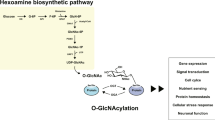
Abstract
Alzheimer’s disease (AD) is the most common cause of dementia and the number of elderly patients suffering from AD has been steadily increasing. Despite worldwide efforts to cope with this disease, little progress has been achieved with regard to identification of effective therapeutics. Thus, active research focusing on identification of new therapeutic targets of AD is ongoing. Among the new targets, post-translational modifications which modify the properties of mature proteins have gained attention. O-GlcNAcylation, a type of PTM that attaches O-linked β-N-acetylglucosamine (O-GlcNAc) to a protein, is being sought as a new target to treat AD pathologies. O-GlcNAcylation has been known to modify the two important components of AD pathological hallmarks, amyloid precursor protein, and tau protein. In addition, elevating O-GlcNAcylation levels in AD animal models has been shown to be effective in alleviating AD-associated pathology. Although studies investigating the precise mechanism of reversal of AD pathologies by targeting O-GlcNAcylation are not yet complete, it is clearly important to examine O-GlcNAcylation regulation as a target of AD therapeutics. This review highlights the mechanisms of O-GlcNAcylation and its role as a potential therapeutic target under physiological and pathological AD conditions.




Similar content being viewed by others
Abbreviations
- AD:
-
Alzheimer’s disease
- AGM:
-
N-acetylglucosamine phosphoglucomutase
- AGX:
-
N-acetylglucosamine-1-phosphorylase
- ALS:
-
Amyotrophic lateral sclerosis
- APH-1:
-
Anterior pharynx-defective 1
- APOE4:
-
Apolipoprotein E4
- APP:
-
Amyloid precursor protein
- ATP5A:
-
ATP synthase subunit α
- Aβ:
-
Amyloid-beta
- BACE1:
-
β-Secretase
- BBB:
-
Blood brain barrier
- CDK2:
-
Cyclin dependent kinase 2
- CEBPβ:
-
CCAAT enhancer-binding protein β
- CT:
-
Computerized tomography
- EOAD:
-
Early onset AD
- ER:
-
Endoplasmic reticulum
- ERβ:
-
Estrogen receptor β
- FAD:
-
Familiar AD
- Fruc-6-P:
-
Fructose-6-phosphate
- G6P-I:
-
Glucose-6-phosphate isomerase
- GFAT:
-
Glutamine–fructose-6-phosphate-transaminase
- Glc-6-P:
-
Glucose-6-phosphate
- GlcN:
-
Glucosamine
- GlcN-6-P:
-
GlcN-6-phosphate
- GlcNAc:
-
N-acetylglucosamine
- GlcNAc-1-P:
-
GlcNAc-1-phosphate
- GlcNAc-6-P:
-
GlcNAc-6-phosphate
- GLUT1:
-
Glucose transporter 1
- GLUT3:
-
Glucose transporter 3
- GNAT:
-
Glucosamine-6-phosphate acetyl transferase
- GSK3β:
-
Glycogen synthase kinase 3β
- HAT:
-
Histone acetyltransferase
- HBP:
-
Hexosamine biosynthetic pathway
- HD:
-
Huntington’s disease
- HK:
-
Hexokinase
- IR:
-
Insulin receptors
- LOAD:
-
Late onset AD
- lOGA:
-
Long OGA
- MAPT:
-
Microtubule-associated protein tau
- MCI:
-
Mild cognitive impairment
- mOGT:
-
Mitochondrial OGT
- MRI:
-
Magnetic resonance imaging
- MTBRs:
-
Microtubule-binding repeats
- NAC:
-
Non-amyloid β component
- ncOGT:
-
Nucleocytoplasmic OGT
- NFs:
-
Neurofilaments
- NFTs:
-
Neurofibrillary tangles
- nNOS:
-
Neuronal nitric oxide synthase
- OGA:
-
O-GlcNAcase
- O-GlcNAc:
-
O-linked β-N-acetylglucosamin
- OGT:
-
O-GlcNAc transferase
- PD:
-
Parkinson’s disease
- PEN2:
-
Presenilin enhancer 2
- PPO:
-
Phosphoinositide binding
- PS1:
-
Presenilin1
- PS2:
-
Presenilin2
- PTMs:
-
Post-translational modifications
- ROS:
-
Reactive oxygen species
- sAPPα:
-
Soluble APPα
- sAPPβ:
-
Soluble APPβ
- SDS:
-
Sodium dodecyl sulfate
- Ser:
-
Serine
- SOD1:
-
Superoxide dismutase 1
- sOGA:
-
Short OGA
- sOGT:
-
Short OGT
- T2DM:
-
Type 2 diabetes mellitus
- Thr:
-
Threonine
- TPR:
-
Tetratricopeptide repeat
- UDP-GlcNAc:
-
Uridine diphosphate N-acetylglucosamine
- XBP1:
-
X-box binding protein1
References
Acosta-Baena, N., Sepulveda-Falla, D., Lopera-Gomez, C. M., Jaramillo-Elorza, M. C., Moreno, S., Aguirre-Acevedo, D. C. et al. (2011). Pre-dementia clinical stages in presenilin 1 E280A familial early-onset Alzheimer's disease: A retrospective cohort study. Lancet Neurology,10, 213–220.
Akan, I., Olivier-Van Stichelen, S., Bond, M. R., & Hanover, J. A. (2018). Nutrient-driven O-GlcNAc in proteostasis and neurodegeneration. Journal of Neurochemistry,144, 7–34.
Akimoto, Y., Comer, F. I., Cole, R. N., Kudo, A., Kawakami, H., Hirano, H., et al. (2003). Localization of the O-GlcNAc transferase and O-GlcNAc-modified proteins in rat cerebellar cortex. Brain Research,966, 194–205.
Alford, S., Patel, D., Perakakis, N., & Mantzoros, C. S. (2018). Obesity as a risk factor for Alzheimer's disease: Weighing the evidence. Obesity Reviews,19, 269–280.
Alonso, A., Zaidi, T., Novak, M., Grundke-Iqbal, I., & Iqbal, K. (2001). Hyperphosphorylation induces self-assembly of tau into tangles of paired helical filaments/straight filaments. Proceedings of the National Academy of Sciences of the USA,98, 6923–6928.
Alonso, J., Schimpl, M., & van Aalten, D. M. F. (2014). O-GlcNAcase: Promiscuous hexosaminidase or key regulator of O-GlcNAc signaling? Journal of Biological Chemistry,289, 34433–34439.
Andersen, K. K., Olsen, T. S., Dehlendorff, C., & Kammersgaard, L. P. (2009). Hemorrhagic and ischemic strokes compared: Stroke severity, mortality, and risk factors. Stroke,40, 2068–2072.
Andrés-Bergós, J., Tardio, L., Larranaga-Vera, A., Gómez, R., Herrero-Beaumont, G., & Largo, R. (2012). The increase in O-linked N-acetylglucosamine protein modification stimulates chondrogenic differentiation both in vitro and in vivo. Journal of Biological Chemistry,287, 33615–33628.
Arnold, C. S., Johnson, G. V., Cole, R. N., Dong, D. L., Lee, M., & Hart, G. W. (1996). The microtubule-associated protein tau is extensively modified with O-linked N-acetylglucosamine. Journal of Biological Chemistry,271, 28741–28744.
Arvanitakis, Z., Wilson, R. S., Bienias, J. L., Evans, D. A., & Bennett, D. A. (2004). Diabetes mellitus and risk of Alzheimer disease and decline in cognitive function. Archives of Neurology,61, 661–666.
Athauda, D., & Foltynie, T. (2016). Insulin resistance and Parkinson's disease: A new target for disease modification? Progress in Neurobiology,145–146, 98–120.
Baek, S. H., & Park, S. J. (2017). Inhibition of Drp1 ameliorates synaptic depression, abeta deposition, and cognitive impairment in an Alzheimer's disease model. Journal of Neuroscience,37, 5099–5110.
Bahn, G., Park, J.-S., Yun, U. J., Lee, Y. J., Choi, Y., Park, J. S. et al. (2019). NRF2/ARE pathway negatively regulates BACE1 expression and ameliorates cognitive deficits in mouse Alzheimer’s models. Proceedings of the National Academy of Sciences of the USA,116, 12516–12523.
Baik, S. H., Fane, M., Park, J. H., Cheng, Y. L., Yang-Wei Fann, D., Yun, U. J. et al. (2015). Pin1 promotes neuronal death in stroke by stabilizing Notch intracellular domain. Annals of Neurology,77, 504–516.
Banerjee, P. S., Lagerlof, O., & Hart, G. W. (2016). Roles of O-GlcNAc in chronic diseases of aging. Molecular Aspects of Medicine,51, 1–15.
Banerjee, P. S., Ma, J., & Hart, G. W. (2015). Diabetes-associated dysregulation of O-GlcNAcylation in rat cardiac mitochondria. Proceedings of the National Academy of Sciences of the USA,112, 6050–6055.
Bell, K. F., Zheng, L., Fahrenholz, F., & Cuello, A. C. (2008). ADAM-10 over-expression increases cortical synaptogenesis. Neurobiology of Aging,29, 554–565.
Benedict, C., Hallschmid, M., Hatke, A., Schultes, B., Fehm, H. L., Born, J., et al. (2004). Intranasal insulin improves memory in humans. Psychoneuroendocrinology,29, 1326–1334.
Bertram, L., Blacker, D., Mullin, K., Keeney, D., Jones, J., Basu, S. et al. (2000). Evidence for genetic linkage of Alzheimer's disease to chromosome 10q. Science,290, 2302–2303.
Bisaglia, M., Trolio, A., Bellanda, M., Bergantino, E., Bubacco, L., & Mammi, S. (2006). Structure and topology of the non-amyloid-beta component fragment of human alpha-synuclein bound to micelles: Implications for the aggregation process. Protein Science,15, 1408–1416.
Biwi, J., & Biot, C. (2018). The many ways by which O-GlcNAcylation may orchestrate the diversity of complex glycosylations. Molecules,23, E2858.
Bloom, G. S. (2014). Amyloid-beta and tau: The trigger and bullet in Alzheimer disease pathogenesis. JAMA Neurol.,71, 505–508.
Bolduc, D. M., Montagna, D. R., Seghers, M. C., Wolfe, M. S., & Selkoe, D. J. (2016). The amyloid-beta forming tripeptide cleavage mechanism of γ-secretase. Elife,5, e17578.
Bomfim, T. R., Forny-Germano, L., Sathler, L. B., Brito-Moreira, J., Houzel, J. C., Decker, H. et al. (2012). An anti-diabetes agent protects the mouse brain from defective insulin signaling caused by Alzheimer's disease-associated Abeta oligomers. Journal of Clinical Investigation,122, 1339–1353.
Bond, M. R., & Hanover, J. A. (2013). O-GlcNAc cycling: A link between metabolism and chronic disease. Annual Review of Nutrition,33, 205–229.
Bond, M. R., & Hanover, J. A. (2015). A little sugar goes a long way: The cell biology of O-GlcNAc. Journal of Cell Biology,208, 869–880.
Bondi, M. W., Edmonds, E. C., & Salmon, D. P. (2017). Alzheimer's disease: Past, present, and future. Journal of the International Neuropsychological Society,23, 818–831.
Borodkin, V. S., Schimpl, M., Gundogdu, M., Rafie, K., Dorfmueller, H. C., Robinson, D. A., et al. (2014). Bisubstrate UDP-peptide conjugates as human O-GlcNAc transferase inhibitors. The Biochemical Journal,457, 497–502.
Braak, H., & Braak, E. (1991). Neuropathological stageing of Alzheimer-related changes. Acta Neuropathologica,82, 239–259.
Brockhausen, I., Nair, D. G., Chen, M., Yang, X., Allingham, J. S., Szarek, W. A., et al. (2016). Human acetyl-CoA:glucosamine-6-phosphate N-acetyltransferase 1 has a relaxed donor specificity and transfers acyl groups up to four carbons in length. Biochemistry and Cell Biology,94, 197–204.
Bronner, I. F., ter Meulen, B. C., Azmani, A., Severijnen, L. A., Willemsen, R., Kamphorst, W. et al. (2005). Hereditary Pick's disease with the G272V tau mutation shows predominant three-repeat tau pathology. Brain,128, 2645–2653.
Bubu, O. M., Brannick, M., Mortimer, J., Umasabor-Bubu, O., Sebastião, Y. V., Wen, Y. et al. (2017). Sleep, cognitive impairment, and Alzheimer’s disease: A systematic review and meta-analysis. Sleep,40, 32.
Buee, L., Bussiere, T., Buee-Scherrer, V., Delacourte, A., & Hof, P. R. (2000). Tau protein isoforms, phosphorylation and role in neurodegenerative disorders. Brain Research Reviews,33, 95–130.
Cai, H., Cong, W.-N., Ji, S., Rothman, S., Maudsley, S., & Martin, B. (2012). Metabolic dysfunction in Alzheimer's disease and related neurodegenerative disorders. Current Alzheimer Research,9, 5–17.
Campion, D., Dumanchin, C., Hannequin, D., Dubois, B., Belliard, S., Puel, M. et al. (1999). Early-onset autosomal dominant Alzheimer disease: Prevalence, genetic heterogeneity, and mutation spectrum. American Journal of Human Genetics,65, 664–670.
Castro, D. M., Dillon, C., Machnicki, G., & Allegri, R. F. (2010). The economic cost of Alzheimer's disease: Family or public health burden? Dementia Neuropsychologia,4, 262–267.
Cha, M. Y., Cho, H. J., Kim, C., Jung, Y. O., Kang, M. J., Murray, M. E. et al. (2015). Mitochondrial ATP synthase activity is impaired by suppressed O-GlcNAcylation in Alzheimer's disease. Human Molecular Genetics,24, 6492–6504.
Chen, R., Gong, P., Tao, T., Gao, Y., Shen, J., Yan, Y. et al. (2017). O-GlcNAc glycosylation of nNOS promotes neuronal apoptosis following glutamate excitotoxicity. Cellular and Molecular Neurobiology,37, 1465–1475.
Chen, Y. X., Du, J. T., Zhou, L. X., Liu, X. H., Zhao, Y. F., Nakanishi, H., et al. (2006). Alternative O-GlcNAcylation/O-phosphorylation of Ser16 induce different conformational disturbances to the N terminus of murine estrogen receptor beta. Chemistry & Biology,13, 937–944.
Cheng, D., Noble, J., Tang, M. X., Schupf, N., Mayeux, R., & Luchsinger, J. A. (2011). Type 2 diabetes and late-onset Alzheimer's disease. Dementia and Geriatric Cognitive Disorders,31, 424–430.
Chiaradonna, F., Ricciardiello, F., & Palorini, R. (2018). The nutrient-sensing hexosamine biosynthetic pathway as the hub of cancer metabolic rewiring. Cells,7, E53.
Chow, V. W., Mattson, M. P., Wong, P. C., & Gleichmann, M. (2010). An overview of APP processing enzymes and products. Neuromolecular Medicine,12, 1–12.
Chun, Y. S., Kwon, O. H., & Chung, S. (2017). O-GlcNAcylation of amyloid-beta precursor protein at threonine 576 residue regulates trafficking and processing. Biochemical and Biophysical Research Communications,490, 486–491.
Chun, Y. S., Park, Y., Oh, H. G., Kim, T. W., Yang, H. O., Park, M. K., et al. (2015). O-GlcNAcylation promotes non-amyloidogenic processing of amyloid-beta protein precursor via inhibition of endocytosis from the plasma membrane. Journal of Alzheimer's Disease,44, 261–275.
Citron, M., Westaway, D., Xia, W., Carlson, G., Diehl, T., Levesque, G. et al. (1997). Mutant presenilins of Alzheimer's disease increase production of 42-residue amyloid β-protein in both transfected cells and transgenic mice. Nature Medicine,3, 67–72.
Cole, R. N., & Hart, G. W. (2001). Cytosolic O-glycosylation is abundant in nerve terminals. Journal of Neurochemistry,79, 1080–1089.
Comtesse, N., Maldener, E., & Meese, E. (2001). Identification of a nuclear variant of MGEA5, a cytoplasmic hyaluronidase and a beta-N-acetylglucosaminidase. Biochemical and Biophysical Research Communications,283, 634–640.
Dayalu, P., & Albin, R. L. (2015). Huntington disease: Pathogenesis and treatment. Neurologic Clinics,33, 101–114.
De Felice, F. G., Lourenco, M. V., & Ferreira, S. T. (2014). How does brain insulin resistance develop in Alzheimer's disease? Alzheimer’s Dementia,10, S26–S32.
De Jonghe, C., Esselens, C., Kumar-Singh, S., Craessaerts, K., Serneels, S., Checler, F. et al. (2001). Pathogenic APP mutations near the gamma-secretase cleavage site differentially affect Abeta secretion and APP C-terminal fragment stability. Human Molecular Genetics,10, 1665–1671.
de Queiroz, R. M., Carvalho, E., & Dias, W. B. (2014). O-GlcNAcylation: The sweet side of the cancer. Frontiers in Oncologys,4, 132–132.
Delbeuck, X., Van der Linden, M., & Collette, F. (2003). Alzheimer's disease as a disconnection syndrome? Neuropsychology Review,13, 79–92.
Deng, Y., Li, B., Liu, F., Iqbal, K., Grundke-Iqbal, I., Brandt, R., et al. (2008). Regulation between O-GlcNAcylation and phosphorylation of neurofilament-M and their dysregulation in Alzheimer disease. The FASEB Journal,22, 138–145.
Deng, Y., Li, B., Liu, Y., Iqbal, K., Grundke-Iqbal, I., & Gong, C. X. (2009). Dysregulation of insulin signaling, glucose transporters, O-GlcNAcylation, and phosphorylation of tau and neurofilaments in the brain: Implication for Alzheimer's disease. American Journal of Pathology,175, 2089–2098.
Donev, R., Kolev, M., Millet, B., & Thome, J. (2009). Neuronal death in Alzheimer's disease and therapeutic opportunities. Journal of Cellular and Molecular Medicine,13, 4329–4348.
Dong, D. L., Xu, Z. S., Chevrier, M. R., Cotter, R. J., Cleveland, D. W., & Hart, G. W. (1993). Glycosylation of mammalian neurofilaments. Localization of multiple O-linked N-acetylglucosamine moieties on neurofilament polypeptides L and M. Journal of Biological Chemistry,268, 16679–16687.
Dorfmueller, H. C., Borodkin, V. S., Blair, D. E., Pathak, S., Navratilova, I., & van Aalten, D. M. (2011). Substrate and product analogues as human O-GlcNAc transferase inhibitors. Amino Acids,40, 781–792.
Dorfmueller, H. C., Borodkin, V. S., Schimpl, M., & van Aalten, D. M. (2009). GlcNAcstatins are nanomolar inhibitors of human O-GlcNAcase inducing cellular hyper-O-GlcNAcylation. The Biochemical Journal,420, 221–227.
Dorfmueller, H. C., & van Aalten, D. M. (2010). Screening-based discovery of drug-like O-GlcNAcase inhibitor scaffolds. FEBS Letters,584, 694–700.
Dorszewska, J., Prendecki, M., Oczkowska, A., Dezor, M., & Kozubski, W. (2016). Molecular basis of familial and sporadic Alzheimer's disease. Current Alzheimer Research,13, 952–963.
Duan, A. R., Jonasson, E. M., Alberico, E. O., Li, C., Scripture, J. P., Miller, R. A. et al. (2017). Interactions between tau and different conformations of tubulin: Implications for tau function and mechanism. Journal of Molecular Biology,429, 1424–1438.
Dulin, F., Léveillé, F., Ortega, J. B., Mornon, J.-P., Buisson, A., Callebaut, I., et al. (2008). p3 peptide, a truncated form of Aβ devoid of synaptotoxic effect, does not assemble into soluble oligomers. FEBS Letters,582, 1865–1870.
Dumurgier, J., Schraen, S., Gabelle, A., Vercruysse, O., Bombois, S., Laplanche, J.-L. et al. (2015). Cerebrospinal fluid amyloid-β 42/40 ratio in clinical setting of memory centers: A multicentric study. Alzheimer’s Research & Therapy,7, 30.
Dunn, L., Allen, G. F., Mamais, A., Ling, H., Li, A., Duberley, K. E. et al. (2014). Dysregulation of glucose metabolism is an early event in sporadic Parkinson's disease. Neurobiology of Aging,35, 1111–1115.
El Haj, M., Gallouj, K., Dehon, H., Roche, J., & Laroi, F. (2018). Hallucinations in Alzheimer's disease: Failure to suppress irrelevant memories. Cognitive Neuropsychiatry,23, 142–153.
Endo, A., Kakiki, K., & Misato, T. (1970). Feedback inhibition of l-glutamine d-fructose 6-phosphate amidotransferase by uridine diphosphate N-acetylglucosamine in Neurospora crassa. Journal of Bacteriology,103, 588–594.
Feigin, V. L., Forouzanfar, M. H., Krishnamurthi, R., Mensah, G. A., Connor, M., Bennett, D. A. et al. (2014). Global and regional burden of stroke during 1990–2010: Findings from the Global Burden of Disease Study 2010. Lancet,383, 245–254.
Ferrarini, L., Palm, W. M., Olofsen, H., van Buchem, M. A., Reiber, J. H., & Admiraal-Behloul, F. (2006). Shape differences of the brain ventricles in Alzheimer's disease. Neuroimage,32, 1060–1069.
Ferreira, L. K., & Busatto, G. F. (2011). Neuroimaging in Alzheimer's disease: Current role in clinical practice and potential future applications. Clinics,66(Suppl 1), 19–24.
Ferrer, C. M., Sodi, V. L., & Reginato, M. J. (2016). O-GlcNAcylation in cancer biology: Linking metabolism and signaling. Journal of Molecular Biology,428, 3282–3294.
Ferri, C. P., Prince, M., Brayne, C., Brodaty, H., Fratiglioni, L., Ganguli, M. et al. (2005). Global prevalence of dementia: A Delphi consensus study. Lancet,366, 2112–2117.
Firbank, M. J., Yarnall, A. J., Lawson, R. A., Duncan, G. W., Khoo, T. K., Petrides, G. S. et al. (2017). Cerebral glucose metabolism and cognition in newly diagnosed Parkinson's disease: ICICLE-PD study. Journal of Neurology, Neurosurgery and Psychiatry,88, 310–316.
Foley, J. M., Salat, D. H., Stricker, N. H., Zink, T. A., Grande, L. J., McGlinchey, R. E. et al. (2014). Interactive effects of apolipoprotein E4 and diabetes risk on later myelinating white matter regions in neurologically healthy older aged adults. American Journal of Alzheimer's Disease & Other Dementias,29, 222–235.
Forster, S., Welleford, A. S., Triplett, J. C., Sultana, R., Schmitz, B., & Butterfield, D. A. (2014). Increased O-GlcNAc levels correlate with decreased O-GlcNAcase levels in Alzheimer disease brain. Biochimica et Biophysica Acta,1842, 1333–1339.
Forstl, H., & Kurz, A. (1999). Clinical features of Alzheimer's disease. European Archives of Psychiatry and Clinical Neuroscience,249, 288–290.
Forsythe, M. E., Love, D. C., Lazarus, B. D., Kim, E. J., Prinz, W. A., Ashwell, G. et al. (2006). Caenorhabditis elegans ortholog of a diabetes susceptibility locus: Oga-1 (O-GlcNAcase) knockout impacts O-GlcNAc cycling, metabolism, and dauer. Proceedings of the National Academy of Sciences of the USA,103, 11952–11957.
Fox, N. C., Freeborough, P. A., & Rossor, M. N. (1996). Visualisation and quantification of rates of atrophy in Alzheimer's disease. Lancet,348, 94–97.
Frolich, L., Blum-Degen, D., Bernstein, H. G., Engelsberger, S., Humrich, J., Laufer, S. et al. (1998). Brain insulin and insulin receptors in aging and sporadic Alzheimer's disease. Journal of Neural Transmission,105, 423–438.
Frozza, R. L., Lourenco, M. V., & De Felice, F. G. (2018). Challenges for Alzheimer's disease therapy: Insights from novel mechanisms beyond memory defects. Frontiers in Neuroscience,12, 37.
Fukushima, N., Furuta, D., Hidaka, Y., Moriyama, R., & Tsujiuchi, T. (2009). Post-translational modifications of tubulin in the nervous system. Journal of Neurochemistry,109, 683–693.
Gao, Y., Wells, L., Comer, F. I., Parker, G. J., & Hart, G. W. (2001). Dynamic O-glycosylation of nuclear and cytosolic proteins: Cloning and characterization of a neutral, cytosolic beta-N-acetylglucosaminidase from human brain. Journal of Biological Chemistry,276, 9838–9845.
Gasparini, L., Terni, B., & Spillantini, M. G. (2007). Frontotemporal dementia with tau pathology. Neurodegenerative Diseases,4, 236–253.
Gatta, E., Lefebvre, T., Gaetani, S., Dos Santos, M., Marrocco, J., Mir, A.-M. et al. (2016). Evidence for an imbalance between tau O-GlcNAcylation and phosphorylation in the hippocampus of a mouse model of Alzheimer’s disease. Pharmacological Research,105, 186–197.
Glenner, G. G., & Wong, C. W. (1984). Alzheimer's disease: Initial report of the purification and characterization of a novel cerebrovascular amyloid protein. Biochemical and Biophysical Research Communications,120, 885–890.
Gloster, T. M., & Vocadlo, D. J. (2010). Mechanism, structure, and inhibition of O-GlcNAc processing enzymes. Current Signal Transduction Therapy,5, 74–91.
Gloster, T. M., Zandberg, W. F., Heinonen, J. E., Shen, D. L., Deng, L., & Vocadlo, D. J. (2011). Hijacking a biosynthetic pathway yields a glycosyltransferase inhibitor within cells. Nature Chemical Biology,7, 174–181.
Goedert, M., Spillantini, M. G., Jakes, R., Rutherford, D., & Crowther, R. A. (1989). Multiple isoforms of human microtubule-associated protein tau: Sequences and localization in neurofibrillary tangles of Alzheimer's disease. Neuron,3, 519–526.
Goetz, C. G., Emre, M., & Dubois, B. (2008). Parkinson's disease dementia: Definitions, guidelines, and research perspectives in diagnosis. Annals of Neurology,64(Suppl 2), S81–92.
Graham, D. L., Gray, A. J., Joyce, J. A., Yu, D., O'Moore, J., Carlson, G. A. et al. (2014). Increased O-GlcNAcylation reduces pathological tau without affecting its normal phosphorylation in a mouse model of tauopathy. Neuropharmacology,79, 307–313.
Griffith, L. S., Mathes, M., & Schmitz, B. (1995). Beta-amyloid precursor protein is modified with O-linked N-acetylglucosamine. Journal of Neuroscience Research,41, 270–278.
Griffith, L. S., & Schmitz, B. (1995). O-linked N-acetylglucosamine is upregulated in Alzheimer brains. Biochemical and Biophysical Research Communications,213, 424–431.
Griffith, L. S., & Schmitz, B. (1999). O-linked N-acetylglucosamine levels in cerebellar neurons respond reciprocally to pertubations of phosphorylation. European Journal of Biochemistry,262, 824–831.
Grima, J. C., Daigle, J. G., Arbez, N., Cunningham, K. C., Zhang, K., Ochaba, J. et al. (2017). Mutant huntingtin disrupts the nuclear pore complex. Neuron,94, 93–107.
Gu, J. H., Shi, J., & Dai, C. L. (2017). O-GlcNAcylation reduces ischemia–reperfusion-induced brain injury. Scientific Reports,7, 10686.
Hackam, A. S., Singaraja, R., Wellington, C. L., Metzler, M., McCutcheon, K., Zhang, T. et al. (1998). The influence of huntingtin protein size on nuclear localization and cellular toxicity. Journal of Cell Biology,141, 1097–1105.
Hanover, J. A., Yu, S., Lubas, W. B., Shin, S. H., Ragano-Caracciola, M., Kochran, J., et al. (2003). Mitochondrial and nucleocytoplasmic isoforms of O-linked GlcNAc transferase encoded by a single mammalian gene. Archives of Biochemistry and Biophysics,409, 287–297.
Hardivillé, S., & Hart, G. W. (2014). Nutrient regulation of signaling, transcription, and cell physiology by O-GlcNAcylation. Cell Metabolism,20, 208–213.
Hart, G. W., Slawson, C., Ramirez-Correa, G., & Lagerlof, O. (2011). Cross talk between O-GlcNAcylation and phosphorylation: Roles in signaling, transcription, and chronic disease. Annual Review of Biochemistry,80, 825–858.
Hastings, N. B., Wang, X., Song, L., Butts, B. D., Grotz, D., Hargreaves, R. et al. (2017). Inhibition of O-GlcNAcase leads to elevation of O-GlcNAc tau and reduction of tauopathy and cerebrospinal fluid tau in rTg4510 mice. Molecular Neurodegeneration,12, 39–39.
He, Y., Ma, X., Li, D., & Hao, J. (2017). Thiamet G mediates neuroprotection in experimental stroke by modulating microglia/macrophage polarization and inhibiting NF-κB p65 signaling. Journal of Cerebral Blood Flow and Metabolism,37, 2938–2951.
Hebert, L. E., Weuve, J., Scherr, P. A., & Evans, D. A. (2013). Alzheimer dsisease in the United States (2010–2050) estimated using the 2010 census. Neurology,80, 1778–1783.
Hick, M., Herrmann, U., Weyer, S. W., Mallm, J. P., Tschape, J. A., Borgers, M. et al. (2015). Acute function of secreted amyloid precursor protein fragment APPsalpha in synaptic plasticity. Acta Neuropathologica,129, 21–37.
Hinderlich, S., Berger, M., Schwarzkopf, M., Effertz, K., & Reutter, W. (2000). Molecular cloning and characterization of murine and human N-acetylglucosamine kinase. European Journal of Biochemistry,267, 3301–3308.
Hippius, H., & Neundörfer, G. (2003). The discovery of Alzheimer's disease. Dialogues in Clinical Neuroscience,5, 101–108.
Hirano, A., Nakano, I., Kurland, L. T., Mulder, D. W., Holley, P. W., & Saccomanno, G. (1984). Fine structural study of neurofibrillary changes in a family with amyotrophic lateral sclerosis. Journal of Neuropathology and Experimental Neurology,43, 471–480.
Hofmann, M., Boles, E., & Zimmermann, F. K. (1994). Characterization of the essential yeast gene encoding N-acetylglucosamine-phosphate mutase. European Journal of Biochemistry,221, 741–747.
Horsch, M., Hoesch, L., Vasella, A., & Rast, D. M. (1991). N-acetylglucosaminono-1,5-lactone oxime and the corresponding (phenylcarbamoyl)oxime Novel and potent inhibitors of beta-N-acetylglucosaminidase. European Journal of Biochemistry,197, 815–818.
Hresko, R. C., Heimberg, H., Chi, M. M., & Mueckler, M. (1998). Glucosamine-induced insulin resistance in 3T3-L1 adipocytes is caused by depletion of intracellular ATP. Journal of Biological Chemistry,273, 20658–20668.
Hsu, D., & Marshall, G. A. (2017). Primary and secondary prevention trials in Alzheimer disease: Looking back, move forward. Current Alzheimer Research,14, 426–440.
Hu, Y., Ye, Y., & Fortini, M. E. (2002). Nicastrin is required for gamma-secretase cleavage of the Drosophila Notch receptor. Developmental Cell,2, 69–78.
Hurlbert, M. S., Zhou, W., Wasmeier, C., Kaddis, F. G., Hutton, J. C., & Freed, C. R. (1999). Mice transgenic for an expanded CAG repeat in the Huntington's disease gene develop diabetes. Diabetes,48, 649–651.
Hwang, S. Y., Shin, J. H., Hwang, J. S., Kim, S. Y., Shin, J. A., Oh, E. S. et al. (2010). Glucosamine exerts a neuroprotective effect via suppression of inflammation in rat brain ischemia/reperfusion injury. Glia,58, 1881–1892.
Ighodaro, O. M., Adeosun, A. M., & Akinloye, O. A. (2017). Alloxan-induced diabetes, a common model for evaluating the glycemic-control potential of therapeutic compounds and plants extracts in experimental studies. Medicina,53, 365–374.
Inzucchi, S. E., Viscoli, C. M., Young, L. H., Furie, K. L., Gorman, M., Lovejoy, A. M. et al. (2016). Pioglitazone prevents diabetes in patients with insulin resistance and cerebrovascular disease. Diabetes Care,39, 1684–1692.
Itkin, A., Salnikov, E. S., Aisenbrey, C., Raya, J., Glattard, E., Raussens, V. et al. (2017). Structural characterization of the amyloid precursor protein transmembrane domain and its γ-cleavage site. ACS Omega,2, 6525–6534.
Itkonen, H. M., Minner, S., Guldvik, I. J., Sandmann, M. J., Tsourlakis, M. C., Berge, V. et al. (2013). O-GlcNAc transferase integrates metabolic pathways to regulate the stability of c-MYC in human prostate cancer cells. Cancer Research,73, 5277–5287.
Jacobsen, K. T., & Iverfeldt, K. (2011). O-GlcNAcylation increases non-amyloidogenic processing of the amyloid-beta precursor protein (APP). Biochemical and Biophysical Research Communications,404, 882–886.
Jaffer, H., Morris, V. B., Stewart, D., & Labhasetwar, V. (2011). Advances in stroke therapy. Drug Delivery and Translational Research,1, 409–419.
Jahn, H. (2013). Memory loss in Alzheimer's disease. Dialogues in Clinical Neuroscience,15, 445–454.
Jia, J., Wei, C., Chen, S., Li, F., Tang, Y., Qin, W. et al. (2018). The cost of Alzheimer's disease in China and re-estimation of costs worldwide. Alzheimer’s Dementia,14, 483–491.
Jiang, J., Lazarus, M. B., Pasquina, L., Sliz, P., & Walker, S. (2011). A neutral diphosphate mimic crosslinks the active site of human O-GlcNAc transferase. Nature Chemical Biology,8, 72–77.
Jiang, M., Yu, S., Yu, Z., Sheng, H., Li, Y., Liu, S. et al. (2017). XBP1 (X-box-binding protein-1)-dependent O-GlcNAcylation Is neuroprotective in ischemic stroke in young mice and its impairment in aged mice is rescued by thiamet-G. Stroke,48, 1646–1654.
Johnson, V. E., Stewart, W., & Smith, D. H. (2010). Traumatic brain injury and amyloid-beta pathology: A link to Alzheimer's disease? Nature Reviews Neuroscience,11, 361–370.
Joiner, C. M., Li, H., Jiang, J., & Walker, S. (2019). Structural characterization of the O-GlcNAc cycling enzymes: Insights into substrate recognition and catalytic mechanisms. Current Opinion in Structural Biology,56, 97–106.
Josefsen, K., Nielsen, M. D., Jorgensen, K. H., Bock, T., Norremolle, A., Sorensen, S. A. et al. (2008). Impaired glucose tolerance in the R6/1 transgenic mouse model of Huntington's disease. Journal of Neuroendocrinology,20, 165–172.
Kadavath, H., Hofele, R. V., Biernat, J., Kumar, S., Tepper, K., Urlaub, H. et al. (2015). Tau stabilizes microtubules by binding at the interface between tubulin heterodimers. Proceedings of the National Academy of Sciences of the USA,112, 7501.
Kametani, F., & Hasegawa, M. (2018). Reconsideration of amyloid hypothesis and tau hypothesis in Alzheimer's disease. Frontiers in Neuroscience,12, 25.
Kang, J., Lemaire, H. G., Unterbeck, A., Salbaum, J. M., Masters, C. L., Grzeschik, K. H. et al. (1987). The precursor of Alzheimer's disease amyloid A4 protein resembles a cell-surface receptor. Nature,325, 733–736.
Keembiyehetty, C. N., Krzeslak, A., Love, D. C., & Hanover, J. A. (2011). A lipid-droplet-targeted O-GlcNAcase isoform is a key regulator of the proteasome. Journal of Cell Science,124, 2851–2860.
Kim, C., Nam, D. W., Park, S. Y., Song, H., Hong, H. S., Boo, J. H. et al. (2013). O-linked beta-N-acetylglucosaminidase inhibitor attenuates beta-amyloid plaque and rescues memory impairment. Neurobiology of Aging,34, 275–285.
Kim, E. J., Kang, D. O., Love, D. C., & Hanover, J. A. (2006). Enzymatic characterization of O-GlcNAcase isoforms using a fluorogenic GlcNAc substrate. Carbohydrate Research,341, 971–982.
Knapp, S., Vocadlo, D., Gao, Z., Kirk, B., Lou, J., & Withers, S. G. (1996). NAG-thiazoline, an N-Acetyl-β-hexosaminidase inhibitor that implicates acetamido participation. Journal of the American Chemical Society,118, 6804–6805.
Kojro, E., & Fahrenholz, F. (2005). The non-amyloidogenic pathway: Structure and function of alpha-secretases. SubCellular Biochemistry,38, 105–127.
Kopke, E., Tung, Y. C., Shaikh, S., Alonso, A. C., Iqbal, K., & Grundke-Iqbal, I. (1993). Microtubule-associated protein tau. Abnormal phosphorylation of a non-paired helical filament pool in Alzheimer disease. Journal of Biological Chemistry,268, 24374–24384.
Kreppel, L. K., Blomberg, M. A., & Hart, G. W. (1997). Dynamic glycosylation of nuclear and cytosolic proteins. Cloning and characterization of a unique O-GlcNAc transferase with multiple tetratricopeptide repeats. Journal of Biological Chemistry,272, 9308–9315.
Kuemmerle, S., Gutekunst, C. A., Klein, A. M., Li, X. J., Li, S. H., Beal, M. F. et al. (1999). Huntington aggregates may not predict neuronal death in Huntington's disease. Annals of Neurology,46, 842–849.
Kumar, A., & Singh, A. (2015). A review on Alzheimer's disease pathophysiology and its management: An update. Pharmacological Reports,67, 195–203.
Kumar, A., Singh, P. K., Parihar, R., Dwivedi, V., Lakhotia, S. C., & Ganesh, S. (2014). Decreased O-linked GlcNAcylation protects from cytotoxicity mediated by huntingtin exon1 protein fragment. Journal of Biological Chemistry,289, 13543–13553.
Lagerlof, O., Slocomb, J. E., Hong, I., Aponte, Y., Blackshaw, S., Hart, G. W., et al. (2016). The nutrient sensor OGT in PVN neurons regulates feeding. Science,351, 1293–1296.
Laird, F. M., Cai, H., Savonenko, A. V., Farah, M. H., He, K., Melnikova, T. et al. (2005). BACE1, a major determinant of selective vulnerability of the brain to amyloid-β amyloidogenesis, is essential for cognitive, emotional, and synaptic functions. Journal of Neuroscience,25, 11693.
Lalic, N. M., Maric, J., Svetel, M., Jotic, A., Stefanova, E., Lalic, K. et al. (2008). Glucose homeostasis in Huntington disease: Abnormalities in insulin sensitivity and early-phase insulin secretion. Archives of Neurology,65, 476–480.
Lauretti, E., Li, J. G., Di Meco, A., & Praticò, D. (2017). Glucose deficit triggers tau pathology and synaptic dysfunction in a tauopathy mouse model. Translational Psychiatry,7, e1020–e1020.
Lee, J. C., Kim, S. J., Hong, S., & Kim, Y. (2019). Diagnosis of Alzheimer’s disease utilizing amyloid and tau as fluid biomarkers. Experimental & Molecular Medicine,51, 53.
Lee, T. N., Alborn, W. E., Knierman, M. D., & Konrad, R. J. (2006). Alloxan is an inhibitor of O-GlcNAc-selective N-acetyl-beta-D-glucosaminidase. Biochemical and Biophysical Research Communications,350, 1038–1043.
Lefebvre, T., Alonso, C., Mahboub, S., Dupire, M. J., Zanetta, J. P., Caillet-Boudin, M. L., et al. (1999). Effect of okadaic acid on O-linked N-acetylglucosamine levels in a neuroblastoma cell line. Biochimica et Biophysica Acta,1472, 71–81.
Lefebvre, T., Ferreira, S., Dupont-Wallois, L., Bussiere, T., Dupire, M. J., Delacourte, A. et al. (2003). Evidence of a balance between phosphorylation and O-GlcNAc glycosylation of Tau proteins: A role in nuclear localization. Biochimica et Biophysica Acta,1619, 167–176.
Levine, P. M., Galesic, A., Balana, A. T., Mahul-Mellier, A. L., Navarro, M. X., De Leon, C. A. et al. (2019). Alpha-synuclein O-GlcNAcylation alters aggregation and toxicity, revealing certain residues as potential inhibitors of Parkinson's disease. Proceedings of the National Academy of Sciences of the USA,116, 1511–1519.
Levine, Z. G., Fan, C., Melicher, M. S., Orman, M., Benjamin, T., Walker, S. (2018). O-GlcNAc transferase recognizes protein substrates using an asparagine ladder in the tetratricopeptide repeat (TPR) superhelix. Journal of the American Chemical Society,140, 3510–3513.
Levine, Z. G., & Walker, S. (2016). The Biochemistry of O-GlcNAc transferase: Which functions make it essential in mammalian cells? Annual Review of Biochemistry,85, 631–657.
Lewis, Y. E., Galesic, A., Levine, P. M., De Leon, C. A., Lamiri, N., Brennan, C. K., et al. (2017). O-GlcNAcylation of alpha-synuclein at serine 87 reduces aggregation without affecting membrane binding. ACS Chemical Biology,12, 1020–1027.
Li, M. V., Chang, B., Imamura, M., Poungvarin, N., & Chan, L. (2006a). Glucose-dependent transcriptional regulation by an evolutionarily conserved glucose-sensing module. Diabetes,55, 1179–1189.
Li, X., Lu, F., Wang, J. Z., & Gong, C. X. (2006b). Concurrent alterations of O-GlcNAcylation and phosphorylation of tau in mouse brains during fasting. European Journal of Neuroscience,23, 2078–2086.
Li, X., Molina, H., Huang, H., Zhang, Y.-Y., Liu, M., Qian, S.-W. et al. (2009). O-linked N-acetylglucosamine modification on CCAAT enhancer-binding protein beta: Role during adipocyte differentiation. Journal of Biological Chemistry,284, 19248–19254.
Li, W., Prakash, R., Chawla, D., Du, W., Didion, S. P., Filosa, J. A. et al. (2013). Early effects of high-fat diet on neurovascular function and focal ischemic brain injury. American Journal of Physiology: Regulatory, Integrative and Comparative Physiology,304, R1001–1008.
Li, W., Wang, T., & Xiao, S. (2016). Type 2 diabetes mellitus might be a risk factor for mild cognitive impairment progressing to Alzheimer's disease. Neuropsychiatric Disease and Treatment,12, 2489–2495.
Lichtenberg, B., Mandelkow, E. M., Hagestedt, T., & Mandelkow, E. (1988). Structure and elasticity of microtubule-associated protein tau. Nature,334, 359–362.
Lim, S., Haque, M. M., Nam, G., Ryoo, N., Rhim, H., & Kim, Y. K. (2015). Monitoring of intracellular tau aggregation regulated by OGA/OGT inhibitors. International Journal of Molecular Sciences,16, 20212–20224.
Lim, G. P., Yang, F., Chu, T., Chen, P., Beech, W., Teter, B. et al. (2000). Ibuprofen suppresses plaque pathology and inflammation in a mouse model for Alzheimer's disease. Journal of Neuroscience,20, 5709–5714.
Little, P. J., Drennon, K. D., & Tannock, L. R. (2008). Glucosamine inhibits the synthesis of glycosaminoglycan chains on vascular smooth muscle cell proteoglycans by depletion of ATP. Archives of Physiology and Biochemistry,114, 120–126.
Liu, F., Iqbal, K., Grundke-Iqbal, I., Hart, G. W., & Gong, C. X. (2004a). O-GlcNAcylation regulates phosphorylation of tau: A mechanism involved in Alzheimer's disease. Proceedings of the National Academy of Sciences of the USA,101, 10804–10809.
Liu, F., Shi, J., Tanimukai, H., Gu, J., Gu, J., Grundke-Iqbal, I. et al. (2009). Reduced O-GlcNAcylation links lower brain glucose metabolism and tau pathology in Alzheimer's disease. Brain,132, 1820–1832.
Liu, K., Paterson, A. J., Zhang, F., McAndrew, J., Fukuchi, K., Wyss, J. M. et al. (2004b). Accumulation of protein O-GlcNAc modification inhibits proteasomes in the brain and coincides with neuronal apoptosis in brain areas with high O-GlcNAc metabolism. Journal of Neurochemistry,89, 1044–1055.
Liu, S., Sheng, H., Yu, Z., Paschen, W., & Yang, W. (2016). O-linked beta-N-acetylglucosamine modification of proteins is activated in post-ischemic brains of young but not aged mice: Implications for impaired functional recovery from ischemic stress. Journal of Cerebral Blood Flow and Metabolism,36, 393–398.
Liu, Y., Cao, Y., Pan, X., Shi, M., Wu, Q., Huang, T. et al. (2018). O-GlcNAc elevation through activation of the hexosamine biosynthetic pathway enhances cancer cell chemoresistance. Cell Death & Disease,9, 485.
Liu, Y., Li, X., Yu, Y., Shi, J., Liang, Z., Run, X. et al. (2012). Developmental regulation of protein O-GlcNAcylation, O-GlcNAc transferase, and O-GlcNAcase in mammalian brain. PLoS ONE,7, e43724.
Liu, Y., Liu, F., Iqbal, K., Grundke-Iqbal, I., & Gong, C.-X. (2008). Decreased glucose transporters correlate to abnormal hyperphosphorylation of tau in Alzheimer disease. FEBS Letters,582, 359–364.
Livingston, G., Sommerlad, A., Orgeta, V., Costafreda, S. G., Huntley, J., Ames, D. et al. (2017). Dementia prevention, intervention, and care. Lancet,390, 2673–2734.
Loeffler, J. P., Picchiarelli, G., Dupuis, L., & De Aguilar, J. L. G. (2016). The role of skeletal muscle in amyotrophic lateral sclerosis. Brain Pathology,26, 227–236.
Loo, D. T., Copani, A., Pike, C. J., Whittemore, E. R., Walencewicz, A. J., & Cotman, C. W. (1993). Apoptosis is induced by beta-amyloid in cultured central nervous system neurons. Proceedings of the National Academy of Sciences of the USA,90, 7951–7955.
Love, D. C., Kochan, J., Cathey, R. L., Shin, S. H., & Hanover, J. A. (2003). Mitochondrial and nucleocytoplasmic targeting of O-linked GlcNAc transferase. Journal of Cell Science,116, 647–654.
Lubas, W. A., Frank, D. W., Krause, M., & Hanover, J. A. (1997). O-Linked GlcNAc transferase is a conserved nucleocytoplasmic protein containing tetratricopeptide repeats. Journal of Biological Chemistry,272, 9316–9324.
Lubas, W. A., & Hanover, J. A. (2000). Functional expression of O-linked GlcNAc transferase. Domain structure and substrate specificity. Journal of Biological Chemistry,275, 10983–10988.
Ludemann, N., Clement, A., Hans, V. H., Leschik, J., Behl, C., & Brandt, R. (2005). O-glycosylation of the tail domain of neurofilament protein M in human neurons and in spinal cord tissue of a rat model of amyotrophic lateral sclerosis (ALS). Journal of Biological Chemistry,280, 31648–31658.
Macauley, M. S., He, Y., Gloster, T. M., Stubbs, K. A., Davies, G. J., & Vocadlo, D. J. (2010). Inhibition of O-GlcNAcase using a potent and cell-permeable inhibitor does not induce insulin resistance in 3T3-L1 adipocytes. Chemistry & Biology,17, 937–948.
Macauley, M. S., & Vocadlo, D. J. (2009). Enzymatic characterization and inhibition of the nuclear variant of human O-GlcNAcase. Carbohydrate Research,344, 1079–1084.
Macauley, M. S., Whitworth, G. E., Debowski, A. W., Chin, D., & Vocadlo, D. J. (2005). O-GlcNAcase uses substrate-assisted catalysis: Kinetic analysis and development of highly selective mechanism-inspired inhibitors. Journal of Biological Chemistry,280, 25313–25322.
Manning, R. W., Reid, C. M., Lampe, R. A., & Davis, L. G. (1988). Identification in rodents and other species of an mRNA homologous to the human β-amyloid precursor. Molecular Brain Research,3, 293–297.
Marcelli, S., Corbo, M., Iannuzzi, F., Negri, L., Blandini, F., Nistico, R., et al. (2018). The involvement of post-translational modifications in Alzheimer's disease. Current Alzheimer Research,15, 313–335.
Marotta, N. P., Lin, Y. H., Lewis, Y. E., Ambroso, M. R., Zaro, B. W., Roth, M. T. et al. (2015). O-GlcNAc modification blocks the aggregation and toxicity of the protein alpha-synuclein associated with Parkinson's disease. Nature Chemistry,7, 913–920.
Marshall, S., Bacote, V., & Traxinger, R. R. (1991). Discovery of a metabolic pathway mediating glucose-induced desensitization of the glucose transport system. Role of hexosamine biosynthesis in the induction of insulin resistance. Journal of Biological Chemistry,266, 4706–4712.
Marshall, S., Nadeau, O., & Yamasaki, K. (2004). Dynamic actions of glucose and glucosamine on hexosamine biosynthesis in isolated adipocytes: Differential effects on glucosamine 6-phosphate, UDP-N-acetylglucosamine, and ATP levels. Journal of Biological Chemistry,279, 35313–35319.
Martin, L., Latypova, X., & Terro, F. (2011). Post-translational modifications of tau protein: Implications for Alzheimer's disease. Neurochemistry International,58, 458–471.
Martinez, M. R., Dias, T. B., Natov, P. S., & Zachara, N. E. (2017). Stress-induced O-GlcNAcylation: An adaptive process of injured cells. Biochemical Society Transactions,45, 237–249.
Masters, C. L., Simms, G., Weinman, N. A., Multhaup, G., McDonald, B. L., & Beyreuther, K. (1985). Amyloid plaque core protein in Alzheimer disease and down syndrome. Proceedings of the National Academy of Sciences of the USA,82, 4245–4249.
Mendez, M. F. (2017). Early-onset alzheimer disease. Neurologic Clinics,35, 263–281.
Mercier, T., Bouvet, M., Dubois-Deruy, E., Dechaumes, A., Beseme, O., Richard, V. et al. (2018). Interplay between phosphorylation and O-GlcNAcylation of sarcomeric proteins in ischemic heart failure. Frontiers in Endocrinology,9, 598–598.
Mergenthaler, P., Lindauer, U., Dienel, G. A., & Meisel, A. (2013). Sugar for the brain: The role of glucose in physiological and pathological brain function. Trend Neuroscience,36, 587–597.
Mio, T., Yabe, T., Arisawa, M., & Yamada-Okabe, H. (1998). The eukaryotic UDP-N-acetylglucosamine pyrophosphorylases. Gene cloning, protein expression, and catalytic mechanism. Journal of Biological Chemistry,273, 14392–14397.
Mooradian, A. D., & Haas, M. J. (2011). Glucose-induced endoplasmic reticulum stress is independent of oxidative stress: A mechanistic explanation for the failure of antioxidant therapy in diabetes. Free Radical Biology and Medicine,50, 1140–1143.
Moran, C., Beare, R., Phan, T. G., Bruce, D. G., Callisaya, M. L., & Srikanth, V., Alzheimer's Disease Neuroimaging. (2015). Type 2 diabetes mellitus and biomarkers of neurodegeneration. Neurology,85, 1123–1130.
Mosconi, L. (2013). Glucose metabolism in normal aging and Alzheimer's disease: Methodological and physiological considerations for PET studies. Clinical and Translational Imaging. https://doi.org/10.1007/s40336-013-0026-y.
Mosconi, L., Pupi, A., & De Leon, M. J. (2008). Brain glucose hypometabolism and oxidative stress in preclinical Alzheimer's disease. Annals of the New York Academy of Sciences,1147, 180–195.
Musuka, T. D., Wilton, S. B., Traboulsi, M., & Hill, M. D. (2015). Diagnosis and management of acute ischemic stroke: Speed is critical. CMAJ,187, 887–893.
Nagel, A. K., & Ball, L. E. (2014). O-GlcNAc transferase and O-GlcNAcase: Achieving target substrate specificity. Amino Acids,46, 2305–2316.
Neugroschl, J., & Wang, S. (2011). Alzheimer's disease: Diagnosis and treatment across the spectrum of disease severity. Mount Sinai Journal of Medicine,78, 596–612.
Neve, R. L., Harris, P., Kosik, K. S., Kurnit, D. M., & Donlon, T. A. (1986). Identification of cDNA clones for the human microtubule-associated protein tau and chromosomal localization of the genes for tau and microtubule-associated protein 2. Brain Research,387, 271–280.
Niikura, T., Tajima, H., & Kita, Y. (2006). Neuronal cell death in Alzheimer's disease and a neuroprotective factor, humanin. Current Neuropharmacology,4, 139–147.
Obeso, J. A., Rodriguez-Oroz, M. C., Goetz, C. G., Marin, C., Kordower, J. H., Rodriguez, M. et al. (2010). Missing pieces in the Parkinson's disease puzzle. Nature Medicine,16, 653–661.
O'Donnell, N., Zachara, N. E., Hart, G. W., & Marth, J. D. (2004). Ogt-dependent X-chromosome-linked protein glycosylation is a requisite modification in somatic cell function and embryo viability. Molecular and Cellular Biology,24, 1680–1690.
Okuyama, R., & Marshall, S. (2003). UDP-N-acetylglucosaminyl transferase (OGT) in brain tissue: Temperature sensitivity and subcellular distribution of cytosolic and nuclear enzyme. Journal of Neurochemistry,86, 1271–1280.
Ortiz-Meoz, R. F., Jiang, J., Lazarus, M. B., Orman, M., Janetzko, J., Fan, C. et al. (2015). A small molecule that inhibits OGT activity in cells. ACS Chemical Biology,10, 1392–1397.
Ott, A., Stolk, R. P., van Harskamp, F., Pols, H. A., Hofman, A., & Breteler, M. M. (1999). Diabetes mellitus and the risk of dementia: The Rotterdam study. Neurology,53, 1937–1942.
Pagano, G., Polychronis, S., Wilson, H., Giordano, B., Ferrara, N., Niccolini, F., et al. (2018). Diabetes mellitus and Parkinson disease. Neurology,90, e1654–e1662.
Pathak, S., Alonso, J., Schimpl, M., Rafie, K., Blair, D. E., Borodkin, V. S. et al. (2015). The active site of O-GlcNAc transferase imposes constraints on substrate sequence. Nature Structural & Molecular Biology,22, 744–750.
Peila, R., Rodriguez, B. L., & Launer, L. J. (2002). Type 2 diabetes, APOE gene, and the risk for dementia and related pathologies: The Honolulu-Asia aging study. Diabetes,51, 1256–1262.
Piaceri, I., Nacmias, B., & Sorbi, S. (2013). Genetics of familial and sporadic Alzheimer's disease. Frontiers in Bioscience,5, 167–177.
Pinho, T. S., Correia, S. C., Perry, G., Ambrósio, A. F., & Moreira, P. I. (2019). Diminished O-GlcNAcylation in Alzheimer's disease is strongly correlated with mitochondrial anomalies. Biochimica et Biophysica Acta,1865, 2048–2059.
Plassman, B. L., Langa, K. M., Fisher, G. G., Heeringa, S. G., Weir, D. R., Ofstedal, M. B. et al. (2007). Prevalence of dementia in the United States: The aging, demographics, and memory study. Neuroepidemiology,29, 125–132.
Postina, R., Schroeder, A., Dewachter, I., Bohl, J., Schmitt, U., Kojro, E. et al. (2004). A disintegrin-metalloproteinase prevents amyloid plaque formation and hippocampal defects in an Alzheimer disease mouse model. Journal of Clinical Investigation,113, 1456–1464.
Prabakaran, S., Lippens, G., Steen, H., & Gunawardena, J. (2012). Post-translational modification: Nature's escape from genetic imprisonment and the basis for dynamic information encoding. Wiley Interdisciplinary Reviews: Systems Biology and Medicine,4, 565–583.
Quaranta, D., Vita, M. G., Bizzarro, A., Masullo, C., Piccininni, C., Gainotti, G., et al. (2015). Cognitive and behavioral determinants of psychotic symptoms in Alzheimer's disease. Dementia and Geriatric Cognitive Disorders,39, 194–206.
Rajapakse, A. G., Ming, X. F., Carvas, J. M., & Yang, Z. (2009). The hexosamine biosynthesis inhibitor azaserine prevents endothelial inflammation and dysfunction under hyperglycemic condition through antioxidant effects. American Journal of Physiology-Heart and Circulatory Physiology,296, H815–822.
Roth, C., Chan, S., Offen, W. A., Hemsworth, G. R., Willems, L. I., King, D. T. et al. (2017). Structural and functional insight into human O-GlcNAcase. Nature Chemical Biology,13, 610–612.
Ruan, H.-B., Singh, J. P., Li, M.-D., Wu, J., & Yang, X. (2013). Cracking the O-GlcNAc code in metabolism. Trends in Endocrinology and Metabolism,24, 301–309.
Ryczko, M. C., Pawling, J., Chen, R., Abdel Rahman, A. M., Yau, K., Copeland, J. K. et al. (2016). Metabolic reprogramming by hexosamine biosynthetic and Golgi N-glycan branching pathways. Scientific Reports,6, 23043.
Ryu, I.-H., Lee, K.-Y., & Do, S.-I. (2016). Aβ-affected pathogenic induction of S-nitrosylation of OGT and identification of Cys-NO linkage triplet. Biochimica et Biophysica Acta,1864, 609–621.
Sadot, E., Heicklen-Klein, A., Barg, J., Lazarovici, P., & Ginzburg, I. (1996). Identification of a tau promoter region mediating tissue-specific-regulated expression in PC12 cells. Journal of Molecular Biology,256, 805–812.
Santos, A. L., & Lindner, A. B. (2017). Protein posttranslational modifications: Roles in aging and age-related disease. Oxidative Medicine and Cellular Longevity,2017, 5716409–5716409.
Schneider, L. S. (2010). The potential and limits for clinical trials for early Alzheimer's disease and some recommendations. The Journal of Nutrition, Health and Aging,14, 295–298.
Schoonenboom, N. S., Pijnenburg, Y. A., Mulder, C., Rosso, S. M., Van Elk, E. J., Van Kamp, G. J. et al. (2004). Amyloid beta(1–42) and phosphorylated tau in CSF as markers for early-onset Alzheimer disease. Neurology,62, 1580–1584.
Schroeder, J. M., Ansfield, F. J., Curreri, A. R., & Lepage, G. A. (1964). Toxicity and clinical trial of azaserine and 6-thioguanine in advanced solid malignant neoplasms. British Journal of Cancer,13, 449–458.
Schuff, N., Woerner, N., Boreta, L., Kornfield, T., Shaw, L. M., Trojanowski, J. Q., Alzheimer's Disease Neuroimaging, et al. (2009). MRI of hippocampal volume loss in early Alzheimer's disease in relation to ApoE genotype and biomarkers. Brain,132, 1067–1077.
Sekerdag, E., Solaroglu, I., & Gursoy-Ozdemir, Y. (2018). Cell death mechanisms in stroke and novel molecular and cellular treatment options. Current Neuropharmacology,16, 1396–1415.
Selnick, H. G., Hess, J. F., Tang, C., Liu, K., Schachter, J. B., Ballard, J. E. et al. (2019). Discovery of MK-8719, a potent O-GlcNAcase inhibitor as a potential treatment for tauopathies. Journal of Medicinal Chemistry,62, 10062–10097.
Shafi, R., Iyer, S. P., Ellies, L. G., O'Donnell, N., Marek, K. W., Chui, D. et al. (2000). The O-GlcNAc transferase gene resides on the X chromosome and is essential for embryonic stem cell viability and mouse ontogeny. Proceedings of the National Academy of Sciences of the USA,97, 5735–5739.
Shan, X., Vocadlo, D. J., & Krieger, C. (2012). Reduced protein O-glycosylation in the nervous system of the mutant SOD1 transgenic mouse model of amyotrophic lateral sclerosis. Neuroscience Letters,516, 296–301.
Shi, J., Gu, J. H., Dai, C. L., Gu, J., Jin, X., Sun, J. et al. (2015). O-GlcNAcylation regulates ischemia-induced neuronal apoptosis through AKT signaling. Scientific Reports,5, 14500.
Skorobogatko, Y., Landicho, A., Chalkley, R. J., Kossenkov, A. V., Gallo, G., & Vosseller, K. (2014). O-linked beta-N-acetylglucosamine (O-GlcNAc) site thr-87 regulates synapsin I localization to synapses and size of the reserve pool of synaptic vesicles. Journal of Biological Chemistry,289, 3602–3612.
Smet-Nocca, C., Broncel, M., Wieruszeski, J. M., Tokarski, C., Hanoulle, X., Leroy, A. et al. (2011). Identification of O-GlcNAc sites within peptides of the Tau protein and their impact on phosphorylation. Molecular BioSystems,7, 1420–1429.
Soldner, J., Meindl, T., Koch, W., Bokde, A. L., Reiser, M. F., Moller, H. J. et al. (2012). Structural and functional neuronal connectivity in Alzheimer's disease: A combined DTI and fMRI study. Der Nervenarzt,83, 878–887.
Squitieri, F., Cannella, M., & Simonelli, M. (2002). CAG mutation effect on rate of progression in Huntington's disease. Neurology Science,23(Suppl 2), S107–108.
Stachelek, C., Stachelek, J., Swan, J., Botstein, D., & Konigsberg, W. (1986). Identification, cloning and sequence determination of the genes specifying hexokinase A and B from yeast. Nucleic Acids Research,14, 945–963.
Stanford, P. M., Halliday, G. M., Brooks, W. S., Kwok, J. B., Storey, C. E., Creasey, H. et al. (2000). Progressive supranuclear palsy pathology caused by a novel silent mutation in exon 10 of the tau gene: Expansion of the disease phenotype caused by tau gene mutations. Brain,123(Pt 5), 880–893.
Steen, E., Terry, B. M., Rivera, E. J., Cannon, J. L., Neely, T. R., Tavares, R. et al. (2005). Impaired insulin and insulin-like growth factor expression and signaling mechanisms in Alzheimer's disease: Is this type 3 diabetes? Journal of Alzheimer's Disease,7, 63–80.
Stubbs, K. A., Macauley, M. S., & Vocadlo, D. J. (2009). A selective inhibitor Gal-PUGNAc of human lysosomal beta-hexosaminidases modulates levels of the ganglioside GM2 in neuroblastoma cells. Angewandte Chemie International Edition in English,48, 1300–1303.
Suh, H. N., Lee, Y. J., Kim, M. O., Ryu, J. M., & Han, H. J. (2014). Glucosamine-induced Sp1 O-GlcNAcylation ameliorates hypoxia-induced SGLT dysfunction in primary cultured renal proximal tubule cells. Journal of Cellular Physiology,229, 1557–1568.
Surmeier, D. J., Guzman, J. N., Sanchez-Padilla, J., & Goldberg, J. A. (2010). What causes the death of dopaminergic neurons in Parkinson's disease? Progress in Brain Research,183, 59–77.
Swamy, M., Pathak, S., Grzes, K. M., Damerow, S., Sinclair, L. V., van Aalten, D. M. F., et al. (2016). Glucose and glutamine fuel protein O-GlcNAcylation to control T cell self-renewal and malignancy. Nature Immunology,17, 712–720.
Talbot, K., Wang, H. Y., Kazi, H., Han, L. Y., Bakshi, K. P., Stucky, A. et al. (2012). Demonstrated brain insulin resistance in Alzheimer's disease patients is associated with IGF-1 resistance, IRS-1 dysregulation, and cognitive decline. Journal of Clinical Investigation,122, 1316–1338.
Tan, M. S., Tan, L., Jiang, T., Zhu, X. C., Wang, H. F., Jia, C. D., et al. (2014). Amyloid-beta induces NLRP1-dependent neuronal pyroptosis in models of Alzheimer's disease. Cell Death Disease,5, e1382.
Telenius, H., Kremer, B., Goldberg, Y. P., Theilmann, J., Andrew, S. E., Zeisler, J., et al. (1994). Somatic and gonadal mosaicism of the Huntington disease gene CAG repeat in brain and sperm. Nature Genetics,6, 409–414.
Terracciano, A., Sutin, A. R., An, Y., O'Brien, R. J., Ferrucci, L., Zonderman, A. B., et al. (2014). Personality and risk of Alzheimer's disease: New data and meta-analysis. Alzheimers Dementia,10, 179–186.
Thompson, J. W., Griffin, M. E., & Hsieh-Wilson, L. C. (2018). Methods for the detection, study, and dynamic profiling of O-GlcNAc glycosylation. Methods in Enzymology,598, 101–135.
Tian, J., Geng, Q., Ding, Y., Liao, J., Dong, M.-Q., Xu, X., et al. (2016). O-GlcNAcylation antagonizes phosphorylation of CDH1 (CDC20 homologue 1). Journal of Biological Chemistry,291, 12136–12144.
Torres, C. R., & Hart, G. W. (1984). Topography and polypeptide distribution of terminal N-acetylglucosamine residues on the surfaces of intact lymphocytes. Evidence for O-linked GlcNAc. Journal of Biological Chemistry,259, 3308–3317.
Tramutola, A., Sharma, N., Barone, E., Lanzillotta, C., Castellani, A., Iavarone, F. et al. (2018). Proteomic identification of altered protein O-GlcNAcylation in a triple transgenic mouse model of Alzheimer's disease. Biochimica et Biophysica Acta,1864, 3309–3321.
Tweedie-Cullen, R. Y., Reck, J. M., & Mansuy, I. M. (2009). Comprehensive mapping of post-translational modifications on synaptic, nuclear, and histone proteins in the adult mouse brain. Journal of Proteome Research,8, 4966–4982.
Uemura, E., & Greenlee, H. W. (2006). Insulin regulates neuronal glucose uptake by promoting translocation of glucose transporter GLUT3. Experimental Neurology,198, 48–53.
Vocadlo, D. J. (2012). O-GlcNAc processing enzymes: Catalytic mechanisms, substrate specificity, and enzyme regulation. Current Opinion in Chemical Biology,16, 488–497.
Wakabayashi, K., Tanji, K., Mori, F., & Takahashi, H. (2007). The Lewy body in Parkinson's disease: Molecules implicated in the formation and degradation of alpha-synuclein aggregates. Neuropathology,27, 494–506.
Walgren, J. L., Vincent, T. S., Schey, K. L., & Buse, M. G. (2003). High glucose and insulin promote O-GlcNAc modification of proteins, including alpha-tubulin. American Journal of Physiology, Endocrinology and Metabolism,284, E424–434.
Walker, B., Brown, M. F., Lynas, J. F., Martin, S.L., McDowell, A., Badet, B., Hill, A. J. (2001). Inhibition of Escherichia coli glucosamine synthetase by novel electrophilic analogues of glutamine comparison with 6-diazo-5-oxo-norleucine. Bioorganic & Medicinal Chemistry Letters,10(2000), 2795–2798.
Walling, A. D. (1999). Amyotrophic lateral sclerosis: Lou Gehrig's disease. American Family Physician,59, 1489–1496.
Wang, A. C., Jensen, E. H., Rexach, J. E., Vinters, H. V., & Hsieh-Wilson, L. C. (2016). Loss of O-GlcNAc glycosylation in forebrain excitatory neurons induces neurodegeneration. Proceedings of the National Academy of Sciences of the USA,113, 15120–15125.
Wang, J., Gu, B. J., Masters, C. L., & Wang, Y. J. (2017a). A systemic view of Alzheimer disease: Insights from amyloid-beta metabolism beyond the brain. Nature Reviews Neurology,13, 612–623.
Wang, P., Lazarus, B. D., Forsythe, M. E., Love, D. C., Krause, M. W., & Hanover, J. A. (2012). O-GlcNAc cycling mutants modulate proteotoxicity in Caenorhabditis elegans models of human neurodegenerative diseases. Proceedings of the National Academy of Sciences of the USA,109, 17669–17674.
Wang, S., Yang, F., Petyuk, V. A., Shukla, A. K., Monroe, M. E., Gritsenko, M. A. et al. (2017b). Quantitative proteomics identifies altered O-GlcNAcylation of structural, synaptic and memory-associated proteins in Alzheimer's disease. The Journal of Pathology,243, 78–88.
Wang, Z., Gucek, M., & Hart, G. W. (2008). Cross-talk between GlcNAcylation and phosphorylation: Site-specific phosphorylation dynamics in response to globally elevated O-GlcNAc. Proceedings of the National Academy of Sciences of the USA,105, 13793–13798.
Wani, W. Y., Chatham, J. C., Darley-Usmar, V., McMahon, L. L., & Zhang, J. (2017). O-GlcNAcylation and neurodegeneration. Brain Research Bulletin,133, 80–87.
Weingarten, M. D., Lockwood, A. H., Hwo, S. Y., & Kirschner, M. W. (1975). A protein factor essential for microtubule assembly. Proceedings of the National Academy of Sciences of the USA,72, 1858.
Weller, J., & Budson, A. (2018). Current understanding of Alzheimer's disease diagnosis and treatment. F1000 Research,7, F1000.
Wells, L., Vosseller, K., & Hart, G. W. (2001). Glycosylation of nucleocytoplasmic proteins: Signal transduction and O-GlcNAc. Science,291, 2376–2378.
Wisniewski, K. E., Wisniewski, H. M., & Wen, G. Y. (1985). Occurrence of neuropathological changes and dementia of Alzheimer's disease in Down's syndrome. Annals of Neurology,17, 278–282.
Woo, H. N., Baik, S. H., Park, J. S., Gwon, A. R., Yang, S., Yun, Y. K., et al. (2011). Secretases as therapeutic targets for Alzheimer's disease. Biochemical and Biophysical Research Communications,404, 10–15.
Woo, H. N., Park, J. S., Gwon, A. R., Arumugam, T. V., & Jo, D. G. (2009). Alzheimer's disease and Notch signaling. Biochemical and Biophysical Research Communications,390, 1093–1097.
Wu, F., Lukinius, A., Bergstrom, M., Eriksson, B., Watanabe, Y., & Langstrom, B. (1999). A mechanism behind the antitumour effect of 6-diazo-5-oxo-l-norleucine (DON): Disruption of mitochondria. European Journal of Cancer,35, 1155–1161.
Xu, J., Murphy, S.L., Kochanek, K.D., Bastian, B., Arias, E. (2018). Deaths: Final data for 2016. National Vital Statistics Reports,67, 1–76.
Xu, H., Gu, H., Yang, Y., Cai, E., Ding, F., & Yu, S. (2019). 2-(4-Methoxyphenyl)ethyl-2-acetamido-2-deoxy-beta-d-pyranoside exerts a neuroprotective effect through regulation of energy homeostasis and O-GlcNAcylation. Journal of Molecular Neuroscience,69, 177–187.
Xu, Q., Park, Y., Huang, X., Hollenbeck, A., Blair, A., Schatzkin, A., et al. (2011). Diabetes and risk of Parkinson's disease. Diabetes Care,34, 910–915.
Xu, S. L., Chalkley, R. J., Maynard, J. C., Wang, W., Ni, W., Jiang, X. et al. (2017). Proteomic analysis reveals O-GlcNAc modification on proteins with key regulatory functions in Arabidopsis. Proceedings of the National Academy of Sciences of the USA,114, e1536–e1543.
Yang, W. H., Kim, J. E., Nam, H. W., Ju, J. W., Kim, H. S., Kim, Y. S., et al. (2006). Modification of p53 with O-linked N-acetylglucosamine regulates p53 activity and stability. Nature Cell Biology,8, 1074–1083.
Yang, X., & Qian, K. (2017). Protein O-GlcNAcylation: Emerging mechanisms and functions. Nature Reviews Molecular Cell Biology,18, 452–465.
Yang, X., Zhang, F., & Kudlow, J. E. (2002). Recruitment of O-GlcNAc transferase to promoters by corepressor mSin3A: Coupling protein O-GlcNAcylation to transcriptional repression. Cell,110, 69–80.
Yang, Y. R., & Suh, P. G. (2014). O-GlcNAcylation in cellular functions and human diseases. Advances in Biological Regulation,54, 68–73.
Yang, Y. W., Hsieh, T. F., Li, C. I., Liu, C. S., Lin, W. Y., Chiang, J. H. et al. (2017). Increased risk of Parkinson disease with diabetes mellitus in a population-based study. Medicine,96, e5921.
Yao, P. J., & Coleman, P. D. (1998). Reduction of O-linked N-acetylglucosamine-modified assembly protein-3 in Alzheimer's disease. Journal of Neuroscience,18, 2399–2411.
Yki-Jarvinen, H., Vogt, C., Iozzo, P., Pipek, R., Daniels, M. C., Virkamaki, A. et al. (1997). UDP-N-acetylglucosamine transferase and glutamine: Fructose 6-phosphate amidotransferase activities in insulin-sensitive tissues. Diabetologia,40, 76–81.
Yuzwa, S. A., Macauley, M. S., Heinonen, J. E., Shan, X., Dennis, R. J., He, Y. et al. (2008). A potent mechanism-inspired O-GlcNAcase inhibitor that blocks phosphorylation of tau in vivo. Nature Chemical Biology,4, 483–490.
Yuzwa, S. A., Shan, X., Jones, B. A., Zhao, G., Woodward, M. L., Li, X. et al. (2014). Pharmacological inhibition of O-GlcNAcase (OGA) prevents cognitive decline and amyloid plaque formation in bigenic tau/APP mutant mice. Molecular Neurodegeneration,9, 42.
Yuzwa, S. A., Shan, X., Macauley, M. S., Clark, T., Skorobogatko, Y., Vosseller, K., et al. (2012). Increasing O-GlcNAc slows neurodegeneration and stabilizes tau against aggregation. Nature Chemical Biology,8, 393–399.
Yuzwa, S. A., & Vocadlo, D. J. (2014). O-GlcNAc and neurodegeneration: Biochemical mechanisms and potential roles in Alzheimer's disease and beyond. Chemical Society Reviews,43, 6839–6858.
Yuzwa, S. A., Yadav, A. K., Skorobogatko, Y., Clark, T., Vosseller, K., & Vocadlo, D. J. (2011). Mapping O-GlcNAc modification sites on tau and generation of a site-specific O-GlcNAc tau antibody. Amino Acids,40, 857–868.
Zhang, H., Gao, G., & Brunk, U. T. (1992). Extracellular reduction of alloxan results in oxygen radical-mediated attack on plasma and lysosomal membranes. Apmis.,100, 317–325.
Zhang, J., Lei, H., Chen, Y., Ma, Y. T., Jiang, F., Tan, J. et al. (2017). Enzymatic O-GlcNAcylation of alpha-synuclein reduces aggregation and increases SDS-resistant soluble oligomers. Neuroscience Letters,655, 90–94.
Zhang, X., Li, Y., Xu, H., & Zhang, Y.-W. (2014). The γ-secretase complex: From structure to function. Frontiers in Cellular Neuroscience,8, 427.
Zheng, H., & Koo, E. H. (2006). The amyloid precursor protein: Beyond amyloid. Molecular Neurodegeneration,1, 5.
Acknowledgements
This study was supported by Grants (2019R1A2C3011422, 2017M3C7A1048268, 2018M3C7A1021851) funded by the Basic Science Research Program through the National Research Foundation of Korea (NRF), the Ministry of Education, Science and Technology, Republic of Korea. The work was also supported by a grant to Korea Polar Research Institute (KOPRI) under project (PE20010).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare no competing financial interests or any conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Park, J., Lai, M.K.P., Arumugam, T.V. et al. O-GlcNAcylation as a Therapeutic Target for Alzheimer’s Disease. Neuromol Med 22, 171–193 (2020). https://doi.org/10.1007/s12017-019-08584-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12017-019-08584-0