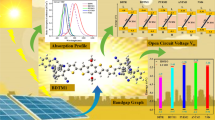
Abstract
This study was aimed at investigating the effect of electron donor and acceptor groups on electrical and optical properties of pi-conjugated [26] annulene. The results of the study showed that almost all selected substitutions reduced the value of \(E_{\mathrm{g}}\) compared with pristine annulene. Calculated dipole moments for the sandwiched forms of annulene are noticeably greater than those of its pristine form, of which the ED2–ANN–QB2 had the maximum value for dipole moment. It was found that the influence of electron donor and acceptor groups on annulene optical properties is very significant. More optical activity improvement was seen in the case of using QB2 and QB3 in one side and other electron donor groups in opposite side of [26] annulene molecule. The higher optical activity of these sandwiched molecules corresponds to higher electron transfer in them. The results of this research may be useful in designing new photosensitizers.













Similar content being viewed by others
References
Adamson A W, Kalyanasundaram K and Grätzel M 1993 Photosensitization and photocatalysis using inorganic and organometallic compounds (Netherlands: Springer Science Business Media B.V.)
Zhang P, Wang M, Li C, Li X, Dong J and Sun L 2010 Chem. Commun. 46 8806
Pfeffer M G, Kowacs T, Wächtler M, Guthmuller J, Dietzek B, Vos J G et al 2015 Angew. Chem. 54 6627
Ravve A 2006 Light-associated reactions of synthetic polymers (New York, NY: Springer)
Wu W, Mao D, Xu S, Kenry, Hu F, Li X et al 2018 Chem 4 1937
Whitehead K and Hedges J I 2005 J. Photochem. Photobiol. B Biol. 80 115
Koh P W, Hatta M H M, Ong S T, Yuliati L and Lee S L 2017 J. Photochem. Photobiol. B Biol. 332 215
Rühle S, Shalom M and Zaban A 2010 ChemPhysChem 11 2290
Huizhi Z, Wu L, Gao Y and Ma T 2011 J. Photochem. Photobiol. A Chem. 219 188
Yamazaki E, Murayama M, Nishikawa N, Hashimoto N, Shoyama M and Kurita O 2007 J. Sol. Energy 81 512
Boyo A O, Shitta M B O, Oluwa T and Adeola S 2012 Trends Appl. Sci. Res. 7 558
Yogo T, Urano Y, Ishitsuka Y, Maniwa F and Nagano T 2005 J. Am. Chem. Soc. 127 12162
Lissi E A, Encinas M V, Lemp E and Rubio M A 1993 Chem. Rev. 93 699
Kou J, Dou D and Yang L 2017 Oncotarget 8 81591
Abrahamse H and Hamblin M R 2016 Biochem. J. 473 347
Ekrami S and Shamlouei H R 2018 Chem. Phys. Lett. 709 26
Becke A D 1988 Phys. Rev. A 38 3098
Lee C, Yang W and Parr R G 1988 Phys. Rev. B 37 785
Frisch M J, Trucks G W, Schlegel H B, Scuseria G E, Robb M A, Cheeseman J R et al 2009 GAUSSIAN 09 (Wallingford, CT: Gaussian, Inc.)
O’boyle N M, Tenderholt A L and Langner K M 2008 J.Comput. Chem.29 839
Runge E and Gross E K 1984 Phys. Rev. Lett.52 997
Gross E and Kohn W 1985 Phys. Rev. Lett.55 2850
Casida M E, Jamorski C, Casida K C and Salahub D R 1998 J. Phys. Chem.108 4439
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Atyabi, S.M., Shamlouei, H.R., Roozbahani, G.M. et al. Tuning the electronic and optical properties of [26] annulene by electron donor and acceptor groups (push–pull system). Bull Mater Sci 43, 72 (2020). https://doi.org/10.1007/s12034-019-2007-x
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s12034-019-2007-x