Abstract
Background
In the medicolegal literature, focal concavities or notching of the corpus callosum has been thought to be associated with fetal alcohol spectrum disorders. Recent work suggests corpus callosum notching is a dynamic and normal anatomical feature, although it has not yet been defined in early life or infancy.
Objective
Our purpose was to characterize the dorsal contour of the corpus callosum during the first 2 years of life by defining the prevalence, onset and trajectory of notching on midsagittal T1-weighted images.
Materials and methods
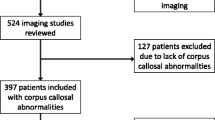
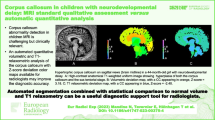
We reviewed retrospectively 1,157 consecutive patients between birth and 2 years of age. Corpus callosum morphology was evaluated and described. A notch was defined as a dorsal concavity of at least 1 mm in depth along the dorsal surface of the corpus callosum. Patient age as well as notch depth, location, number and presence of the pericallosal artery in the notch were noted.
Results
Two hundred thirty-three notches were identified in 549 patients: 36 anterior, 194 posterior and 3 patients with undulations. A statistically significant (R2=0.53, Beta=0.021, P=0.002) positive correlation between posterior notch prevalence and age in months was noted. A positive correlation between age and depth of the posterior notch was also statistically significant (r=0.32, n=179, P≤0.001). A trend for increased anterior notch prevalence with age was identified with significant correlation between visualized pericallosal artery indentation and anterior notching (r=0.20, n=138, P=0.016). Sub-analysis of the first month of life showed corpus callosum notching was not present.
Conclusion
The presence of posterior notching increased significantly with age and was more frequent than that of anterior notching. Corpus callosum notching was absent in the first week of life, building on prior studies suggesting corpus callosum notching is acquired. This study provides baseline data on normative corpus callosum notching trajectories by age group during early life, a helpful correlate when associating corpus callosum morphology with disease.





Similar content being viewed by others
References
Meissner TW, Friedrich P, Ocklenburg S et al (2017) Tracking the functional development of the corpus callosum in children using behavioral and evoked potential interhemispheric transfer times. Dev Neuropsychol 42:172–186
Lenroot RK, Giedd JN (2006) Brain development in children and adolescents: insights from anatomical magnetic resonance imaging. Neurosci Biobehav Rev 30:718–729
Luders E, Thompson PM, Toga AW (2010) The development of the corpus callosum in the healthy human brain. J Neurosci 30:10985–10990
Edwards TJ, Sherr EH, Barkovich AJ, Richards LJ (2014) Clinical, genetic and imaging findings identify new causes for corpus callosum development syndromes. Brain 137(Pt 6):1579–1613
Ture U, Yasargil MG, Krisht AF (1996) The arteries of the corpus callosum: a microsurgical anatomic study. Neurosurgery 39:1075–1084
Kahilogullari G, Comert A, Ozdemir M et al (2013) Arterial vascularization patterns of the splenium: an anatomical study. Clin Anat 26:675–681
Sakai T, Mikami A, Suzuki J et al (2017) Developmental trajectory of the corpus callosum from infancy to the juvenile stage: comparative MRI between chimpanzees and humans. PLoS One 12:e0179624
Hasan KM, Kamali A, Iftikhar A et al (2009) Diffusion tensor tractography quantification of the human corpus callosum fiber pathways across the lifespan. Brain Res 1249:91–100
Hasan KM, Kamali A, Kramer LA et al (2008) Diffusion tensor quantification of the human midsagittal corpus callosum subdivisions across the lifespan. Brain Res 1227:52–67
Imperati D, Colcombe S, Kelly C et al (2011) Differential development of human brain white matter tracts. PLoS One 6:e23437
Lebel C, Beaulieu C (2011) Longitudinal development of human brain wiring continues from childhood into adulthood. J Neurosci 31:10937–10947
McLaughlin NC, Paul RH, Grieve SM et al (2007) Diffusion tensor imaging of the corpus callosum: a cross-sectional study across the lifespan. Int J Dev Neurosci 25:215–221
Pujol J, Vendrell P, Junque C et al (1993) When does human brain development end? Evidence of corpus callosum growth up to adulthood. Ann Neurol 34:71–75
Ren T, Anderson A, Shen WB et al (2006) Imaging, anatomical, and molecular analysis of callosal formation in the developing human fetal brain. Anat Rec A Discov Mol Cell Evol Biol 288:191–204
Rakic P, Yakovlev PI (1968) Development of the corpus callosum and cavum septi in man. J Comp Neurol 132:45–72
Luders E, Narr KL, Bilder RM et al (2007) Positive correlations between corpus callosum thickness and intelligence. Neuroimage 37:1457–1464
Andronikou S, Pillay T, Gabuza L et al (2015) Corpus callosum thickness in children: an MR pattern-recognition approach on the midsagittal image. Pediatr Radiol 45:258–272
Malavolti AM, Chau V, Brown-Lum M et al (2017) Association between corpus callosum development on magnetic resonance imaging and diffusion tensor imaging, and neurodevelopmental outcome in neonates born very preterm. Dev Med Child Neurol 59:433–440
Cantlon JF, Davis SW, Libertus ME et al (2011) Inter-parietal white matter development predicts numerical performance in young children. Learn Individ Differ 21:672–680
Fornari E, Knyazeva MG, Meuli R, Maeder P (2007) Myelination shapes functional activity in the developing brain. Neuroimage 38:511–518
Hynd GW, Semrud-Clikeman M, Lorys AR et al (1991) Corpus callosum morphology in attention deficit-hyperactivity disorder: morphometric analysis of MRI. J Learn Disabil 24:141–146
Belmonte M, Egaas B, Townsend J, Courchesne E (1995) NMR intensity of corpus callosum differs with age but not with diagnosis of autism. Neuroreport 6:1253–1256
David AS (1993) Callosal transfer in schizophrenia: too much or too little? J Abnorm Psychol 102:573–579
Raine A, Harrison GN, Reynolds GP et al (1990) Structural and functional characteristics of the corpus callosum in schizophrenics, psychiatric controls, and normal controls. A magnetic resonance imaging and neuropsychological evaluation. Arch Gen Psychiatry 47:1060–1064
Riley EP, Mattson SN, Sowell ER et al (1995) Abnormalities of the corpus callosum in children prenatally exposed to alcohol. Alcohol Clin Exp Res 19:1198–1202
Donald KA, Eastman E, Howells FM et al (2015) Neuroimaging effects of prenatal alcohol exposure on the developing human brain: a magnetic resonance imaging review. Acta Neuropsychiatr 27:251–269
Roebuck TM, Mattson SN, Riley EP (1998) A review of the neuroanatomical findings in children with fetal alcohol syndrome or prenatal exposure to alcohol. Alcohol Clin Exp Res 22:339–344
Hoyme HE, Kalberg WO, Elliott AJ et al (2016) Updated clinical guidelines for diagnosing fetal alcohol spectrum disorders. Pediatrics:138. https://doi.org/10.1542/peds.2015-4256
Bocci T, Caleo M, Giorli E et al (2011) Transcallosal inhibition dampens neural responses to high contrast stimuli in human visual cortex. Neuroscience 187:43–51
Bocci T, Caleo M, Restani L et al (2016) Altered recovery from inhibitory repetitive transcranial magnetic stimulation (rTMS) in subjects with photosensitive epilepsy. Clin Neurophysiol 127:3353–3361
Unterberger I, Bauer R, Walser G, Bauer G (2016) Corpus callosum and epilepsies. Seizure 37:55–60
Mardia K, Bookstein F, Kent J (2013) Alcohol, babies and the death penalty: saving lives by analysing the shape of the brain. Significance 10:12–16
Bookstein FL, Connor PD, Huggins JE et al (2007) Many infants prenatally exposed to high levels of alcohol show one particular anomaly of the corpus callosum. Alcohol Clin Exp Res 31:868–879
Krause KL, Howard D, Pettersson DR et al (2019) Defining the normal dorsal contour of the corpus callosum with time. AJNR Am J Neuroradiol 40:86–91
Garel C, Cont I, Alberti C et al (2011) Biometry of the corpus callosum in children: MR imaging reference data. AJNR Am J Neuroradiol 32:1436–1443
Westerhausen R, Fjell AM, Krogsrud SK et al (2016) Selective increase in posterior corpus callosum thickness between the age of 4 and 11 years. Neuroimage 139:17–25
Giedd JN, Blumenthal J, Jeffries NO et al (1999) Development of the human corpus callosum during childhood and adolescence: a longitudinal MRI study. Prog Neuro-Psychopharmacol Biol Psychiatry 23:571–588
Keshavan MS, Diwadkar VA, DeBellis M et al (2002) Development of the corpus callosum in childhood, adolescence and early adulthood. Life Sci 70:1909–1922
Bullitt E, Zeng D, Mortamet B et al (2010) The effects of healthy aging on intracerebral blood vessels visualized by magnetic resonance angiography. Neurobiol Aging 31:290–300
May PA, Baete A, Russo J et al (2014) Prevalence and characteristics of fetal alcohol spectrum disorders. Pediatrics 134:855–866
May PA, Gossage JP, Kalberg WO et al (2009) Prevalence and epidemiologic characteristics of FASD from various research methods with an emphasis on recent in-school studies. Dev Disabil Res Rev 15:176–192
Fenlon LR, Richards LJ (2015) Contralateral targeting of the corpus callosum in normal and pathological brain function. Trends Neurosci 38:264–272
Tovar-Moll F, Monteiro M, Andrade J et al (2014) Structural and functional brain rewiring clarifies preserved interhemispheric transfer in humans born without the corpus callosum. Proc Natl Acad Sci U S A 111:7843–7848
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflicts of interest
None
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Simpson, L.N., Schneble, E.J., Griffin, E.D. et al. Morphological changes of the dorsal contour of the corpus callosum during the first two years of life. Pediatr Radiol 50, 543–549 (2020). https://doi.org/10.1007/s00247-019-04585-0
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00247-019-04585-0