Abstract
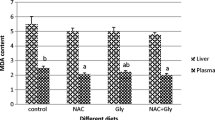
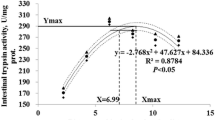
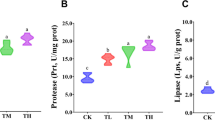
This study was conducted to investigate the effects of diet 7.5 g/kg α-ketoglutarate (AKG) on the growth performance, antioxidant defense system, digestive enzymes, and immune response of grass carp (Ctenopharyngodon idellus). A total of 400 grass carp with an average body weight 10.81 ± 0.68 g was randomly allocated into 2 groups with 4 replicates of 50 fish respectively. The experiment was conducted in net cages (1.5 m × 1.5 m × 1.5 m) suspended in an indoor cement pool. Fish were fed a basic diet containing either 0 (control) or 7.5 g/kg AKG (supplemented diet). The experiment lasted for 8 weeks (56 days). Results indicated that compared with the control group, the final weight (FW), weight gain rate (WGR), specific growth rate (SGR), and protein efficiency ratio (PER) in the AKG group were increased significantly (P < 0.05). However, the feed conversion ratio (FCR) was decreased significantly (P < 0.05). The 7.5 g/kg AKG supplementation significantly increased the activities of glutamine synthetase (GS), glutathione peroxidase (GSH-Px), catalase (CAT), total superoxide dismutase (T-SOD), and hexokinase (HK), as well as the concentrations of glutathione (GSH), total antioxidant capacity (T-AOC), and complement 3 (C3) in blood (P < 0.05), while significantly decreased the concentrations of malondialdehyde (MDA) and hemoglobin (Hb) (P < 0.05). The GS activity and GSH concentration in hepatopancreas were increased significantly (P < 0.05), whereas the glycogen concentration in hepatopancreas, and the glycogen concentration and GS activity in the muscle were significantly decreased (P < 0.05). In addition, 7.5 g/kg AKG supplementation significantly increased the concentration of GSH and the activities of amylase, protease, and lipase in fore-gut, alkaline phosphates (ALP) in the mid-gut, and Na-ATP and Ca-ATP in the gill (P < 0.05), as well as γ-glutamyl transpeptidase (γ-GT) both in fore-gut and mid-gut (P < 0.05), whereas the activity of acid phosphatase (ACP) in the mid-gut was decreased significantly (P < 0.05). In conclusion, diet 7.5 g/kg AKG supplementation in grass carp may improve the growth performance and immune response and play crucial roles in regulating the activities of GS, antioxidant defense system, and digestive enzymes.
Similar content being viewed by others
Abbreviations
- ROS:
-
Reactive oxygen species
- ACP:
-
Acid phosphatase
- ALP:
-
Alkaline phosphates
- Gln:
-
Glutamine
- GS:
-
Glutamine synthetase
- AKG:
-
α-Ketoglutarate
- TCA:
-
Tricarboxylic acid cycle
- IW:
-
Initial weight
- FW:
-
Final weight
- WGR:
-
Weight gain rate
- SGR:
-
Specific growth rate
- FCR:
-
Feed conversion ratio
- PER:
-
Protein efficiency ratio
- TP:
-
Total protein
- ALB:
-
Albumin
- LSZ:
-
Lysozyme
- C3:
-
Complement 3
- γ-GT:
-
γ-Glutamyl transpeptidase
- ALT:
-
Alanine aminotransferase
- AST:
-
Aspartate aminotransferase
- GSH:
-
Glutathione
- GST:
-
Glutathione-S-transferase
- GSH-Px:
-
Glutathione peroxidase
- SOD:
-
Superoxide dismutase
- CAT:
-
Catalase
- T-AOC:
-
Total antioxidant capacity
- MDA:
-
Malondialdehyde
- NO:
-
Nitric oxide
- Hb:
-
Hemoglobin
- HK:
-
Hexokinase
- INS:
-
Insulin
- ADA:
-
Adenosine deaminase
References
Abhijith BD, Ramesh M, Poopal RK (2016) Responses of metabolic and antioxidant enzymatic activities in gill, liver and plasma of Catla catla during methyl parathion exposure. J Basic Appl Zool 77(C):31–40. https://doi.org/10.1016/j.jobaz.2015.11.002
Ai F, Wang L, Li J, Xu Q (2019) Effects of a-ketoglutarate (AKG) supplementation in low phosphorous diets on the growth, phosphorus metabolism and skeletal development of juvenile mirror carp (Cyprinus carpio). Aquaculture 507:393–401. https://doi.org/10.1016/j.aquaculture.2019.03.047
Anderson PM, Broderius MA, Fong KC, Tsui KNT, Chew SF, Ip YK (2002) Glutamine synthetase expression in liver, muscle, stomach and intestine of Bostrichthys sinensis in response to exposure to a high exogenous ammonia concentration. J Exp Biol 205(14):2053–2065
Bondad-Reantaso MG, Subasinghe RP, Arthur JR, Ogawa K, Chinabut S, Adlard R, Tan Z, Shariff M (2005) Disease and health management in Asian aquaculture. Vet Parasitol 132(3-4):249–272. https://doi.org/10.1016/j.vetpar.2005.07.005
Caballero-Solares A, Viegas I, Salgado MC, Siles AM, Sáez A, Metón I, Baanante IV, Fernández F (2015) Diets supplemented with glutamate or glutamine improve protein retention and modulate gene expression of key enzymes of hepatic metabolism in gilthead seabream (Sparus aurata) juveniles. Aquaculture 444:79–87. https://doi.org/10.1016/j.aquaculture.2015.03.025
Carey BW, Finley LW, Cross JR, Allis CD, Thompson CB (2015) Intracellular alpha-ketoglutarate maintains the pluripotency of embryonic stem cells. Nature 518(7539):413–416. https://doi.org/10.1038/nature13981
Chen JS, Wu F, Yang HS, Li FN, Jiang Q, Liu SJ, Kang BJ, Li S, Adebowale TO, Huang N, Li H, Yin YL, Fu CX, Yao K (2017) Growth performance, nitrogen balance, and metabolism of calcium and phosphorus in growing pigs fed diets supplemented with alpha-ketoglutarate. Anim Feed Sci Technol 226:21–28. https://doi.org/10.1016/j.anifeedsci.2016.12.013
Cock J, Gitterle T, Salazar M, Rye M (2009) Breeding for disease resistance of Penaeid shrimps. Aquaculture 286(1-2):1–11. https://doi.org/10.1016/j.aquaculture.2008.09.011
Coutinho F, Castro C, Rufino-Palomares E, Ordonez-Grande B, Gallardo MA, Oliva-Teles A, Peres H (2016) Dietary glutamine supplementation effects on amino acid metabolism, intestinal nutrient absorption capacity and antioxidant response of gilthead sea bream (Sparus aurata) juveniles. Comp Biochem Physiol A Mol Integr Physiol 191:9–17. https://doi.org/10.1016/j.cbpa.2015.09.012
De Almeida LMV, Piñeiro CC, Leite MC, Brolese G, Leal RB, Gottfried C, Gonçalves CA (2008) Protective effects of resveratrol on hydrogen peroxide induced toxicity in primary cortical astrocyte cultures. Neurochem Res 33(1):8–15. https://doi.org/10.1007/s11064-007-9399-5
De Lange CFM, Pluske J, Gong J, Nyachoti CM, Torrallardona D, Brufau J, Estevegarcia E, Lizardo R, Gasa J, Aguilera JF (2010) Strategic use of feed ingredients and feed additives to stimulate gut health and development in young pigs. Livest Sci 134(1):124–134. https://doi.org/10.1016/j.livsci.2010.06.117
Deng Y-P, Jiang W-D, Yang L, Jiang J, Kuang S-Y, Tang L, Wu P, Zhang Y-A, Lin F, Zhou X-Q (2014) Differential growth performance, intestinal antioxidant status and relative expression of Nrf2 and its target genes in young grass carp (Ctenopharyngodon idella) fed with graded levels of leucine. Aquaculture 434:66–73. https://doi.org/10.1016/j.aquaculture.2014.07.026
Dong X, Wei Y, Yu J, Yang W, Qiyou X (2014) Glutamine precursor supplementation increases glutamine synthetase gene expression in intestine of common carp (Cyprinus carpio). Aquac Res 45(9):1559–1566. https://doi.org/10.1111/are.12354
Doucette CD, Schwab DJ, Wingreen NS, Rabinowitz JD (2011) alpha-Ketoglutarate coordinates carbon and nitrogen utilization via enzyme I inhibition. Nat Chem Biol 7(12):894–901. https://doi.org/10.1038/nchembio.685
Enes P, Panserat S, Kaushik S, Oliva-Teles A (2009) Nutritional regulation of hepatic glucose metabolism in fish. Fish Physiol Biochem 35(3):519–539. https://doi.org/10.1007/s10695-008-9259-5
Glasauer A, Chandel NS (2014) Targeting antioxidants for cancer therapy. Biochem Pharmacol 92(1):90–101. https://doi.org/10.1016/j.bcp.2014.07.017
Hegazi MM, Attia ZI, Ashour OA (2010a) Oxidative stress and antioxidant enzymes in liver and white muscle of Nile tilapia juveniles in chronic ammonia exposure. Aquat Toxicol 99(2):118–125. https://doi.org/10.1016/j.aquatox.2010.04.007
Hegazi MM, Attia ZI, Hegazi MAM, Hasanein SS (2010b) Metabolic consequences of chronic sublethal ammonia exposure at cellular and subcellular levels in Nile tilapia brain. Aquaculture 299(1-4):149–156. https://doi.org/10.1016/j.aquaculture.2009.11.020
Hoseini SM, Vatnikov YA, Kulikov EV, Petrov AK, Hoseinifar SH, Van Doan H (2019) Effects of dietary arginine supplementation on ureagenesis and amino acid metabolism in common carp (Cyprinus carpio) exposed to ambient ammonia. Aquaculture 511:734209. https://doi.org/10.1016/j.aquaculture.2019.734209
Hou YQ, Wang L, Ding BY, Liu YL, Zhu HL, Liu JA, Li YT, Wu X, Yin YL, Wu GY (2010) Dietary alpha-ketoglutarate supplementation ameliorates intestinal injury in lipopolysaccharide-challenged piglets. Amino Acids 39(2):555–564. https://doi.org/10.1007/s00726-010-0473-y
Hou Y, Wang L, Ding B, Liu Y, Zhu H, Liu J, Li Y, Kang P, Yin Y, Wu G (2011) alpha-Ketoglutarate and intestinal function. Front Biosci 16(2009):1186–1196. https://doi.org/10.1216/JIE-2009-21-1-1
Hu R, Fufa Q, Tang J, Zhao Q, Yan J, Zhou Z, Zhou Y, Liu Z (2017) Cloning, expression, and nutritional regulation of the glutamine synthetase gene in Ctenopharyngodon idellus. Comp Biochem Physiol B: Biochem Mol Biol 212:70–76. https://doi.org/10.1016/j.cbpb.2017.06.004
Ko J-Y, Kim E-A, Lee J-H, Kang M-C, Lee J-S, Kim J-S, Jung W-K, Jeon Y-J (2014) Protective effect of aquacultured flounder fish-derived peptide against oxidative stress in zebrafish. Fish Shellfish Immunol 36(1):320–323. https://doi.org/10.1016/j.fsi.2013.11.018
Krogdahl A, Hemre GI, Mommsen TP (2005) Carbohydrates in fish nutrition: digestion and absorption in postlarval stages. Aquac Nutr 11(2):103–122. https://doi.org/10.1111/j.1365-2095.2004.00327.x
Li HT, Feng L, Jiang WD, Liu Y, Jiang J, Li SH, Zhou XQ (2013) Oxidative stress parameters and anti-apoptotic response to hydroxyl radicals in fish erythrocytes: protective effects of glutamine, alanine, citrulline and proline. Aquat Toxicol 126:169–179. https://doi.org/10.1016/j.aquatox.2012.11.005
Li H-T, Jiang W-D, Yang L, Jiang J, Zhang Y-A, Wu P, Zeng Y-Y, Zhou X-Q, Lin F (2017) Dietary glutamine improves the function of erythrocytes through its metabolites in juvenile carp (Cyprinus carpio var. Jian). Aquaculture 474:86–94. https://doi.org/10.1016/j.aquaculture.2017.03.041
Lin Y, Miao L-H, Pan W-J, Huang X, Dengu JM, Zhang W-X, Ge X-P, Liu B, Ren M-C, Zhou Q-L, Xie J, Pan L-k, Xi B-w (2018) Effect of nitrite exposure on the antioxidant enzymes and glutathione system in the liver of bighead carp, Aristichthys nobilis. Fish Shellfish Immunol 76:126–132. https://doi.org/10.1016/j.fsi.2018.02.015
Liu CP, Fu J, Xu FP, Wang XS, Li S (2015) The role of heat shock proteins in oxidative stress damage induced by Se deficiency in chicken livers. Biometals 28(1):163–173. https://doi.org/10.1007/s10534-014-9812-x
Livingstone DR (2003) Oxidative stress in aquatic organisms in relation to pollution and aquaculture. Rev Med Vet 154(6):427–430 https://www.revmedvet.com/2003/RMV154_427_430.pdf
Loro VL, Jorge MB, Da Silva KR, Wood CM (2012) Oxidative stress parameters and antioxidant response to sublethal waterborne zinc in a euryhaline teleost Fundulus heteroclitus: protective effects of salinity. Aquat Toxicol 110-111(4):187–193. https://doi.org/10.1016/j.aquatox.2012.01.012
Mailloux RJ, Singh R, Brewer G, Auger C, Lemire J, Appanna VD (2009) α-Ketoglutarate dehydrogenase and glutamate dehydrogenase work in tandem to modulate the antioxidant α-ketoglutarate during oxidative stress in pseudomonas fluorescens. J Bacteriol 191(12):3804–3810. https://doi.org/10.1128/jb.00046-09
Martínez-Álvarez RM, Morales AE, Sanz A (2005) Antioxidant defenses in fish: biotic and abiotic factors. Rev Fish Biol Fish 15(1-2):75–88. https://doi.org/10.1007/s11160-005-7846-4
Mian J, Siddiqui PZJ (2014) Effect of stocking density and protein level on behaviour survival growth rate crowding status stress response food consumption protein efficiency and body composition of hybrid (Oreochromis mossambicus× Oreochromis niloticus) in saline environment. Int J Fish Aquat Stud IJFAS 1(4):72–78 http://www.fisheriesjournal.com/archives/?year = 2014&vol = 1&issue = 4&part = B&ArticleId = 66
Molina R, Moreno I, Pichardo S, Jos A, Moyano R, Monterde J, Camean A (2005) Acid and alkaline phosphatase activities and pathological changes induced in Tilapia fish (Oreochromis sp.) exposed subchronically to microcystins from toxic cyanobacterial blooms under laboratory conditions. Toxicon 46(7):725–735. https://doi.org/10.1016/j.toxicon.2005.07.012
Moon TW (2001) Glucose intolerance in teleost fish: fact or fiction? Comp Biochem Physiol B: Biochem Mol Biol 129(2-3):243–249. https://doi.org/10.1016/S1096-4959(01)00316-5
Peh WYX, Chew SF, Ching BY, Loong AM, Ip YK (2010) Roles of intestinal glutamate dehydrogenase and glutamine synthetase in environmental ammonia detoxification in the euryhaline four-eyed sleeper, Bostrychus sinensis. Aquat Toxicol 98(1):91–98. https://doi.org/10.1016/j.aquatox.2010.01.018
Pohlenz C, Buentello A, Criscitiello MF, Mwangi W, Smith R, Gatlin DM 3rd (2012) Synergies between vaccination and dietary arginine and glutamine supplementation improve the immune response of channel catfish against Edwardsiella ictaluri. Fish Shellfish Immunol 33(3):543–551. https://doi.org/10.1016/j.fsi.2012.06.005
Putker M, O’Neill JS (2016) Reciprocal control of the circadian clock and cellular redox state - a critical appraisal. Mol Cells 39(1):6–19. https://doi.org/10.14348/molcells.2016.2323
Ransberry VE, Morash AJ, Blewett TA, Wood CM, Mcclelland GB (2015) Oxidative stress and metabolic responses to copper in freshwater- and seawater-acclimated killifish, Fundulus heteroclitus. Aquat Toxicol 161:242–252. https://doi.org/10.1016/j.aquatox.2015.02.013
Shaojuan L, Liuqin H, Kang Y (2018) The antioxidative function of alpha-ketoglutarate and its applications. Biomed Res Int 2018:1–6. https://doi.org/10.1155/2018/3408467
Sies H (1999) Glutathione and its role in cellular functions. Free Radic Biol Med 27(9–10):916–921. https://doi.org/10.1016/s0891-5849(99)00177-x
Sliwa E, Tatara MR, Nowakowski H, Pierzynowski SG, Studzinski T (2006) Effect of maternal dexamethasone and alpha-ketoglutarate administration on skeletal development during the last three weeks of prenatal life in pigs. J Matern Fetal Neonatal Med 19(8):489–493. https://doi.org/10.1080/14767050600850381
Suzer C, Aktulun S, Coban D, Okan Kamaci H, Saka S, Firat K, Alpbaz A (2007) Digestive enzyme activities in larvae of sharpsnout seabream (Diplodus puntazzo). Comp Biochem Physiol A Mol Integr Physiol 148(2):470–477. https://doi.org/10.1016/j.cbpa.2007.06.418
Wang H-q, Zhao Y-r, Jin Bi-tao, Jian Liang (2016a) Effects of dietary alpha-ketoglutarate supplementation on growth and serum biochemical parameters of grass carp (Ctenopharyngodon idella) fingerlings. Isr J Aquacult Bamidgeh. http://hdl.handle.net/10524/54930
Wang L, Xu Q, Wang C’a, Li J, Chen D, Zhao Z, Luo L, Xue D (2016b) Effects of dietary α-ketoglutarate supplementation on the growth performance, glutamine synthesis and amino acid concentrations of juvenile hybrid sturgeon Acipenser schrenckii ♀×Acipenser baerii ♂ fed high levels of soy protein concentrate." Animal Feed Science and Technology 211:199-207. https://doi.org/10.1016/j.anifeedsci.2015.11.016
Wang L, Wei Y, Wang C, Li J, Zhao Z, Luo L, Du X, Qiyou X (2017a) Effects of α-ketoglutarate on the growth performance, amino acid metabolism and related gene expression of mirror carp (Cyprinus carpio). Aquac Nutr 23(5):926–933. https://doi.org/10.1111/anu.12460
Wang L, Xu Q, Wang C’a, Li J, Chen D, Zhao Z, Liang L, Xue D (2017b) Effects of dietary α-ketoglutarate supplementation on the antioxidant defense system and HSP 70 and HSP 90 gene expression of hybrid sturgeon Acipenser schrenckii ♀ × A. baerii ♂exposed to ammonia-N stress. Aquac Res 48(5):2266–2277. https://doi.org/10.1111/are.13063
Wicher KB, Fries E (2006) Haptoglobin, a hemoglobin-binding plasma protein, is present in bony fish and mammals but not in frog and chicken. Proc Natl Acad Sci 103(11):4168–4173. https://doi.org/10.1073/pnas.0508723103
Wu G, Bazer FW, Burghardt RC, Johnson GA, Kim SW, Knabe DA, Li P, Li X, Mcknight JR, Satterfield MC (2011) Proline and hydroxyproline metabolism: implications for animal and human nutrition. Amino Acids 40(4):1053–1063. https://doi.org/10.1007/s00726-010-0715-z
Xu Q, Gatlin DM 3rd (2018) Effects of alpha-ketoglutarate (AKG) on growth performance and non-specific immunity of juvenile red drum fed diets with low or adequate phosphorus levels. Fish Physiol Biochem 44(2):573–582. https://doi.org/10.1007/s10695-017-0454-0
Yao K, Yin Y, Li X, Xi P, Wang J, Lei J, Hou Y, Wu G (2012) Alpha-ketoglutarate inhibits glutamine degradation and enhances protein synthesis in intestinal porcine epithelial cells. Amino Acids 42(6):2491–2500. https://doi.org/10.1007/s00726-011-1060-6
Zhang J, Shen H, Wang X, Wu J, Xue Y (2004) Effects of chronic exposure of 2,4-dichlorophenol on the antioxidant system in liver of freshwater fish Carassius auratus. Chemosphere 55(2):167–174. https://doi.org/10.1016/j.chemosphere.2003.10.048
Zhang W, Chen Q, Mai K, Xu W, Wang X, Liufu Z (2010) Effects of dietary α-lipoic acid on the growth and antioxidative responses of juvenile abalone Haliotis discus hannai Ino. Aquac Res 41(11):e781–e787. https://doi.org/10.1111/j.1365-2109.2010.02592.x
Funding
This study was funded by National Key R&D Program of China (No. 2018YFD0900302), National Natural Science Foundation of China (31470132), and Natural Science Foundation of Hunan Province (14JJ4039).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of Interest
The authors declare that they have no conflict of interest.
Ethical approval
All applicable international, national, and/or institutional guidelines for the care and use of animals were followed by the authors.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Lin, X., Jin, B., Wang, H. et al. Effects of diet α-ketoglutarate (AKG) supplementation on the growth performance, antioxidant defense system, intestinal digestive enzymes, and immune response of grass carp (Ctenopharyngodon idellus). Aquacult Int 28, 511–524 (2020). https://doi.org/10.1007/s10499-019-00475-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10499-019-00475-2