Abstract
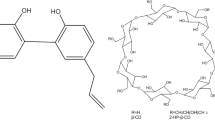
In this study, the antifungal compound natamycin was encapsulated in methyl-β-cyclodextrin (heptakis(2,6-di-O-methyl)-β-cyclodextrin, Me-β-CD) to improve its aqueous solubility and stability. The aqueous solubilities of natamycin in the presence of β-CD, 2-hydroxypropyl-β-CD, 6-O-α-maltosyl-β-CD, and Me-β-CD were compared. The Me-β-CD showed the best result to increase the solubility of natamycin in aqueous. The pH stability of natamycin was improved by the formation of inclusion complex with Me-β-CD, especially at acidic conditions. The degradation of natamycin under UV-light exposure followed first-order kinetics with half-life times (t1/2) of 59.2 and 157.5 min in pure form and Me-β-CD inclusion complex, respectively. The in vitro antifungal activities of natamycin/Me-β-CD complex against Aspergillus niger food pathogen were evaluated. The results demonstrated that the natamycin/Me-β-CD complex could effectively improve the aqueous solubility and photostability of natamycin without compromising in antifungal activities. Finally, the molecular inclusion mechanisms and geometrical configurations of the natamycin/Me-β-CD complex were studied using molecular dynamics simulations. This research may lead to the development of more effective inclusion-based delivery systems to encapsulate and protect lipophilic antimicrobial agents for food applications.






Similar content being viewed by others
References
C.P.O. Resa, R.J. Jagusb, L.N. Gerschenson, Food Control 35, 101–108 (2014)
J.L. Koontz, J.E. Marcy, J. Agric. Food Chem. 57, 7106–7110 (2003)
E. Cevher, D. Şensoy, M. Zloh, L. Mülazımoğlu, J. Pharm. Sci. 97, 4319–4335 (2008)
S. Ho, Y.Y. Thoo, D.J. Young, L.F. Siow, LWT–Food Sci. Technol. 86, 555–565 (2017)
E.J. Pérez-Monterroza, A.M. Chaux-Gutiérrez, C.M.L. Franco, V.R. Nicoletti, Food Biophys. 13, 343–352 (2018)
S. Ho, Y.Y. Thoo, D.J. Young, L.F. Siow, LWT–Food Sci. Technol. 100, 368–373 (2019)
H. Pu, Q. Sun, P. Tang, L. Zhao, Q. Li, Y. Liu, Food Chem. 260, 183–192 (2018)
A. Celebioglu, Z.I. Yildiz, T. Uyar, J. Agric. Food Chem. 66, 457–466 (2018)
J.L. Koontz, J.E. Marcy, W.E. Barbeau, S.E. Duncan, J. Agric. Food Chem. 51, 7111–7114 (2003)
Y.F. Li, J. Jin, Q. Guo, Y.M. Ha, Q.P. Li, Carbohydr. Polym. 125, 288–300 (2015)
L. Szente, J. Szejtli, Adv. Drug Deliv. Rev. 36, 17–28 (1999)
L. Angiolini, M. Agnes, B. Cohen, K. Yannakopoulou, A. Douhal, Int. J. Pharm. 531(2), 668–675 (2017)
A. Duarte, A. Martinho, A. Luis, A. Figueiras, M. Oleastro, F.C. Domingues, LWT–Food Sci. Technol. 63, 1254–1260 (2015)
L.L. Wang, S.S. Li, P.X. Tang, J. Yan, K.L. Xu, H. Li, Carbohydr. Polym. 129, 9–16 (2015)
B.G. Liu, Y. Li, H.C. Xiao, Y.L. Liu, H.Z. Mo, H.J. Ma, J. Food Sci. 80(6), C1156–C1161 (2015)
B.G. Liu, W. Li, J. Zhao, Y. Liu, X.A. Zhu, G.Z. Liang, Food Chem. 141(2), 900–906 (2013)
Q. Ding, X. Cui, G.H. Xu, C.H. He, K.J. Wu, AICHE J. 64, 4080–4088 (2018)
S. Fang, H.J. Xie, H.Y. Chen, L. Wang, S.Y. Tian, J. Chem. Thermodyn. 113, 144–150 (2017)
J. He, C. Christophe, X.G. Shao, W.S. Cai, J. Phys. Chem. C 118, 24173–24180 (2014)
Y.M. Te Welscher, H.H. Ten Napel, M.M. Balagué, C.M. Souza, H. Riezman, B. De Kruijff, E. Breukink, J. Biol. Chem. 283, 6393–6401 (2008)
J.P. Fan, T.T. Yuan, J.X. Yu, X.H. Zhang, Y.H. Cao, J. Chem. Eng. Data 63, 642–650 (2018)
W. Khuntawe, M. Karttunend, J. Wong-Ekkabut, Phys. Chem. Chem. Phys. 19, 24219–24229 (2017)
P.M. Kells, H. Ouellet, J. Santos-Aberturas, J.F. Aparicio, L.M. Podust, Chem. Biol. 17(8), 841–851 (2010)
M.J. Abraham, T. Murtola, R. Schulz, S. Páll, J.C. Smith, B. Hess, SoftwareX 1(2), 19–25 (2015)
X. Daura, A.E. Mark, W.F. van Gunsteren, J. Comput. Chem. 19, 535–547 (1998)
B. Hess, C. Kutzner, D. van der Spoel, E. Lindahl, J. Chem. Theory Comput. 4, 435–447 (2008)
G. Bussi, D. Donadio, M. Parrinello, J. Chem. Phys. 126, 014101 (2007)
M. Parrinello, A. Rahman, J. Appl. Phys. 52, 7182–7190 (1981)
A.R. Green, J.K. Guillory, J. Pharm. Sci. 78, 427–431 (1989)
G.J. You, L.L. Sun, X.X. Cao, H.H. Li, M. Wang, Y.N. Liu, X.L. Ren, LWT–Food Sci. Technol. 94, 172–177 (2018)
C.Q. Sun, P. Lalitha, N.V. Prajna, R. Karpagam, M. Geetha, K.S. O'Brien, Ophthalmology 121, 1495–1500 (2014)
T. Loftsson, M.E. Brewster, J. Pharm. Sci. 101, 3019–3032 (2012)
J.M. Hamilton-Miller, J. Pharm. Pharmacol. 25, 401–407 (1973)
Z. Shao, S. Fang, Y. Li, J. Chen, Y. Meng, Int. J. Biol. Macromol. 118(Pt B), 2208–2215 (2018)
Acknowledgments
The authors acknowledge support from the Key Research and Development Project of Zhejiang Province, China (2019C02088).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare no conflict of interest.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Fang, S., Peng, X., Liang, X. et al. Enhancing Water Solubility and Stability of Natamycin by Molecular Encapsulation in Methyl-β-Cyclodextrin and its Mechanisms by Molecular Dynamics Simulations. Food Biophysics 15, 188–195 (2020). https://doi.org/10.1007/s11483-019-09620-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11483-019-09620-z