Abstract
Objective
To evaluate the most commonly used medications and in-hospital morbidities and mortality in infants born 22–24 weeks of gestation.
Study design
Multicenter retrospective cohort study of infants born 22–24 weeks of gestation (2006–2016), without major congenital anomalies and with available medication data obtained from neonatal intensive care units managed by the Pediatrix Medical Group.
Results
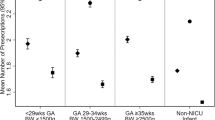
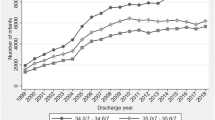
This study included 7578 infants from 195 sites. Median (25th, 75th percentile): birthweight was 610 g (540, 680); the number of distinct medications used was 13 (8, 18); and different antimicrobial exposure was 4 (2, 5). The most common morbidities were BPD (41%) and grade III or IV IVH (20%), and overall survival varied from 46% (2006) to 57% (2016).
Conclusions
A large number of medications were used in periviable infants. There was a high prevalence of in-hospital morbidities, and survival of this population increased over the study period.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
Ecker JL, Kaimal A, Mercer BM, Blackwell SC, deRegnier RA.American College of Obstetricians and Gynecologists and the Society for Maternal–Fetal Medicine, et al. Periviable birth: interim update. Am J Obstet Gynecol. 2016;215:B2–B12.e1.
Raju TN, Mercer BM, Burchfield DJ, Joseph GF. Periviable birth: executive summary of a joint workshop by the Eunice Kennedy Shriver National Institute of Child Health and Human Development, Society for Maternal-Fetal Medicine, American Academy of Pediatrics, and American College of Obstetricians and Gynecologists. J Perinatol. 2014;34:333–42.
Mercer BM. Periviable birth and the shifting limit of biability. Clin Perinatol. 2017;44:283–6.
Brumbaugh JE, Hansen NI, Bell EF, Sridhar A, Carlo WA, Hintz SR, et al. Outcomes of extremely preterm infants with birth weight less than 400 g. JAMA Pediatr. 2019;173:434–45.
Stoll BJ, Hansen NI, Bell EF, Shankaran S, Laptook AR, Walsh MC, et al. Neonatal outcomes of extremely preterm infants from the NICHD Neonatal Research Network. Pediatrics. 2010;126:443–56.
Rysavy MA, Li L, Bell EF, Das A, Hintz SR, Stoll BJ, et al. Between-hospital variation in treatment and outcomes in extremely preterm infants. N Engl J Med. 2015;372:1801–11.
Tucker Edmonds B, McKenzie F, Macheras M, Srinivas SK, Lorch SA. Morbidity and mortality associated with mode of delivery for breech periviable deliveries. Am J Obstet Gynecol. 2015;213:70.e1–2.
Horbar JD, Carpenter JH, Badger GJ, Kenny MJ, Soll RF, Morrow KA, et al. Mortality and neonatal morbidity among infants 501 to 1500 grams from 2000 to 2009. Pediatrics. 2012;129:1019–26.
Younge N, Goldstein RF, Bann CM, Hintz SR, Patel RM, Smith PB, et al. Survival and neurodevelopmental outcomes among periviable infants. N. Engl J Med. 2017;376:617–28.
Clark RH, Bloom BT, Spitzer AR, Gerstmann DR. Reported medication use in the neonatal intensive care unit: data from a large national data set. Pediatrics. 2006;117:1979–87.
Hsieh EM, Hornik CP, Clark RH, Laughon MM, Benjamin DK Jr, Smith PB. Medication use in the neonatal intensive care unit. Am J Perinatol. 2014;31:811–21.
Nau R, Sörgel F, Eiffert H. Penetration of drugs through the blood-cerebrospinal fluid/blood-brain barrier for treatment of central nervous system infections. Clin Microbiol Rev. 2010;23:858–83.
Wu TW, Azhibekov T, Seri I. Transitional hemodynamics in preterm neonates: clinical relevance. Pediatr Neonatol. 2016;57:7–18.
Fernandez E, Perez R, Hernandez A, Tejada P, Arteta M, Ramos JT. Factors and mechanisms for pharmacokinetic differences between pediatric population and adults. Pharmaceutics. 2011;3:53–72.
Crane JM, Magee LA, Lee T, Synnes A, von Dadelszen P, Dahlgren L, et al. Maternal and perinatal outcomes of pregnancies delivered at 23 weeks’ gestation. J Obstet Gynaecol Can. 2015;37:214–24.
Spitzer AR, Ellsbury DL, Handler D, Clark RH. The Pediatrix BabySteps Data Warehouse and the Pediatrix QualitySteps improvement project system-tools for “meaningful use” in continuous quality improvement. Clin Perinatol. 2010;37:49–70.
Olsen IE, Groveman SA, Lawson ML, Clark RH, Zemel BS. New intrauterine growth curves based on United States data. Pediatrics. 2010;125:e214–24.
Trembath A, Hornik CP, Clark R, Smith PB, Daniels J, Laughon M. Comparative effectiveness of surfactant preparations in premature infants. J Pediatr. 2013;163:955–60.e1.
Papile LA, Burstein J, Burstein R, Koffler H. Incidence and evolution of subependymal and intraventricular hemorrhage: a study of infants with birth weights less than 1,500 gm. J Pediatr. 1978;92:529–34.
Nir-Neuman H, Abu-Kishk I, Toledano M, Heyman E, Ziv-Baran T, Berkovitch M. Unlicensed and off-label medication use in pediatric and neonatal intensive care units: no change over a decade. Adv Ther. 2018;35:1122–32.
Polin RA, Carlo WA. Committee on fetus and newborn; American Academy of Pediatrics Surfactant replacement therapy for preterm and term neonates with respiratory distress. Pediatrics. 2014;133:156–63.
American Regent, Inc. Caffeine citrate drug label information. DAILYMED. 2019. https://dailymed.nlm.nih.gov/dailymed/drugInfo.cfm?setid=d17e3be5-514f-454a-8347-d804cd5ec37f. Accessed 5 July 2019.
Mori R, Kusuda S, Fujimura M. Neonatal Research Network Japan. Antenatal corticosteroids promote survival of extremely preterm infants born at 22 to 23 weeks of gestation. J Pediatr. 2011;159:110–4.e1.
Carlo WA, McDonald SA, Fanaroff AA, Vohr BR, Stoll BJ, Ehrenkranz RA, et al. Association of antenatal corticosteroids with mortality and neurodevelopmental outcomes among infants born at 22 to 25 weeks’ gestation. J Am Med Assoc. 2011;306:2348–58.
Ehret DEY, Edwards EM, Greenberg LT, et al. Association of antenatal steroid exposure with survival among infants receiving postnatal life support at 22 to 25 weeks’ gestation. JAMA Netw Open. 2018;1:e183235.
Roberts D, Brown J, Medley N, Dalziel S. Antenatal corticosteroids for accelerating fetal lung maturation for women at risk of preterm birth. Cochrane Database Syst Rev. 2006;3:CD004454.
Travers CP, Clark RH, Spitzer AR, Das A, Garite TJ, Carlo WA. Exposure to any antenatal corticosteroids and outcomes in preterm infants by gestational age: prospective cohort study. BMJ. 2017;356:j1039.
Wapner RJ. Antenatal corticosteroids for periviable birth. Semin Perinatol. 2013;37:410–3.
Tyson JE, Parikh NA, Langer J, Green C, Higgins RD. National Institute of Child Health Human Development Neonatal Research Network Intensive care for extreme prematurity-moving beyond gestational age. N Engl J Med. 2008;358:1672–81.
Kitchen WH, Permezel MJ, Doyle LW, Ford GW, Rickards AL, Kelly EA. Changing obstetric practice and 2-year outcome of the fetus of birth weight under 1000 g. Obstet Gynecol. 1992;79:268–75.
Patel RM, Kandefer S, Walsh MC, Bell EF, Carlo WA, Laptook AR, et al. Causes and timing of death in extremely premature infants from 2000 through 2011. N Engl J Med. 2015;372:331–40.
Kawakita T, Reddy UM, Grantz KL, Landy HJ, Desale S, Igbal SN. Maternal outcomes associated with early preterm cesarean delivery. Am J Obstet Gynecol. 2017;216:312.e1–9.
Malloy MH. Impact of cesarean section on neonatal mortality rates among very preterm infants in the United States, 2000–2003. Pediatrics. 2008;122:285–92.
Deulofeut R, Sola A, Lee B, Buchter S, Rahman M, Rogido M. The impact of vaginal delivery in premature infants weighing less than 1,251 grams. Obstet Gynecol. 2005;105:525–31.
Holzer I, Lehner R, Ristl R, Husslein PW, Berger A, Farr A. Effect of delivery mode on neonatal outcome among preterm infants: an observational study. Wien Klin Wochenschr. 2017;129:612–7.
Bottoms SF, Paul RH, Iams JD, Mercer BM, Thom EA, Roberts JM, et al. Obstetric determinants of neonatal survival: influence of willingness to perform cesarean delivery on survival of extremely low-birth-weight infants. National Institute of Child Health and Human Development Network of Maternal-Fetal Medicine Units. Am J Obstet Gynecol. 1997;176:960–6.
Patel RM, Rysavy MA, Bell EF, Tyson JE. Survival of infants born at periviable gestational ages. Clin Perinatol. 2017;44:287–303.
Stoll BJ, Hansen NI, Bell EF, Walsh MC, Carlo WA, Shankaran S, et al. Trends in care practices, morbidity, and mortality of extremely preterm neonates, 1993–2012. J Am Med Assoc. 2015;314:1039–51.
Ishii N, Kono Y, Yonemoto N, Kusuda S, Fujimura M. Neonatal Research Network, Japan Outcomes of infants born at 22 and 23 weeks’ gestation. Pediatrics. 2013;132:62–71.
Gunasekera H, Isaacs D. Outcome for 22–24 week gestation newborns. J Paediatr Child Health. 2017;53:1240.
Moore T, Hennessy EM, Myles J, Johnson SJ, Draper ES, Costeloe KL, et al. Neurological and developmental outcome in extremely preterm children born in England in 1995 and 2006: the EPICure studies. BMJ. 2012;345:e7961.
American College of Obstetricians and Gynecologists; Society for Maternal-Fetal Medicine. Obstetric care consensus no. 6: periviable birth. Obstet Gynecol. 2017;130:e187–99.
Brunkhorst J, Weiner J, Lantos J. Infants of borderline viability: the ethics of delivery room care. Semin Fetal Neonatal Med. 2014;19:290–5.
Guillén U, Weiss EM, Munson D, Maton P, Jefferies A, Norman M, et al. Guidelines for the management of extremely premature deliveries: a systematic review. Pediatrics. 2015;136:343–50.
Chervenak FA, McCullough LB. Ethical issues in periviable birth. Semin Perinatol. 2013;37:422–5.
Edmonds BT, McKenzie F, Hendrix KS, Perkins SM, Zimet GD. The influence of resuscitation preferences on obstetrical management of periviable deliveries. J Perinatol. 2015;35:161–6.
Allen KM, Smith B, Iliev I, Evans J, Werthammer J. Short term cost of care for the surviving periviable neonate. J Neonatal Perinat Med. 2017;10:191–4.
Jadcherla SR, Khot T, Moore R, Malkar M, Gulati IK, Slaughter JL. Feeding methods at discharge predict long-term feeding and neurodevelopmental outcomes in preterm infants referred for gastrostomy evaluation. J Pediatr. 2017;181:125–30.
Batton DG. Committee on Fetus and Newborn Clinical report—antenatal counseling regarding resuscitation at an extremely low gestational age. Pediatrics. 2009;124:422–7.
Acknowledgements
PTN Steering Committee Members: Daniel K. Benjamin Jr., Christoph Hornik, Kanecia Zimmerman, Phyllis Kennel, and Rose Beci, Duke Clinical Research Institute, Durham, NC, USA; Chi Dang Hornik, Duke University Medical Center, Durham, NC, USA; Gregory L. Kearns, Independent; Matthew Laughon, University of North Carolina at Chapel Hill, Chapel Hill, NC USA; Ian M. Paul, Penn State College of Medicine, Hershey, PA, USA; Janice Sullivan, University of Louisville, Louisville, KY, USA; Kelly Wade, Children’s Hospital of Philadelphia, Philadelphia, PA, USA; Paula Delmore, Wichita Medical Research and Education Foundation, Wichita, KS, USA. The Eunice Kennedy Shriver National Institute of Child Health and Human Development (NICHD): Perdita Taylor-Zapata and June Lee. The Emmes Company, LLC (Data Coordinating Center): Ravinder Anand, Gaurav Sharma, Gina Simone, Kim Kaneshige, and Lawrence Taylor. PTN Publications Committee: Chaired by Thomas Green, Ann & Robert H. Lurie Children’s Hospital of Chicago, Chicago, IL, USA
Funding
This work was funded under National Institute of Child Health and Human Development (NICHD) contract (HHSN275201000003I) for the Pediatric Trials Network (PI Daniel K. Benjamin Jr.). The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Institutes of Health. This work was also supported by Duke Clinical Research Institute’s R25 Summer Training in Academic Research (STAR) Program (grant #5R25HD076475–07).
Author information
Authors and Affiliations
Contributions
MP-D had full access to all of the data in the study and takes responsibility for the integrity of the data and the accuracy of the data analysis and contributed to the conception and design of the study, the data analysis, the data interpretation, the manuscript drafting, and the critical revision of the manuscript. NY contributed to the data analysis and interpretation, the manuscript drafting, and the critical revision of the manuscript. DKB contributed to the data interpretation, the manuscript drafting, and the critical revision of the manuscript. KL contributed to the data interpretation, the manuscript drafting, and the critical revision of the manuscript. CH contributed to the data interpretation, the manuscript drafting, and the critical revision of the manuscript. KH contributed to the data interpretation, the manuscript drafting, and the critical revision of the manuscript. JM contributed to the data interpretation, the manuscript drafting, and the critical revision of the manuscript. AW contributed to the data interpretation, the manuscript drafting, and the critical revision of the manuscript. KOZ contributed to the data interpretation, the manuscript drafting, and the critical revision of the manuscript. KA contributed to the data interpretation, the manuscript drafting, and the critical revision of the manuscript. RGG contributed to the conception and design of the study, the supervision, data interpretation, and the critical revision of the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
NY received support from the NIH (K23DK120960). MP-D, DKB, KL, CH, KH, JM, AW, KOZ, KA, and RGG report no relevant disclosures.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
About this article
Cite this article
Puia-Dumitrescu, M., Younge, N., Benjamin, D.K. et al. Medications and in-hospital outcomes in infants born at 22–24 weeks of gestation. J Perinatol 40, 781–789 (2020). https://doi.org/10.1038/s41372-020-0614-4
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41372-020-0614-4
This article is cited by
-
Neuroprotection care bundle implementation is associated with improved long-term neurodevelopmental outcomes in extremely premature infants
Journal of Perinatology (2022)
-
Drug utilisation in neonatal units in England and Wales: a national cohort study
European Journal of Clinical Pharmacology (2022)
-
Respiratory, growth, and survival outcomes of infants with tracheostomy and ventilator dependence
Pediatric Research (2021)