Abstract
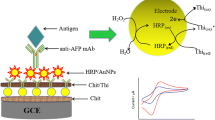
In the current study, an electrochemical biosensing signal amplification system was utilized with thionine-chitosan-gold nanoparticles (Chit-GNPs) that absorbed horseradish peroxidase (HRP) and anti-His tagged protein monoclonal antibody derived from Balb/c mice. In addition, transmission electron microscopy (TEM) was used to characterize the nanogold solution and atomic force microscopy (AFM) was used to characterize the sensor assembly. To evaluate the quality of the immunosensor, the amperometric I-t curve method was applied to determine His-IL23 in PBS. The results indicated that the response current exhibited an optimal linear correlation with the His-IL23 concentration that ranged from 0.01 to 103 ng/ml. The lowest detection limit was noted at 3.3 pg/ml (S/N = 3). The linear equation was deduced as follows: △I = 0.02lgC + 0.037 (R2 = 0.9628). Moreover, it was validated with high sensitivity, reproducibility and rapid response. Apparently, the immunosensor may be a very useful tool for the detection and quantification of His-tagged proteins. In addition, the signal amplification system can be used for the preparation of other immunosensors and to assist in bioassays.






Similar content being viewed by others
Abbreviations
- Ab:
-
antibody
- Ag:
-
antigen
- BSA:
-
bovine serum albumin
- ELISA:
-
enzyme-linked immunosorbent assay
- LOD:
-
the limit of detection
- mAb:
-
monoclonal antibodies
- His-tags:
-
Poly-histidine tags
- PBS:
-
phosphate bufered saline
- PBS-T:
-
0.01 M PBS contains 0.05%(v/v) Tween-20
- GCE:
-
glassy carbon electrode
- Chit:
-
chitosan
- GNPs:
-
Gold nanoparticles
- HRP:
-
horseradish peroxidase
- Thi:
-
thionine
- NTA:
-
nitrilotriacetic acid
- TEM:
-
transmission electron microscope
- AFM:
-
atomic force microscope
- SPR:
-
surface plasmon resonance
- CV:
-
cyclic voltammetry
- Ra:
-
average roughness
- R:
-
the correlation coefcient value
- SD:
-
standard deviation
References
P. Ball, L. Garwin, Science at the atomic scale. Nature 355, 761–766 (1992). https://doi.org/10.1038/355761a0
B. Brizzard, R. Chubet, Epitope Tagging of Recombinant Proteins (Current Protocols in Neuroscience, John Wiley & Sons, Inc, 2001). https://doi.org/10.1002/0471142301.ns0508s00
R.E. Cavicchi, R.H. Silsbee, Coulomb suppression of tunneling rate from small metal particles. Phys. Rev. Lett. 52, 1453–1456 (1984). https://doi.org/10.1103/PhysRevLett.52.1453
F. Darain, C. Ban, Y.B. Shim, Development of a new and simple method for the detection of histidine-tagged proteins. Biosens. Bioelectron. 20, 857–863 (2004). https://doi.org/10.1016/j.bios.2004.03.028
N. Debeljak, L. Feldman, K.L. Davis, R. Komel, A.J. Sytkowski, Variability in the immunodetection of his-tagged recombinant proteins. Anal. Biochem. 359, 216–223 (2006). https://doi.org/10.1016/j.ab.2006.09.017
P.K. Deshmukh, K.P. Ramani, S.S. Singh, A.R. Tekade, V.K. Chatap, G.B. Patil, S.B. Bari, Stimuli-sensitive layer-by-layer (lbl) self-assembly systems: Targeting and biosensory applications. J. Control. Release 166, 294–306 (2013). https://doi.org/10.1016/j.jconrel.2012.12.033
G. Frens, Controlled nucleation for the regulation of the particle size in monodisperse gold suspensions. Nat. Phys. Sci. 241, 20–22 (1973). https://doi.org/10.1038/physci241020a0
L. Gerbino, J.S. Riva, M.C. Strumia, A.M. Baruzzi, Thionine immobilized in crosslinked chitosan films. Sensors Actuators B Chem. 131, 455–461 (2008). https://doi.org/10.1016/j.snb.2007.12.006
L. Ji, Z.K. Guo, T. Yan, H.M. Ma, B. Du, Y.Y. Li, Q. Wei, Ultrasensitive sandwich-type electrochemical immunosensor based on a novel signal amplification strategy using highly loaded palladium nanoparticles/carbon decorated magnetic microspheres as signal labels. Biosens. Bioelectron. 68, 757–762 (2015). https://doi.org/10.1016/j.bios.2015.02.010
S. Kang, K.K. Shiu, Scanning tunneling microscopic and Voltammetric studies of the surface structures of an electrochemically activated glassy carbon electrode. Anal. Chem. 74, 879–885 (2002). https://doi.org/10.1021/ac010734+
X.B. Kang, G.C. Pang, Q.S. Chen, X.Y. Liang, Fabrication of bacillus cereus, electrochemical immunosensor based on double-layer gold nanoparticles and chitosan. Sensors Actuators B Chem. 177, 1010–1016 (2013). https://doi.org/10.1016/j.snb.2012.12.018
X.B. Kang, G.C. Pang, X.Y. Liang, M. Wang, J. Liu, W.M. Zhu, Study on a hydrogen peroxide biosensor based on horseradish peroxidase/GNPs-thionine/chitosan. Electrochim. Acta 62, 327–334 (2012). https://doi.org/10.1016/j.electacta.2011.12.034
C. Ley, D. Holtmann, K.M. Mangold, J. Schrader, Immobilization of histidine-tagged proteins on electrodes. Colloids Surf. B: Biointerfaces 88(2), 539–551 (2011). https://doi.org/10.1016/j.colsurfb.2011.07.044
R.P. Liang, J.D. Qiu, P.X. Cai, A novel amperometric immunosensor based on three-dimensional sol–gel network and nanoparticle self-assemble technique. Anal. Chim. Acta 534, 223–229 (2005). https://doi.org/10.1016/j.aca.2004.11.064
G.Z. Liu, W.Q. Guo, D.D. Song, A multianalyte electrochemical immunosensor based on patterned carbon nanotubes modified substrates for detection of pesticides. Biosens. Bioelectron. 52(9), 360–366 (2014). https://doi.org/10.1016/j.bios.2013.09.009
D.Q. Lu, F.P. Lu, G.C. Pang, A novel glutathione-s transferase immunosensor based on horseradish peroxidase and double-layer gold nanoparticles. Biomed. Microdevice. 18(3), 2–9 (2016). https://doi.org/10.1007/s10544-016-0075-x
H.H. Ma, J.Z. Sun, Y. Zhang, T. Zhen, Label-free immunosensor based on one-step electrodeposition of chitosan-gold nanoparticles biocompatible film on au microelectrode for determination of aflatoxin b1, in maize. Biosens. Bioelectron. 80, 222–229 (2016). https://doi.org/10.1016/j.bios.2016.01.063
X. Mao, J.H. Jiang, J.W. Chen, R.Q. Yu, Cyclic accumulation of nanoparticles: A new strategy for electrochemical immunoassay based on the reversible reaction between dethiobiotin and avidin. Anal. Chim. Acta 557(1), 159–163 (2006). https://doi.org/10.1016/j.aca.2005.09.078
G. Merola, E. Martini, M. Tomassetti, L.G. Campanella, Simple and suitable immunosensor for β-lactam antibiotics analysis in real matrixes: Milk, serum, urine. J. Pharmaceut. Biomed. 106, 186–196 (2014). https://doi.org/10.1016/j.jpba.2014.08.005
M. Moreno-Guzmán, I. Ojeda, R. Villalonga, A. González-Cortés, P. Yáñez-Sedeño, J.M. Pingarrón, Ultrasensitive detection of adrenocorticotropin hormone (acth) using disposable phenylboronic-modified electrochemical immunosensors. Biosens. Bioelectron. 35, 82–86 (2012). https://doi.org/10.1016/j.bios.2012.02.015
T. Nagaoka, T. Yoshino, Surface properties of electrochemically pretreated glassy carbon. Anal. Chem. 58(6), 1037–1042 (1986). https://doi.org/10.1021/ac00297a012
M.O. Noor, U.J. Krull, Silicon nanowires as field-effect transducers for biosensor development: A review. Anal. Chim. Acta 825, 1–25 (2014). https://doi.org/10.1016/j.aca.2014.03.016
A.S. Pina, C.R. Lowe, A.C.A. Roque, Challenges and opportunities in the purification of recombinant tagged proteins. Biotechnol. Adv. 32(2), 366–381 (2014). https://doi.org/10.1016/j.biotechadv.2013.12.001
V.K. Rao, G.P. Rai, G.S. Agarwal, S. Suresh, Amperometric immunosensor for detection of antibodies of salmonella typhi, in patient serum. Anal. Chim. Acta 531(2), 173–177 (2005). https://doi.org/10.1016/j.aca.2004.10.015
R. Ravikumar, L.H. Chen, P. Jayaraman, C.L. Poh, C.C. Chan, Chitosan-nickel film based interferometric optical fiber sensor for label-free detection of histidine tagged proteins. Biosens. Bioelectron. 99, 578–585 (2017). https://doi.org/10.1016/j.bios.2017.08.012
X. Ren, P. Lu, R. Feng, T. Zhang, Y. Zhang, D. Wu, Q. Wei, An ITO-based point-of-care colorimetric immunosensor for ochratoxin a detection. Talanta 188, 593–599 (2018). https://doi.org/10.1016/j.talanta.2018.06.004
X. Ren, H. Ma, T. Zhang, Y. Zhang, T. Yan, B. Du, Q. Wei, A sulfur-doped graphene-based immunological biosensing platform for multianalysis of cancer biomarkers. ACS Appl. Mater. Interfaces 9, 37637–37644 (2017a). https://doi.org/10.1021/acsami.7b13416
X. Ren, J. Yan, D. Wu, Q. Wei, Y. Wan, Nanobody-based apolipoprotein E immunosensor for point-of-care testing. ACS Sensors 2, 1267–1271 (2017b). https://doi.org/10.1021/acssensors.7b00495
X. Ren, T. Zhang, D. Wu, T. Yan, X. Pang, B. Du, W. Lou, Q. Wei, Increased electrocatalyzed performance through high content potassium doped graphene matrix and aptamer tri infinite amplification labels strategy: Highly sensitive for matrix metalloproteinases-2 detection. Biosens. Bioelectron. 94, 694–700 (2017c). https://doi.org/10.1016/j.bios.2017.03.064
K.G. Shevchenko, V.R. Cherkasov, A.A. Tregubov, P.I. Nikitin, M.P. Nikitin, Surface plasmon resonance as a tool for investigation of non-covalent nanoparticle interactions in heterogeneous self-assembly & disassembly systems. Biosens. Bioelectron. 88, 3–8 (2017). https://doi.org/10.1016/j.bios.2016.09.042
G.B. Sigal, C. Bamdad, A. Barberis, J. Strominger, G.M. Whitesides, A self-assembled monolayer for the binding and study of histidine-tagged proteins by surface plasmon resonance. Anal. Chem. 68(3), 490–497 (1996). https://doi.org/10.1021/ac9504023
A. Spriestersbach, J. Kubicek, F. Schäfer, H. Block, B. Maertens, Purification of his-tagged proteins. Method. Enzymol. 559, 1–15 (2015). https://doi.org/10.1016/bs.mie.2014.11.003
S.A. Tria, D. Lopez-Ferber, C. Gonzalez, I. Bazin, A. Guiseppi-Elie, Microfabricated biosensor for the simultaneous amperometric and luminescence detection and monitoring of ochratoxin a. Biosens. Bioelectron. 79(1), 835–842 (2016). https://doi.org/10.1016/j.bios.2016.01.018
J.Q. Tang, G.C. Pang, J.R. Gao, X.Y. Liang, Development of casein immunosensor modified by chitosan and nano-gold. Food Sci. Biotech. 29, 533–53 (2011). http://en.cnki.com.cn/Article_en/CJFDTOTAL-SPKX201104063.htm
A. Tinazli, J. Tang, R. Valiokas, S. Picuric, S. Lata, J. Piehler, B. Liedberg, R. Tampé, High-affinity chelator thiols for switchable and oriented immobilization of histidine-tagged proteins: A generic platform for protein chip technologies. Chem. Eur. J. 11(18), 5249–5259 (2005). https://doi.org/10.1002/chem.200500154
M. Varga, Chapter 3–Self-assembly of nanobiomaterials, in: Grumezescu, A., (Eds.), Fabrication and Self-Assembly of Nanobiomaterials: Applications of Nanobiomaterials Volume 1, William Andrew pub., pp.57–90, (2016). https://doi.org/10.1016/B978-0-323-41533-0.00003-9
X.Y. Wang, G.C. Pang, Amplification systems of weak interaction biosensors: Applications and prospects. Sensor Rev. 35(1), 30–42 (2015). https://doi.org/10.1108/SR-03-2014-629
Y.L. Wang, X.J. Li, W. Cao, Y.Y. Li, H. Li, B. Du, Q. Wei, Ultrasensitive sandwich-type electrochemical immunosensor based on a novel signal amplification strategy using highly loaded toluidine blue/gold nanoparticles decorated kit-6/carboxymethyl chitosan/ionic liquids as signal labels. Biosens. Bioelectron. 61(1), 618–624 (2014). https://doi.org/10.1016/j.bios.2014.05.059
M. Wąsowicz, S. Viswanathan, A. Dvornyk, K. Grzelak, B. Kłudkiewicz, H. Radeck, Comparison of electrochemical immunosensors based on gold nano materials and immunoblot techniques for detection of histidine-tagged proteins in culture medium. Biosens. Bioelectron. 24(2), 284–289 (2008). https://doi.org/10.1016/j.bios.2008.04.002
T. Yang, S. Wang, H.L. Jin, W.W. Bao, S.M. Huang, J.C. Wang, An electrochemical impedance sensor for the label-free ultrasensitive detection of interleukin-6 antigen. Sensors Actuators B Chem. 178(3), 310–315 (2013). https://doi.org/10.1016/j.snb.2012.12.107
T. Zhang, N. Ma, A. Ali, Q. Wei, D. Wu, X. Ren, Electrochemical ultrasensitive detection of cardiac troponin i using covalent organic frameworks for signal amplification. Biosens. Bioelectron. 119, 176–181 (2018a). https://doi.org/10.1016/j.bios.2018.08.020
T. Zhang, B. Xing, Q. Han, Y. Lei, D. Wu, X. Ren, Q. Wei, Electrochemical immunosensor for ochratoxin a detection based on au octahedron plasmonic colloidosomes. Anal. Chim. Acta 1032, 114–121 (2018b). https://doi.org/10.1016/j.aca.2018.05.035
Acknowledgments
This research was financially supported by National Natural Science Foundation of China (Grant No. 31671857, 31901782).
Author information
Authors and Affiliations
Contributions
Guangchang Pang conceived of and designed the experiments. Dingqiang Lu performed the experiments and wrote the paper. Ruijuan Ren participated in the basic part of the experiment, including the establishment of electrochemical signal amplification systems and data analysis, as well as editing and modification of the manuscript.
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare no conflict of interest.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Ren, R., Lu, D. & Pang, G. Development of a new and simple method for the detection of histidine-tagged proteins based on thionine-chitosan/gold nanoparticles/horseradish peroxidase. Biomed Microdevices 22, 11 (2020). https://doi.org/10.1007/s10544-019-0464-z
Published:
DOI: https://doi.org/10.1007/s10544-019-0464-z