Abstract
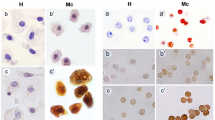
Tunicata is an interesting phylogenetic group, at the base of the branch leading to Chordata. Ascidians (Tunicata, Ascidiacea) morula blood cells are involved in defense reactions and in the formation of the tunic extracellular matrix. The ascidian tunic is hardened as a result of sclerotization of matrix proteins due to the action of phenol oxidase enzyme, contained in morula cells. Morula cells of the ascidian Styela rustica contain two major proteins, the function of which is still unknown; one of them is a protein with molecular mass of 48 kDa – p48. The aim of present study was to search for possible homologues of p48 in ascidians belonging to the Stolidobranchia order: Styela coriacea (Styelidae), Molgula citrina (Molgulidae), Boltenia echinata, Halocynthia aurantium (Pyuridae) and to determine the tissue distribution of those homologues. In order to show the presence of p48 in the tissues of sea squirts we used indirect immunolabeling method on paraffin sections.It was shown that the antibodies bind with morula cells of Styela rustica, Styela coreacea, Boltenia echinata, with the tunic matrix in all studied species and with test cells of Styela rustica, Styela coreacea, Boltenia echinata, Molgula citrina. It gives us a ground to assume the existence of p48 homologues in all studied ascidians and to expect a common mechanism of their participation in the tunic formation, as possible substrates of the phenol oxidase system.




Similar content being viewed by others
REFERENECS
Abebe, A., Kuang, Q.F., Evans, J., Robinson, W.E., and Sugumaran, M., Oxidative transformation of a tunichrome model compound provides new insight into the crosslinking and defense reaction of tunichromes, Bioorg. Chem., 2017, vol. 71, pp. 219–229.
Alié, A., Hiebert, L.S., Simion, P., Scelzo, M., Prünster, M.M., Lotito, S., Delsuc, F., Douzery, E.J.P., Dantec, C., Lemaire, P., Darras, S., Kawamura, K., Brown, F.D., and Tiozzo, S., Convergent acquisition of nonembryonic development in styelid ascidians, Mol. Biol. Evol., 2018, vol. 35, pp. 1728–1743.
Amparyup, P., Charoensapsri, W., and and, Tassanakajon, A., Prophenoloxidase system and its role in shrimp immune responses against major pathogens, Fish Shellfish Immunol., 2013, vol. 34, pp. 990–1001.
Andersen, S.O., Insect cuticular sclerotization: a review, Insect Biochem. Mol. Biol., 2010, vol. 40, pp. 166–178.
Ballarin, L., Franchi, N., Schiavon, F., Tosatto, S.C.E., Mičetić, I., and Kawamura, K., Looking for putative phenoloxidases of compound ascidians: haemocyanin-like proteins in Polyandrocarpa misakiensis and Botryllus schlosseri, Dev. Comp. Immunol., 2012, vol. 38, pp. 232–242.
Bella, M.A., Carbone, M.C., D’Amato, M., Alessandro, R., and De Leo, G., The identification and localization of two intermediate filament proteins in the tunic of Styela plicata (Tunicata, Styelidae), Tissue Cell, 2009, vol. 41, pp. 381–389.
Berrill, N.J., II. Studies in tunicate development, part V: the evolution and classification of ascidians, Phil. Trans. R. Soc. Lond. B, 1936, vol. 226, pp. 43–70.
Burighel, P. and Cloney, R.A., Hemichordata, chaetognatha, and the invertebrate chordates, in Microscopic Anatomy of Invertebrates, New York: Wiley-Liss, 1997, pp. 221–347.
Cammarata, M. and Parrinello, N., The ascidian prophenoloxidase activating system, Invertebr. Survival J., 2009, vol. 6, pp. 67–76.
Cammarata, M., Arizza, V., Cianciolo, C., Parrinello, D., Vazzana, M., Vizzini, A., Salerno, G., and Parrinello, N., The prophenoloxidase system is activated during the tunic inflammatory reaction of Ciona intestinalis, Cell Tiss. Res., 2008, vol. 333, pp. 481–492.
Chaga, O.Yu., Morphology and histochemistry of blood cells of an ascidian Molgula citrina Alder and Hancook, Tsitologiia, 1980a, vol. 55, no. 3, pp. 287–295.
Chaga, O.Yu., Ortho-diphenoloxidase system of ascidians, Tsitologiia, 1980b, vol. 22, no. 6, pp. 619–625.
Chaga, O.Yu., Blood cells of the ascidian Styela (Goniocarpa) rustica, Tsitologiia, 1998, vol. 40, no. 1, pp. 31–44.
Cloney, R.A., Ascidian larvae and the events of metamorphosis, Integ. Comp. Biol., 1982, vol. 22, pp. 817–826.
Cloney, R.A. and Hansson, L.J., Ascidian larvae: the role of test cells in preventing hydrophobicity, Acta Zool., 1996, vol. 77, pp. 73–78.
Delsuc, F., Brinkmann, H., Chourrout, D., and Philippe, H., Tunicates and not cephalochordates are the closest living relatives of vertebrates, Nature, 2006, vol. 439, pp. 965–968.
Delsuc F., Philippe H., Tsagkogeorga, G., Simion P., Tilak M.-K., Turon X., López-Legentil S., Piette J., Lemaire P., Douzery, E.J.P. A phylogenomic framework and timescale for comparative studies of tunicates. BMC Biol. 2018. vol. 16, pp. 16.
Hirose, E., Ascidian tunic cells: morphology and functional diversity of free cells outside the epidermis, Invert. Biol., 2009, vol. 128, pp. 83–96.
Hirose, E., Shirae, M., and Saito, Y., Ultrastructures and classification of circulating hemocytes in 9 botryllid ascidians (Chordata: Ascidiacea), Zool. Sci., 2003, vol. 20, pp. 647–656.
Immesberger, A. and Burmester, T., Putative Phenoloxidases in the Tunicate Ciona Intestinalis and the Origin of the Arthropod Hemocyanin Superfamily, J. Comp. Physiol., 2004, vol. B 174, pp. 169–180.
Isaeva, V.V. and Golubev, A.G., Tunicates: our closest invertebrate relatives, Biosfera, 2017, vol. 99, no. 3, pp. 242–260.
Kocot, K.M., Tassia, M.G., Halanych, K.M., and Swalla, B.J., Phylogenomics offers resolution of major tunicate relationships, Mol. Phylogenet. Evol., 2018, vol. 121, pp. 166–173.
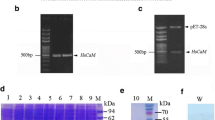
Laemmli, U.K., Cleavage of structural proteins during the assembly of the head of bacteriophage T4, Nature, 1970, vol. 227, pp. 680–685.
Lahille, F., Sur la Classification des Tuniciers, 1886, pp. 446–448. Cited by: Kocot et al., 2018.
Lee, I.H., Cho, Y., and Lehrer, R.I., Styelins, broad-spectrum antimicrobial peptides from the solitary tunicate, Styela clava, Comp. Biochem. Physiol. B. Biochem. Mol. Bio-l., 1997, vol. 118, pp. 515–521.
Lehmann, E.L., Introduction to Student (1908) the probable error of a mean, in Breakthroughs in Statistics: Methodology and Distribution, New York: Springer, 1992, pp. 29–32.
Mann, H.B. and Whitney, D.R., On a test of whether one of two random variables is stochastically larger than the other, Ann. Math. Statist., 1947, vol. 18, pp. 50–60.
Parrinello, N., Arizza, V., Chinnici, C., Parrinello, D., and Cammarata, M., Phenoloxidases in ascidian hemocytes: characterization of the pro-phenoloxidase activating system, Comp. Biochem. Physiol. B: Biochem. Mol. Biol., 2003, vol. 135, pp. 583–591.
Parrinello, N., Arizza, V., Cammarata, M., Giaramita, F.T., Pergolizzi, M., Vazzana, M., Vizzini, A., and Parrinello, D., Inducible lectins with galectin properties and human IL1α epitopes opsonize yeast during the inflammatory response of the ascidian Ciona intestinalis, Cell Tiss. Res., 2007, vol. 329, pp. 379–390.
Parrinello, D., Sanfratello, M.A., Parisi, M.G., Vizzini, A., and Cammarata, M., In the ovary of Ciona intestinalis (type A), immune-related galectin and phenoloxidase genes are differentially expressed by the follicle accessory cells, Fish Shellfish Immunol., 2018, vol. 72, pp. 452–458.
Podgornaya, O.I. and Shaposhnikova, T.G., Antibodies with the cell-type specificity to the morula cells of the solitary ascidians Styela rustica and Boltenia echinata, Cell Struct. Funct., 1998, vol. 23, pp. 349–355.
Rowley, A.F., Ultrastructural and cytochemical studies on the blood cells of the sea squirt, Ciona intestinalis, Cell Tiss. Res., 1982, vol. 223, pp. 403–414.
Sato, Y., Terakado, K., and Morisawa, M., Test cell migration and tunic formation during post-hatching development of the larva of the ascidian, Ciona intestinalis, Dev. Growth Differ., 1997, vol. 39, pp. 117–126.
Shaposhnikova, T.G., Stolbovaya, A. Yu., Ponomartsev, N.V., and Podgornaya, O.I., Immuno- and histochemistry characteristics of morula and test cells in three ascidian species, Tsitologiia, 2011, vol. 53, no. 12, pp. 986–991.
Shirae, M. and Saito, Y., A comparison of hemocytes and their phenoloxidase activity among botryllid ascidians, Zoo. Sci., 2000, vol. 17, pp. 881–891.
Smith, M.J., The blood cells and tunic of the ascidian Halocynthia aurantium (Pallas), I. Hematology, tunic morphology, and partition of cells between blood and tunic, Biol. Bull., 1970, vol. 138, pp. 354–378.
Smith, V.J. and Peddie, C.M., Cell cooperation during host defense in the solitary tunicate Ciona intestinalis (L), Biol. Bull., 1992, vol. 183, pp. 211–219.
Stach, T. and Turbeville, J.M., Phylogeny of Tunicata inferred from molecular and morphological characters, Mol. Phylogenet. Evol., 2002, vol. 25, pp. 408–428.
Sugumaran, M., Unified mechanism for sclerotization of insect cuticle, in Advances in Insect Physiology, New York: Academic Press, 1998, pp. 229–334.
Sukhachev, A.N., Dyatchkov, I.S., Romanyuk, D.S., Kumeyko, V.V., Sinitsina, V.F., Korolkova, E.D., Kharazova, A.D, and Polevschikov, A.V., Morphological analysis of hemocytes of ascidian Halocynthia aurantium, Tsitologiia, 2013, vol. 55, no. 12, pp. 901–906.
Swalla, B.J., Cameron, C.B., Corley, L.S., and Garey, J.R., Urochordates are monophyletic within the deuterostomes, Syst. Biol., 2000, vol. 49, pp. 52–64.
Taylor, S.W., Kammerer, B., Nicholson, G.J., Pusecker, K., Walk, T., Bayer, E., Scippa, S., and de Vincentiis, M., Morulin Pm: a modified polypeptide containing TOPA and 6-bromotryptophan from the morula cells of the ascidian, Phallusia mammillata, Arch. Biochem. Biophys., 1997, vol. 348, pp. 278–288.
Taylor, S.W., Craig, A.G., Fischer, W.H., Park, M., and Lehrer, R.I., Styelin D, an extensively modified antimicrobial peptide from ascidian hemocytes, J. Biol. Chem., 2000, vol. 275, pp. 38417–38426.
Towbin, H., Staehelin, T., and Gordon, J., Electrophoretic transfer of proteins from polyacrylamide gels to nitrocellulose sheets: procedure and some applications, Proc. Natl. Acad. Sci. U. S. A., 1979, vol. 76, pp. 4350–4354.
Winchell, C.J., Sullivan, J., Cameron, C.B., Swalla, B.J., and Mallatt, J., Evaluating hypotheses of deuterostome phylogeny and chordate evolution with new LSU and SSU ribosomal DNA data, Mol. Biol. Evol., 2002, vol. 19, pp. 762–776.
Zeng, L., Jacobs, M.W., and Swalla, B.J., Coloniality has evolved once in stolidobranch ascidians, Integr. Comp. Biol., 2006, vol. 46, pp. 255–268.
ACKNOWLEDGMENTS
We are grateful to the staff of the Kartesh White Sea Biological Station (Zoological Institute, Russian Academy of Sciences) for the opportunity to carry out the work and for assistance in its implementation. We are also grateful to A.I. Solovyeva for assistance in collecting the material and L.S. Adonin for help in obtaining antibodies.
Funding
This work was supported by the Russian Foundation for Basic Research (project no. 15-04-06008), Russian Science Foundation (project no. 19-74-20102) and by the Program of the Presidium of Russian Academy of Sciences “Molecular and Cellular Biology” (no. 01.2.01457147).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest. The authors declare that they have no conflict of int-erest.
Statement on the welfare of animals. The authors declare that all experiments on guinea pigs were carried out in compliance with animal rights and according to generally accepted international ethical standards guaranteed by a certificate of the Institute of Cytology, Russian Academy of Sciences, identification no. F18-00380 (Animal Welfare Assurance).
Additional information
Translated by A. Barkhash
Abbreviations: DOPA—dioxyphenylalanine, BSA—bovine serum albumin, DAB—diaminobenzidine, SDS—sodium dodecyl sulfate, TBST (TBS-tween)—mixture of isotonic Tris-buffered saline solution and Tween 20 polysorbate.
Rights and permissions
About this article
Cite this article
Tylets, M.I., Daugavet, M.A., Savelieva, A.V. et al. Homologues of p48 Protein from Morula Cells of Ascidian Styela rustica in Other Species of Stolidobranchia. Cell Tiss. Biol. 13, 388–396 (2019). https://doi.org/10.1134/S1990519X19050080
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1990519X19050080