/10.7554/eLife.07370
Gottlieb PA (2016) A tour de force: the discovery, properties, and function of Piezo channels. Curr Top Membr 79:1–36. ISSN 1063-5823. https://doi.org/10.1016/bs.ctm.2016.11.007
Alper SL (2017) Genetic diseases of PIEZO1 and PIEZO2 dysfunction. Curr Top Membr 79:97–126. https://doi.org/10.1016/bs.ctm.2017.01.001
Gottlieb PA, Sachs F (2012) Piezo1: properties of a cation-selective mechanical channel. Channels 6(4):214–219
Miller DR, Rickles FR, Lichtman MA, La Celle PL, Bates J, Weed RI (1971) A new variant of hereditary hemolytic anemia with stomatocytosis and erythrocyte cation abnormality. Blood 38(2):184–204
Fermo E, Bogdanova A, Petkova-Kirova P, Zaninoni A, Marcello AP, Makhro A, Hänggi P, Hertz L, Danielczok J, Vercellati C, Mirra N, Zanella A, Cortelezzi A, Barcellini W, Kaestner L, Bianchi P (2017) Gardos channelopathy: a variant of hereditary Stomatocytosis with complex molecular regulation. Sci Rep 7:1744. https://doi.org/10.1038/s41598-017-01591-w
Andolfo I, Russo R, Manna F, Shmukler BE, Gambale A, Vitiello G, De Rosa G, Brugnara C, Alper SL, Snyder LM, Iolascon A (2015) A novel Gardos channel variants linked to dehydrated hereditary stomatocytosis (xerocytosis). Am J Hematol 90(10):921–926
Rapetti-Mauss R, Lacoste C, Picard V, Guitton C, Lombard E, Loosveld M, Nivaggioni V, Dasilva N, Salgado D, Desvignes JP, Béroud C, Viout P, Bernard M, Soriani O, Vinti H, Lacroze V, Feneant-Thibault M, Thuret I, Guizouarn H, Badens CA (2015) Mutation in the Gardos channel is associated with hereditary xerocytosis. Blood 126(11):1273–1280
Glogowska E, Lezon-Geyda K, Maksimova Y, Schulz VP, Gallagher PG (2015) Red cells, iron, and erythropoiesis variants in the Gardos channel (Kcnn4) are associated with hereditary xerocytosis. Blood 126(11):1281–1284
Delaunay J (2004) The hereditary stomatocytosis: genetic disorders of the red cell membrane permeability to monovalent cations. Semin Hematol 41(2):165–172
Delaunay J (2007) The molecular basis of hereditary red cell membrane disorders. Blood Rev 21(1):1–20
Archer NM, Shmukler BE, Andolfo I, Vandorpe DH, Gnanasambandam R, Higgins JM, Rivera A, Fleming MD, Sachs F, Gottlieb PA, Iolascon A, Brugnara C, Alper SL, Nathan DG (2014) Hereditary xerocytosis revisited. Am J Hematol 89:1142–1146
Kaufman HW, Niles JK, Gallagher DR, Rivera A, Alper SL, Brugnara C, Snyder LM. (2018) The revised prevalence estimate of possible hereditaryxerocytosis as derived from a large U.S. Laboratory database. Am J Hematol 93(1):E9-E12. https://doi.org/10.1002/ajh.24923
Albuisson J, Murthy SE, Bandell M, Coste B, Louis-Dit-Picard H, Mathur J, Fénéant-Thibault M, Tertian G, de Jaureguiberry JP, Syfuss PY, Cahalan S, Garçon L, Toutain F, Simon Rohrlich P, Delaunay J, Picard V, Jeunemaitre X, Patapoutian A (2013) Dehydrated hereditary stomatocytosis linked to gain-of-function variants in mechanically activated PIEZO1 ion channels. Nat Commun 4:1884
Andolfo I, Alper SL, Franceschi LD, Auriemma C, Russo R, Falco LD, Vallefuoco F, Esposito MR, Vandorpe DH, Shmukler BE, Narayan R, Montanaro D, Armiento MD, Vetro A, Limongelli I, Zuffardi O, Glader BE, Schrier SL, Brugnara C, Stewart GW, Delaunay J, Iolascon A (2013) Multiple clinical forms of dehydrated hereditary stomatocytosis arisefrom variants in PIEZO1. Blood 121(19):3925–3935. https://doi.org/10.1182/blood-2013-02-482489
Da Costa L, Suner L, Galimand J, Bonnel A, Pascreau T, Couque N, Fenneteau O, Mohandas N (2016) Diagnostic tool for red blood cell membrane disorders: assessment of a new generation ektacytometer. Blood Cells Mol Dis 56:9–22
Syfuss PY, Ciupea A, Brahimi S, Cynober T, Stewart GW, Grandchamp B, Beaumont C, Tchernia G, Delaunay J, Wagner JC (2006) Mild dehydrated hereditary stomatocytosis revealed by marked hepatosiderosis. Clin Lab Haematol 28(4):270–274
Grootenboer S, Barro C, Cynober T, Olivier Schischmanoff P, Ayoubi JM, Tchernia G, Delaunay J, Pons JC (2001) Dehydrated hereditary stomatocytosis: a cause of prenatal ascites. Prenat Diagn 21(13):1114–1118
Iolascon A, Stewart GW, Ajetunmobi JF, Perrotta S, Delaunay J, Carella M, Zelante L, Gasparini P (1999) Familial pseudohyperkalemia maps to the same locus as dehydrated hereditary stomatocytosis (hereditary xerocytosis). Blood 93(9):3120–3123
Stewart GW, Amess JAL, Eber SW, Kingswood C, Lane PA, Smith BD, Mentzer WC (1996) Thromboembolic disease after splenectomy for hereditary stomatocytosis. Br J Haematol 93(2):303–310
Jaïs X, Till SJ, Cynober T, Ioos V, Garcia G, Tchernia G, Dartevelle P, Simonneau G, Delaunay J, Humbert M (2003) An extreme consequence of splenectomy in dehydrated hereditary stomatocytosis: gradual thromboembolic pulmonary hypertension and lung-heart transplantation. Hemoglobin 27(3):139–147
Glogowska E, Schneider ER, Maksimova Y, Schulz VP, Lezon-Geyda K, Wu J, Radhakrishnan K, Keel SB, Mahoney D, Freidmann AM, Altura RA, Gracheva EO, Bagriantsev SN, Kalfa TA, Gallagher PG (2017) Novel mechanisms of PIEZO1 dysfunction in hereditary xerocytosis. Blood 130(16):1845–1856. https://doi.org/10.1182/blood-2017-05-786004
Andolfo I, Russo R, Gambale A, Iolascon A (2017) Hereditary stomatocytosis: an underdiagnosed condition. Am J Hematol 93:107–121. https://doi.org/10.1002/ajh.24929
Zarychanski R, Schulz VP, Houston BL, Maksimova Y, Houston DS, Smith B, Rinehart J, Gallagher PG (2012) Variants in the mechanotransduction protein PIEZO1 are associated with hereditary xerocytosis. Blood 120:1908–1915. https://doi.org/10.1182/blood-2012-04-422253
Houston BL, Zelinski T, Israels SJ, Coghlan G, Chodirker BN, Gallagher PG, Houston DS, Zarychanski R (2011) Refinement of the hereditary xerocytosis locus on chromosome 16q in a large Canadian kindred. Blood Cells Mol Dis 47(4):226–231
Gnanasambandam R, Bae C, Gottlieb PA, Sachs F (2003) Ionic selectivity and permeation properties of human PIEZO1 channels. PLoS One 10:e0125503. https://doi.org/10.1371/journal.pone.0125503
Cinar E, Zhou S, De Courcey J, Wang Y, Waugh RE, Wan J (2015) Piezo1 regulates the mechanotransductive release of ATP from human RBCs. Proc Natl Acad Sci U S A 112:11783–11788. https://doi.org/10.1073/pnas.1507309112
Faucherre A, Kissa K, Nargeot J, Mangoni ME, Jopling C (2014) Piezo1 plays a role in erythrocyte volume homeostasis. Haematologica 99:70–75. https://doi.org/10.3324/haematol.2013.086090
Li J, Hou B, Tumova S, Muraki K, Bruns A, Ludlow MJ, Sedo A, Hyman AJ, McKeown L, Young RS, Yuldasheva NY, Majeed Y, Wilson LA, Rode B, Bailey MA, Kim HR, Fu Z, Carter DA, Bilton J, Imrie H, Ajuh P, Dear TN, Cubbon RM, Kearney MT, Prasad RK, Evans PC, Ainscough JF, Beech DJ (2014) Piezo l integration of vascular architecture with physiological force. Nature 515:279–U308
Ranade SS, Qiu Z, Woo SH, Hur SS, Murthy SE, Cahalan SM, Xu J, Mathur J, Bandell M, Coste B, Li YS, Chien S, Patapoutian A (2014) Piezo1, a mechanically activated ion channel, is required for vascular development in mice. Proc Natl Acad Sci 111:10347–10352
Ma S, Cahalan S, LaMonte G, Grubaugh ND, Zeng W, Murthy SE, Paytas E, Gamini R, Lukacs V, Whitwam T, Loud M, Lohia R, Berry L, Khan SM, Janse CJ, Bandell M, Schmedt C, Wengelnik K, Su AI, Honore E, Winzeler EA, Andersen KG, Patapoutian A (2018) Common PIEZO1 allele in African populations causes RBC dehydration and attenuates plasmodium infection. Cell 173(2):443–455.e12
Ge J, Li W, Zhao Q, Li N, Chen M, Zhi P, Li R, Gao N, Xiao B, Yang M (2015) Architecture of the mammalian mechanosensitive Piezo1 channel. Nature 527:64–69
Zhao Q, Zhou H, Chi S, Wang Y, Wang J, Geng J, Wu K, Liu W, ZhangMeng-Qiu Dong T, Wang J, Li X, Xiao B (2018) Structure and mechanogating mechanism of the Piezo1 channel. Nature 554:487–492
Bae C, Gnanasambandam R, Nicolai C, Sachs F, Gottlieb PA (2013) Xerocytosis is caused by variants that alter the kinetics of the mechanosensitive channel PIEZO1. Proc. Natl. Acad. Sci 110:E1162–E1168. https://doi.org/10.1073/pnas.1219777110
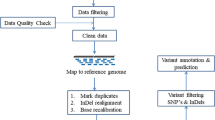
Kedar PS, Harigae H, Ito E, Muramatsu H, Kojima S, Okuno Y, Fujiwara T, Dongerdiye R, Warang PP, Madkaikar MR (2019) Study of pathophysiology and molecular characterization of congenital anemia in India using targeted next-generation sequencing approach. Int J Hematol 110:618-626 https://doi.org/10.1007/s12185-019-02716-9
Clarke GM, Higins T (2000) Laboratory investigation of hemoglobinopathies and thalassemias: review and update. Clin Chem 46(8):1284–1290
Dacie JV, Lewis SM (1999) Practical hematology, 8th edn. Churchill Livingston, London
Warang P, Gupta M, Kedar P, Ghosh K, Colah R (2011) Flow cytometric osmotic fragility-an effective screening approach for red cell membranopathies. Cytometry B Clin Cytom 80(3):186–190
Kedar PS, Colah RB, Kulkarni S, Ghosh K, Mohanty D (2003) Experience with eosin-5′-maleimide as a diagnostic tool for red cell membrane disorders. Clin Lab Haematol 25(6):373–376
King MJ, Garçon L, Hoyer JD, Iolascon A, Picard V, Stewart G, Bianchi P, Lee SH, Zanella A (2015) International Council for Standardization in Haematology ICSH guidelines for the laboratory diagnosis of nonimmune hereditary red cell membrane disorders. Int J Lab Hematol 37(3):304–325
Beutler E (1984) Red cell metabolism: a manual of biochemical methods, 3rd edn. Grune & Stratton, Orlando
Sambrook J, Fritsch EF, Maniatis T (1989) Molecular cloning, a laboratory manual, 2nd edn. Cold Spring Harbor Laboratory Press, New York
Li H, Durbin R (2010) Fast and accurate long-read alignment with burrows-wheeler transform. Bioinformatics 26(5):589–595. https://doi.org/10.1093/bioinformatics/btp698
Kircher M, Witten DM, Jain P, O'Roak BJ, Cooper GM, Shendure J (2014) A general framework for estimating the relative pathogenicity of human genetic variants. Nat Genet 46(3):310–315
Coste B, Murthy SE, Mathur J, Schmidt M, Mechioukhi Y, Delmas P, Patapoutian A (2015) Piezo1 ion channel pore properties are dictated by C-terminal region. Nat Commun 6:7223. https://doi.org/10.1038/Ncomms8223
Moroni M, Servin-Vences MR, Fleischer R, Sánchez-Carranza O, Lewin GR (2018) Voltage gating of mechanosensitive Piezo channels. Nat Commun 9:1096. https://doi.org/10.1038/S41467-018-03502-7
Picard V, Guitton C, Thuret I, Rose C, Bendelac L, Ghazal K, Aguilar-Martinez P, Badens C, Barro C, Bénéteau C, Berger C, Cathébras P, Deconinck E, Delaunay J, Durand JM, Firah N, Galactéros F, Godeau B, Jaïs X, Jaureguiberry JP, Stradic CL, Lifermann F, Maffre R, Morin G, Perrin J, JPM R, Toutain F, Lahary A, Garçon L (2019) Clinical and biological features in PIEZO1-hereditary xerocytosis and gardoschannelopathy: a retrospective series of 126 patients. Haemtologica 104(6):11. https://doi.org/10.3324/haematol.2018.205328
Bakker E (2006) Is the DNA sequence the gold standard in genetic testing? Clin Chem 52(4):557–558. https://doi.org/10.1373/clinchem.2005.066068
Katsanis SH, Katsanis N (2013) Molecular genetic testing and the future of clinical genomics. Nat Rev Genet 14(6):415–426. https://doi.org/10.1038/nrg3493
Park J, Jang W, Han E, Chae H, Yoo J, Kim Y, Kim M (2018) Hereditary dehydrated stomatocytosis with splicing site mutation of PIEZO1 mimicking myelodysplastic syndrome diagnosed by targeted next-generation sequencing. Pediatr Blood Cancer 65:e27053
Acknowledgments
We would like to thank patients and family members for their cooperation and participation in this study. This study was performed with the support of the Indian Council of Medical Research New Delhi, and the Department of Science and Technology (DST) New Delhi for financial support.
Funding
This study was performed with the financial support of the Indian Council of Medical Research New Delhi and the Department of Science and Technology New Delhi (SERB Project No. EEQ/2017/000115).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
The study protocol was approved by the Institutional Ethical Committee of ICMR-National Institute of Immunohematology, Mumbai. Informed consent was obtained from the parents or guardians of patients prior to enrollment in the study. All procedures performed in our study were in accordance with the 1964 Helsinki declaration.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
ESM 1
(DOCX 40 kb)
Rights and permissions
About this article
Cite this article
More, T.A., Dongerdiye, R., Devendra, R. et al. Mechanosensitive Piezo1 ion channel protein (PIEZO1 gene): update and extended mutation analysis of hereditary xerocytosis in India. Ann Hematol 99, 715–727 (2020). https://doi.org/10.1007/s00277-020-03955-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00277-020-03955-1