Abstract
Introduction
Fine lavender and lavandin are perfume and medicinal plants originate from the South of France and are widely cultivated for their essential oils. Recently, cultivated plants suffered from a severe decline in France, due to the propagation of the yellow decline disease. This disease is caused by the stolbur phytoplasma, a bacterium transmitted by a sap-sucking insect, the planthopper.
Objectives
In order to understand the complex relationships between host plant, pest, pathogen and environment responsible for the yellow decline of lavender, we use a metabolomic approach to highlight changes in chemical emissions from asymptomatic (“healthy”) and symptomatic (“infected”) plants.
Methods
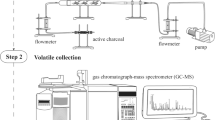
Volatile compounds produced by fine lavender and lavandin were collected in the field using a dynamic headspace extraction approach. Afterwards, compounds trapped on Tenax adsorbent were thermodesorbed and analysed using an automated thermal desorption-gas chromatography–mass spectrometry (ATD-GC–MS). Multivariate statistical analyses was performed using principal component analysis and partial least square discriminant analyses.
Results
The untargeted screening of volatiles allowed the separation of asymptomatic and symptomatic plants according to their emissions. The approach was sufficiently accurate so as to separate the emissions according to the different stages of infection. Twelve compounds were found to be deregulated metabolites of yellow disease infection, common to fine lavender (variety 7713) and lavandin (variety abrial).
Conclusion
The metabolomic approach allowed for the effective identification of chemical variations between infected and healthy plants in a complex field environment.

modified from CRIEPPAM) (Color figure online)







Similar content being viewed by others
References
An, M., Haig, T., & Hatfield, P. (2001). On-site field sampling and analysis of fragrance from living Lavender (Lavandula angustifolia L.) flowers by solid-phase microextraction coupled to gas chromatography and ion-trap mass spectrometry. Journal of Chromatography A,917(1–2), 245–250. https://doi.org/10.1016/S0021-9673(01)00657-4.
Becker, C., Desneux, N., Monticelli, L., Fernandez, X., Michel, T., & Lavoir, A.-V. (2015). Effects of abiotic factors on HIPV-mediated interactions between plants and parasitoids. BioMed Research International,2015, 1–18. https://doi.org/10.1155/2015/342982.
Bertaccini, A., & Duduk, B. (2009). Phytoplasma and phytoplasma diseases: A review of recent research. Phytopathologia Mediterranea,48, 355–378.
Bertamini, M., & Nedunchezhian, N. (2001). Effects of phytoplasma [Stolbur-Subgroup (Bois Noir-BN)] on photosynthetic pigments, saccharides, ribulose 1,5-bisphosphate carboxylase, nitrate and nitrite reductases, and photosynthetic activities in field-grown grapevine (Vitis vinifera L. cv. Chardonnay) leaves. Photosynthetica,39(1), 119–122. https://doi.org/10.1023/A:1012412406727.
Bruce, T. J. A., & Pickett, J. A. (2011). Perception of plant volatile blends by herbivorous insects—Finding the right mix. Phytochemistry,72(13), 1605–1611. https://doi.org/10.1016/j.phytochem.2011.04.011.
Bruce, T. J. A., Midega, C. A. O., Birkett, M. A., Pickett, J. A., & Khan, Z. R. (2010). Is quality more important than quantity? Insect behavioural responses to changes in a volatile blend after stemborer oviposition on an African grass. Biology Letters,6(3), 314–317. https://doi.org/10.1098/rsbl.2009.0953.
Chaisse, E., Fontaine, M., Costes, T., Taquin, S., Vial, G., Perrot, S., & Bringer-Guerin, C. (2017). L’Essentiel, Le bulletin technique et économique de la filière plantes à parfum. p 90.
Chuche, J., Danet, J.-L., Rivoal, J.-B., Arricau-Bouvery, N., & Thiéry, D. (2018). Minor cultures as hosts for vectors of extensive crop diseases: Does Salvia sclarea act as a pathogen and vector reservoir for lavender decline? Journal of Pest Science,91(1), 145–155. https://doi.org/10.1007/s10340-017-0885-5.
Da Porto, C., & Decorti, D. (2008). Analysis of the volatile compounds of flowers and essential oils from Lavandula angustifolia cultivated in Northeastern Italy by Headspace Solid-Phase Microextraction coupled to Gas Chromatography-Mass Spectrometry. Planta Medica,74(2), 182–187. https://doi.org/10.1055/s-2008-1034295.
De Moraes, C. M., Lewis, W. J., Paré, P. W., Alborn, H. T., & Tumlinson, J. H. (1998). Herbivore-infested plants selectively attract parasitoids. Nature,393(6685), 570–573. https://doi.org/10.1038/31219.
Dicke, M. (2009). Behavioural and community ecology of plants that cry for help. Plant, Cell & Environment,32(6), 654–665. https://doi.org/10.1111/j.1365-3040.2008.01913.x.
Domingo-Almenara, X., Brezmes, J., Vinaixa, M., Samino, S., Ramirez, N., Ramon-Krauel, M., et al. (2016). eRah: A computational tool integrating spectral deconvolution and alignment with quantification and identification of metabolites in GC/MS-based metabolomics. Analytical Chemistry,88(19), 9821–9829. https://doi.org/10.1021/acs.analchem.6b02927.
Dong, F., Fu, X., Watanabe, N., Su, X., & Yang, Z. (2016). Recent advances in the emission and functions of plant vegetative volatiles. Molecules,21(2), 124. https://doi.org/10.3390/molecules21020124.
Engelberth, J., Alborn, H. T., Schmelz, E. A., & Tumlinson, J. H. (2004). Airborne signals prime plants against insect herbivore attack. Proceedings of the National Academy of Sciences,101(6), 1781–1785. https://doi.org/10.1073/pnas.0308037100.
Fabre, A., Danet, J.-L., & Foissac, X. (2011). The stolbur phytoplasma antigenic membrane protein gene stamp is submitted to diversifying positive selection. Gene,472(1–2), 37–41. https://doi.org/10.1016/j.gene.2010.10.012.
Giray, F. H. (2018). An analysis of world lavender oil markets and lessons for Turkey. Journal of Essential Oil Bearing Plants,21(6), 1612–1623. https://doi.org/10.1080/0972060X.2019.1574612.
Harshman, S. W., Mani, N., Geier, B. A., Kwak, J., Shepard, P., Fan, M., et al. (2016). Storage stability of exhaled breath on Tenax TA. Journal of Breath Research,10(4), 046008. https://doi.org/10.1088/1752-7155/10/4/046008.
Hervé, M. R., Nicolè, F., & Lê Cao, K.-A. (2018). Multivariate analysis of multiple datasets: A practical guide for chemical ecology. Journal of Chemical Ecology,44(3), 215–234. https://doi.org/10.1007/s10886-018-0932-6.
Hren, M., Nikolić, P., Rotter, A., Blejec, A., Terrier, N., Ravnikar, M., et al. (2009). “Bois noir” phytoplasma induces significant reprogramming of the leaf transcriptome in the field grown grapevine. BMC Genomics,10(1), 460. https://doi.org/10.1186/1471-2164-10-460.
Kang, S., & Paul Thomas, C. L. (2016). How long may a breath sample be stored for at − 80 °C? A study of the stability of volatile organic compounds trapped onto a mixed Tenax: Carbograph trap adsorbent bed from exhaled breath. Journal of Breath Research,10(2), 026011. https://doi.org/10.1088/1752-7155/10/2/026011.
Kessler, S., Schaerer, S., Delabays, N., Turlings, T. C. J., Trivellone, V., & Kehrli, P. (2011). Host plant preferences of Hyalesthes obsoletus, the vector of the grapevine yellows disease ‘bois noir’, in Switzerland: Host plant preferences of Hyalesthes obsoletus. Entomologia Experimentalis et Applicata,139(1), 60–67. https://doi.org/10.1111/j.1570-7458.2011.01107.x.
Lavoir, A.-V., Staudt, M., Schnitzler, J.-P., Landais, D., Massol, F., Rocheteau, A., et al. (2009). Drought reduced monoterpene emissions from Quercus ilex trees: Results from a throughfall displacement experiment within a forest ecosystem. Biogeosciences Discussions,6, 863–893.
Linskens, H. F., & Allen, M. S. (Eds.). (1997). Plant volatile analysis. Berlin Milan Paris Santa Clara Singapore Tokyo: Springer.
Lis-Balchin, M. (2002). Lavender: The genus lavandula. London: Taylor & Francis.
Llusia, J., & Penuelas, J. (2000). Seasonal patterns of terpene content and emission from seven Mediterranean woody species in field conditions. American Journal of Botany,87(1), 133–140. https://doi.org/10.2307/2656691.
Mayer, C. J., Vilcinskas, A., & Gross, J. (2008). Pathogen-induced release of plant allomone manipulates vector insect behavior. Journal of Chemical Ecology,34(12), 1518–1522. https://doi.org/10.1007/s10886-008-9564-6.
Ormeño, E., Mévy, J. P., Vila, B., Bousquet-Mélou, A., Greff, S., Bonin, G., & Fernandez, C. (2007). Water deficit stress induces different monoterpene and sesquiterpene emission changes in Mediterranean species. Relationship between terpene emissions and plant water potential. Chemosphere,67(2), 276–284. https://doi.org/10.1016/j.chemosphere.2006.10.029.
Prince, E. K., & Pohnert, G. (2010). Searching for signals in the noise: Metabolomics in chemical ecology. Analytical and Bioanalytical Chemistry,396(1), 193–197. https://doi.org/10.1007/s00216-009-3162-5.
Proffit, M., Schatz, B., Bessière, J.-M., Chen, C., Soler, C., & Hossaert McKey, M. (2008). Signalling receptivity: Comparison of the emission of volatile compounds by figs of Ficus hispida before, during and after the phase of receptivity to pollinators. Balaban, Philadelphia/Rehovot,45, 15–24.
Riolo, P., Minuz, R. L., Anfora, G., Stacconi, M. V. R., Carlin, S., Isidoro, N., et al. (2012). Perception of host plant volatiles in Hyalesthes obsoletus: Behavior, morphology, and electrophysiology. Journal of Chemical Ecology,38(8), 1017–1030. https://doi.org/10.1007/s10886-012-0154-2.
Ross, C. F. (2012). Headspace analysis. In: Comprehensive sampling and sample preparation. Amsterdam: Elsevier, p 27–50.
Sardans, J., Peñuelas, J., & Rivas-Ubach, A. (2011). Ecological metabolomics: Overview of current developments and future challenges. Chemoecology,21(4), 191–225. https://doi.org/10.1007/s00049-011-0083-5.
Sémétey, O., Gaudin, J., Danet, J. L., Salar, P., Theil, S., Fontaine, M., et al. (2018). Lavender decline in France is associated with chronic infection by Lavender-specific strains of Candidatus Phytoplasma solani. Applied and Environmental Microbiology,84(24), e01507. https://doi.org/10.1128/AEM.01507-18.
Shah, J. (2009). Plants under attack: Systemic signals in defence. Current Opinion in Plant Biology,12(4), 459–464. https://doi.org/10.1016/j.pbi.2009.05.011.
Soria, A. C., García-Sarrió, M. J., & Sanz, M. L. (2015). Volatile sampling by headspace techniques. TrAC Trends in Analytical Chemistry,71, 85–99. https://doi.org/10.1016/j.trac.2015.04.015.
Stewart-Jones, A., & Poppy, G. M. (2006). Comparison of glass vessels and plastic bags for enclosing living plant parts for headspace analysis. Journal of Chemical Ecology,32(4), 845–864. https://doi.org/10.1007/s10886-006-9039-6.
Tholl, D., Boland, W., Hansel, A., Loreto, F., Röse, U. S. R., & Schnitzler, J.-P. (2006). Practical approaches to plant volatile analysis. The Plant Journal,45(4), 540–560. https://doi.org/10.1111/j.1365-313X.2005.02612.x.
Yvin, C., Cansière, R., Chuche, J., Thierry, D., Nicolè, F., Kuntzmann, P., et al. (2018). Étude des facteurs sensoriels intervenant dans le choix de plantes et dans le comportement alimentaire des adultes de Hyalesthes obsoletus (Echostol). Innovations Agronomiques,63, 445–456.
Acknowledgements
The authors acknowledge all the partners involved in the EUCLIDE project (No. 154827) co-funded by SPLP Funds (https://www.sauvegarde-lavandes-provence.org/fr/) and SUD PACA region (France). Eric Chaisse is also thanked for his support, expertise and comments on the project.
Author information
Authors and Affiliations
Contributions
ES performed the experiments and analysed the data. ES, FN, XF and TM wrote the article. FN, TC, XF and TM supervised the research work. All authors read and approved the article.
Corresponding author
Ethics declarations
Conflict of interest
The authors have no conflict of interest to declare.
Research involving human or animal rights
This article does not contain any studies with human participants or animals performed by any of the authors.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Stierlin, É., Nicolè, F., Costes, T. et al. Metabolomic study of volatile compounds emitted by lavender grown under open-field conditions: a potential approach to investigate the yellow decline disease. Metabolomics 16, 31 (2020). https://doi.org/10.1007/s11306-020-01654-6
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11306-020-01654-6