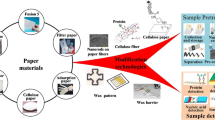
Abstract
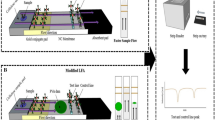
Green/sustainable cellulose paper-based platforms at point-of-care testing have received much interest in biomedical fields at resource-limited settings. In such platforms, biomolecules would have to be immobilized onto paper substrate to achieve detection. Therefore, effective molecule immobilization is critical for the performance of paper-based testing, especially the detection sensitivity. Although various methods have been developed to improve the detection sensitivity, the immobilization capacity of biomolecules has not been considered yet. In this study, we developed a facile method for modifying paper materials by incorporating chitosan into NC membrane, which has positive effect on the pore size, porosity, and surface groups of NC membrane, hence, enhancing the immobilization capacity of biomolecules on paper substrate. We demonstrated the effectiveness of the above mentioned approach by using nucleic acid lateral flow assay (NALFAs) as a model paper-based platform; in comparison with the unmodified NALFAs, the results showed ten-fold increase in the detection sensitivity for HBV. We envision that this facile modification method will have a great potential for developing other highly sensitive paper-based detecting platforms.
Graphic abstract







Similar content being viewed by others
References
Abou Hammad AB, Elnahrawy AM, Youssef AM, Youssef AM (2019) Sol gel synthesis of hybrid chitosan/calcium aluminosilicate nanocomposite membranes and its application as support for CO2 sensor. Int J Biol Macromol 125:503–509. https://doi.org/10.1016/j.ijbiomac.2018.12.077
Barros RJ, Wehtje E, Garcia FAP, Adlercreutz P (2009) Physical characterization of porous materials and correlation with the activity of immobilized enzyme in organic medium. Biocatal Biotransfor 16:67–85. https://doi.org/10.3109/10242429809040111
Bin L, Wen HM, Wang H, Wu H, Tyagi M, Yildirim T, Zhou W, Chen B (2014) A porous metal–organic framework with dynamic pyrimidine group exhibiting record high methane storage working capacity. J Am Chem Soc 136:6207–6210
Branca C, D’Angelo G, Crupi C, Khouzami K, Rifici S, Ruello G, Wanderlingh U (2016) Role of the OH and NH vibrational groups in polysaccharide-nanocomposite interactions: a FTIR-ATR study on chitosan and chitosan/clay films. Polymer-Basel 99:614–622
Chiu RY et al (2014) Simultaneous concentration and detection of biomarkers on paper. Lab Chip 14:3021–3028. https://doi.org/10.1039/c4lc00532e
Choi JR et al (2016) An integrated paper-based sample-to-answer biosensor for nucleic acid testing at the point of care. Lab Chip 16:611–621. https://doi.org/10.1039/c5lc01388g
Choi JR et al (2017) Advances and challenges of fully integrated paper-based point-of-care nucleic acid testing. TrAC-Trend Anal Chem 93:37–50. https://doi.org/10.1016/j.trac.2017.05.007
Freedman DL, Cashwell JM, Kim BJ (2002) Biotransformation of explosive-grade nitrocellulose under denitrifying and sulfidogenic conditions. Waste Manage 22:283–292
Fu X, Cheng Z, Yu J, Choo P, Chen L, Choo J (2016) A SERS-based lateral flow assay biosensor for highly sensitive detection of HIV-1 DNA. Biosens Bioelectron 78:530–537. https://doi.org/10.1016/j.bios.2015.11.099
Gabriel EF, Garcia PT, Cardoso TM, Lopes FM, Martins FT, Coltro WK (2016) Highly sensitive colorimetric detection of glucose and uric acid in biological fluids using chitosan-modified paper microfluidic devices. Analyst 141:4749–4756. https://doi.org/10.1039/c6an00430j
Hartmann M, Kostrov X (2013) Immobilization of enzymes on porous silicas–benefits and challenges. Chem Soc Rev 42:6277–6289. https://doi.org/10.1039/c3cs60021a
Hu J, Wang L, Li F, Han YL, Lin M, Lu TJ, Xu F (2013) Oligonucleotide-linked gold nanoparticle aggregates for enhanced sensitivity in lateral flow assays. Lab Chip 13:4352–4357. https://doi.org/10.1039/c3lc50672j
Hu J et al (2017) Multiple test zones for improved detection performance in lateral flow assays. Sens Actuators B Chem 243:484–488. https://doi.org/10.1016/j.snb.2016.12.008
Jimenez-Cervantes E, López-Barroso J, Martínez-Hernández AL, Velasco-Santos C (2016) Graphene-based materials functionalization with natural polymeric biomolecules. Recent Adv Gr Res IntechOpen. https://doi.org/10.5772/64001
Kang YDLWYLHHCCFYJ, Yu CMLL (2018) Chitosan functionalization to prolong stable hydrophilicity of cotton thread for thread-based analytical device application. Cellulose 25:4831–4840. https://doi.org/10.1007/s10570-018-1891-3
Katis IN, He PJW, Eason RW, Sones CL (2018) Improved sensitivity and limit-of-detection of lateral flow devices using spatial constrictions of the flow-path. Biosens Bioelectron 113:95–100. https://doi.org/10.1016/j.bios.2018.05.001
Kong F, Hu YF (2012) Biomolecule immobilization techniques for bioactive paper fabrication. Anal Bioanal Chem 403:7–13. https://doi.org/10.1007/s00216-012-5821-1
Książczak ARA, Zielenkiewicz T (2003) Nitrocellulose porosity-thermoporometry. J Therm Anal Calorim 74:559–568
Liu H, Wang H, Yang W, Cheng Y (2012) Disulfide cross-linked low generation dendrimers with high gene transfection efficacy, low cytotoxicity, and low cost. J Am Chem Soc 134:17680–17687. https://doi.org/10.1021/ja307290j
Liu M-M et al (2019) A colorimetric assay for sensitive detection of hydrogen peroxide and glucose in microfluidic paper-based analytical devices integrated with starch-iodide-gelatin system. Talanta 200:511–517. https://doi.org/10.1016/j.talanta.2019.03.089
Lu X et al (2018) Improved performance of lateral flow immunoassays for alpha-fetoprotein and vanillin by using silica shell-stabilized gold nanoparticles. Microchim Acta 186:2. https://doi.org/10.1007/s00604-018-3107-9
Marín-Barroso E et al (2018) Paper based analytical device modified with nanoporous material for the fluorescent sensing of gliadin content in different food samples. Microchem J 142:78–84. https://doi.org/10.1016/j.microc.2018.06.005
Moghadam BY, Connelly KT, Posner JD (2014) Isotachophoretic preconcenetration on paper-based microfluidic devices. Anal Chem 86:5829–5837. https://doi.org/10.1021/ac500780w
Mohite BV, Patil SV (2014) Bacterial cellulose of Gluconoacetobacter hansenii as a potential bioadsorption agent for its green environment applications. J Biomater Sci Polymer Ed 25:2053–2065. https://doi.org/10.1080/09205063.2014.970063
Mu M, Li X, Tong A, Guo G (2019) Multi-functional chitosan-based smart hydrogels mediated biomedical application. Expert Opin Drug Del 16:1–12. https://doi.org/10.1080/17425247.2019.1580691
Preechakasedkit P, Siangproh W, Khongchareonporn N, Ngamrojanavanich N, Chailapakul O (2018) Development of an automated wax-printed paper-based lateral flow device for alpha-fetoprotein enzyme-linked immunosorbent assay. Biosens Bioelectron 102:27–32. https://doi.org/10.1016/j.bios.2017.10.051
Ren X, Lu P, Feng R, Zhang T, Zhang Y, Wu D, Wei Q (2018) An ITO-based point-of-care colorimetric immunosensor for ochratoxin a detection. Talanta 188:593–599. https://doi.org/10.1016/j.talanta.2018.06.004
Roy RD, Rosenmund C, Stefan MI (2017) Cooperative binding mitigates the high-dose hook effect. BMC Syst Biol 11:74. https://doi.org/10.1186/s12918-017-0447-8
Shen SS, Yang JJ, Liu CX, Bai RB (2017) Immobilization of copper ions on chitosan/cellulose acetate blend hollow fiber membrane for protein adsorption. RSC Adv 7:10424–10431. https://doi.org/10.1039/c7ra00148g
Sicard C, Brennan JD (2013) Bioactive paper: biomolecule immobilization methods and applications in environmental monitoring. MRS Bull 38:331–334. https://doi.org/10.1557/mrs.2013.61
Tang R et al (2016) Improved sensitivity of lateral flow assay using paper-based sample concentration technique. Talanta 152:269–276. https://doi.org/10.1016/j.talanta.2016.02.017
Tang R et al (2017a) Improved analytical sensitivity of lateral flow assay using sponge for HBV nucleic acid detection. Sci Rep 7:1360. https://doi.org/10.1038/s41598-017-01558-x
Tang R et al (2017b) A fully disposable and integrated paper-based device for nucleic acid extraction, amplification and detection. Lab Chip 17:1270–1279. https://doi.org/10.1039/c6lc01586g
Tang R et al (2017c) Paper-based device with on-chip reagent storage for rapid extraction of DNA from biological samples. Microchim Acta 184:2141–2150. https://doi.org/10.1007/s00604-017-2225-0
Tsai TT, Huang TH, Chen CA, Ho NY, Chou YJ, Chen CF (2018) Development a stacking pad design for enhancing the sensitivity of lateral flow immunoassay. Sci Rep 8:17319. https://doi.org/10.1038/s41598-018-35694-9
Verma MS, Tsaloglou M-N, Sisley T, Christodouleas D, Chen A, Milette J, Whitesides GM (2018) Sliding-strip microfluidic device enables ELISA on paper. Biosens Bioelectron 99:77–84. https://doi.org/10.1016/j.bios.2017.07.034
Wang S, Ge L, Song X, Yu J, Ge S, Huang J, Zeng F (2012) Paper-based chemiluminescence ELISA: lab-on-paper based on chitosan modified paper device and wax-screen-printing. Biosens Bioelectron 31:212–218. https://doi.org/10.1016/j.bios.2011.10.019
Wang P, Ge L, Ge S, Yu J, Yan M, Huang J (2013) A paper-based photoelectrochemical immunoassay for low-cost and multiplexed point-of-care testing. Chem Commun (Camb) 49:3294–3296. https://doi.org/10.1039/c3cc00149k
Wang W et al (2019) Reusable surface plasmon resonance sensor for rapid detection of Cu2 + based on modified-chitosan thin film as an active layer. Sens Actuat A Phys 286:59–67. https://doi.org/10.1016/j.sna.2018.12.025
Wong SY, Cabodi M, Rolland J, Klapperich CM (2014) evaporative concentration on a paper-based device to concentrate analytes in a biological fluid. Anal Chem 86:11981–11985. https://doi.org/10.1021/ac503751a
Yamada K, Henares TG, Suzuki K, Citterio D (2015) Paper-based inkjet-printed. microfluidic. analytical device. Angew Chem Int Ed 54:2-19. https://doi.org/10.1002/anie.201411508, https://doi.org/10.1002/ange.201411508
Yetisen AK, Akram MS, Lowe CR (2013) Paper-based microfluidic point-of-care diagnostic devices. Lab Chip 13:2210–2251. https://doi.org/10.1039/c3lc50169h
Yew CT, Azari P, Choi JR, Li F, Pingguan-Murphy B (2018) Electrospin-coating of nitrocellulose membrane enhances sensitivity in nucleic acid-based lateral flow assay. Anal Chim Acta 1009:81–88. https://doi.org/10.1016/j.aca.2018.01.016
Zhang H, Lei Z, Tian R, Wang Z (2018) Polyamidoamine starburst dendrimer-activated chromatography paper-based assay for sensitive detection of telomerase activity. Talanta 178:116–121. https://doi.org/10.1016/j.talanta.2017.09.034
Acknowledgments
This work was supported by the National Natural Science Foundation of China (21808132), the China Postdoctoral Science Foundation (2018M633525), the Project of Shaanxi Provincial Education department (18JK0096), the Natural Science Basic Research Plan of Shaanxi Province (2019JQ-517), the Opening Project of National Experimental Teaching Demonstration Center of Light Chemical Engineering (2018QGSJ02-10), the High-level Foreign Experts Project (GD20186100425), “Biomass Chemistry and Materials” Academician Workstation Project in SUST (134090002).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
All authors declare that they have no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Tang, R.H., Li, M., Liu, L.N. et al. Chitosan-modified nitrocellulose membrane for paper-based point-of-care testing. Cellulose 27, 3835–3846 (2020). https://doi.org/10.1007/s10570-020-03031-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10570-020-03031-x