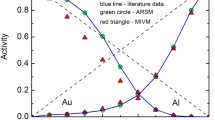
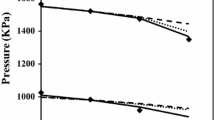
Abstract
Polynomial representation of partial excess Gibbs energy (i.e., activity coefficient) in multicomponent dilute solution has been widely used after Wagner. Although Wagner’s Interaction Parameter Formalism has been known to be only strictly valid at infinite dilute concentration, it has been used often, even at finite concentration, due to its mathematical simplicity. Nevertheless, several attempts have been made to correct the formalism to be thermodynamically consistent at finite concentration. Among those, Unified Interaction Parameter Formalism proposed by Pelton and Bale, which may be considered as an extension of Darken’s quadratic formalism, has obtained much attention. However, there have been much confusion and debate about the way of the correction. Recently, a thermodynamic analysis was reported that there are infinite numbers of ways to correct Wagner’s formalism to be thermodynamically consistent, which may prevent one from using the Unified Interaction Parameter Formalism with confidence. In the present article, the correction to the Wagner’s formalism is discussed by revisiting Darken’s condition of the thermodynamic consistency. It is shown that the correction to the Wagner’s formalism can be made uniquely. It is pointed out that to ensure the thermodynamic consistency among the activity coefficients of all components, Gibbs–Duhem relation and Maxwell relation among all components including solvent–solute, must be obeyed. Derived expressions for activity coefficients of all components by path-independent integration are also shown to be the same as those obtained by differentiating a corresponding integral excess partial Gibbs energy.
Similar content being viewed by others
References
C. Wagner: Thermodynamics of Alloys, Addison-Wesley Pub. Co., Reading, MA, 1951, pp. 51–53.
L. S. Darken: Trans. Metall. Soc. AIME, 1967, vol. 239, pp. 80–89.
L. S. Darken: Trans. Metall. Soc. AIME, 1967, vol. 239, pp. 90–96.
A.D. Pelton and C.W. Bale: Metall. Mater. Trans. A, 1986, vol. 17A, pp. 1211–1215.
C.W. Bale and A.D. Pelton: Metall. Mater. Trans. A, 1990, vol. 21, pp. 1997–2002.
A.D. Pelton: Metall. Mater. Trans. B, 1997, vol. 28B, pp. 869–76.
C.H.P. Lupis and J.F. Elliott: Acta Metall., 1966, vol. 14, pp. 529–38.
M. Margules: Sitz. Akad. Wissen. Wien, Math.–Nat., 1985, vol. 104, pp. 1243–78.
D. Bouchard and C.W. Bale: J. Phase Equil., 1995, vol. 16, pp. 16–23.
D. Bouchard and C.W. Bale: Can. Metall. Q., 1995, vol. 34, pp. 343–346.
H. Li and A. Morris: Metall. Mater. Trans. B, 1997, vol. 28B, pp. 553–62.
I.-H. Jung, S.D. Decterov and A.D. Pelton: Metall. Mater. Trans. B, 2004, vol. 35B, pp. 493–507.
Y.-B. Kang and H.-G. Lee: ISIJ Int., 2004, vol. 44, pp. 1006–15.
S. Srikanth and K.T. Jacob: Metall. Mater. Trans. B, 1988, vol. 19B, pp. 269–75.
J. Hajra: Metall. Mater. Trans. B, 1991, vol. 22B, pp. 583–91.
J. Hajra and M. Frohberg: Metall. Mater. Trans. B, 1992, vol. 23, pp. 23–28.
J. Hajra, S. Reddy, and M. Frohberg: Metall. Mater. Trans. B, 1995, vol. 26B, pp. 495–502.
D. V. Malakhov: Calphad, 2013, vol. 41, pp. 16–19.
K. T. F. Eleno, C. G. Schön: Braz. J. Phys., 2014, vol. 44, pp. 208–214.
C. Wagner: Acta Metall., 1973, vol. 21, pp. 1297–1303.
R. Schmid, J.-C. Lin and Y.A. Chang: Zeit. fuer Metall., 1984, vol. 75, pp. 730–37.
Y.A. Chang, K. Fitzner, and M.-X. Chang: Prog. Mater. Sci., 1988, vol. 32, pp. 97–259.
Y.-B. Kang: Metall. Mater. Trans. B, 2019, vol. 50B, pp. 2942–58.
G. Sigworth and J. F. Elliott: Metal Sci. 1974, vol. 8, pp. 298–310.
C.H.P. Lupis and J.F. Elliott: Acta Metall., 1966, vol. 14, pp. 1019–31.
G. Kaptay: Calphad, 2017, vol. 56, pp. 169–184.
S.-M. Liang, P. Wang, and R. Schmid-Fetzer: Calphad, 2016, vol. 54, pp. 82–96.
Y.-B. Kang and A.D. Pelton: J. Chem. Thermodynamics, 2013, vol. 60, pp. 19–24.
Acknowledgments
The present author would like to express sincere gratitude to Prof. Emeritus A.D. Pelton, Polytechnique de Montréal, Canada, for his critical comment and valuable suggestions during the preparation of this manuscript.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Manuscript submitted October 4, 2019.
Appendices
Appendices
Appendix A
Extension of Darken’s Thermodynamic Consistency into Multicomponent Solution including Solvent
As Gibbs energy, G, is a state function, and its differential is exact, the Maxwell’s relation is obeyed:
where \(n_i\), \(n_j\) are the number of moles of components i and j, and
and
By differentiating G with respect to \(n_j\), while all other \(n_i(i \ne j)\) are kept constant,
The last term in the Eq. [A4] is zero due to the Gibbs–Duhem relation. From the above,
Similarly,
Therefore, the following relationship between activity coefficients of components i and j are obtained:
which is the same as the Eq. [14]. Taking all \(X_i\)’s except for \(X_1\) as independent variables, and using the following derivatives of \(X_k\) with respect to \(n_i\),
the above equation is expressed as:
Similarly,
Therefore, the following equation is obtained which represents the conditions of thermodynamic consistency between two components (i and j) in N-component system, including solvent (\(i=1\) or \(j=1\)) and solute (\(i,j\ne 1\)):
Appendix B
Derivation of \(\ln \gamma _1\) in Multicomponent Solution
From the Eq. [57],
Then,
Therefore by comparison with the Eq. [56],
According to Raoult’s law (\(\frac{\partial \ln \gamma _1}{\partial X_i} \rightarrow 0\) when \(X_i,X_j \rightarrow 0\)), it can be shown that \(c_i = c_j = 0\). Furthermore, by inspection, \(a_{ii}=-\frac{1}{2}\epsilon _{ii}\), \(a_{jj}=-\frac{1}{2}\epsilon _{jj}\), \(b_{ij} = b_{ji} = -\frac{1}{2}\epsilon _{ij}\), etc. Therefore,
Rights and permissions
About this article
Cite this article
Kang, YB. The Uniqueness of a Correction to Interaction Parameter Formalism in a Thermodynamically Consistent Manner. Metall Mater Trans B 51, 795–804 (2020). https://doi.org/10.1007/s11663-020-01792-1
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11663-020-01792-1