Abstract
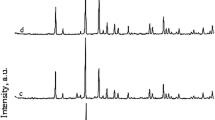
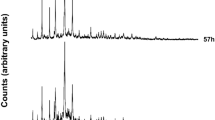
Nano sized ZSM-5 zeolite samples were synthesized successively from kaolin clay as alumina source having a large amount of quartz (39%) and silicic acid as silica source by hydrothermal treatment with NaOH in the presence of tetrapropylammonium hydroxide as a template. Then the effect of kaolin content, crystallization temperature and time on the size and crystallinity of the products were investigated. The prepared samples were characterized using XRD, SEM, EDS and FT-IR techniques. The results showed that the synthesized ZSM-5 zeolite samples were almost pure and their crystallization was almost complete. The average particle size, as determined by Debye-Scherrer equation, was in the range of 20-42 nm. Increasing kaolin content on crystal size was more effective than increase in crystallization temperature and time. Additional evidences for the nano sized ZSM-5 zeolite were the asymmetric stretch vibration band at 1225 cm-1 in the FT-IR spectra and TEM images. The scanning electron micrographs of the synthesized zeolites showed that they are spherical shape crystals.
Similar content being viewed by others
Refrences
R.J. Argauer, G.R. Landolt, US Patent 3,702,886 (1972).
C. Falamaki, M. Edrissi, M. Sohrabi, Zeolite 19 (1997) 2.
N. Kumar, V. Nieminen, K. Demirkan, T. Salmi, D.Y. Murzin, E. Laine, Appl. Catal. A-Gen. 235 (2002) 113.
E. Davis, R. Lubo, Chem. Mater. 4 (1992) 756.
L.F. Petrik, C.T. Oconnor, S. Schwar, in: H.K. Beyer, H.G. Karage, I. Kiricsi, J.B. Nagy (Eds.), Catalysis by Microporous Materials, Studies in Surface Science and Catalysis, Vol. 94, Elsevier, 1995, p. 517.
A. Nastro, Z. Gabelica, P. Bodart, J.B. Nagy, in: S. Kaliiaguine, A. Mabay (Eds.), Catalysis on Theenergyscene, Studies in Surface Science and Catalysis, 19, Elsevier, 1984, p. 138.
B.M. Lowe, J.R.D. Nee, J.L. Casi, Zeolite 14 (1994) 610.
D.T. Hayburst, G. Evanina, F. Hung, J. Chem. 64 (1990) 295.
M. Padovan, G. Leofanti, M. Solari, E. Moretti, Zeolites 4 (1984) 295.
E.G. Derouane, Z. Gabelica, J. Solid State Chem. 64 (1986) 296.
B.M. Lowe, in: P.J. Grobet, W.J. Mortier, E.F. Vansant, G. Schulz-Ekloff (Eds.), Innovation in Zeolite Materials Science, Studies in surface Science and Catalysis, 37, Elsevier, 1998, p. 1.
L. Tosheva, V.P. Valtchev, Chem. Mater. 17 (2005) 2494.
J. Miller, US Patent 00555 88 51 A.
M. Khatamian, M. Dolatyari, Asian J. Chem. 19 (2007) 5199.
R.M. Mohamed, O.A. Fouad, A.A. Ismail, I.A. Ibrahim, Mater. Lett. 59 (2005) 3441.
Y. Chang, L. Jun, J.-S. Li, Y.-C. Yang, X.-Y. Sun, Mater. Lett. 59 (2005) 3427.
D.W. Breck, Zeolite Molecular Sieves, Willey, New York, 1974.
J. Aguado, D.P. Serrano, J.M. Escola, J.M. Rodriguez, Micropor. Mesopor. Mater. 75 (2004) 41.
S.L. Burkett, M.E. Daivis, J. Phys. Chem. B 98 (1994) 4647.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Khatamian, M., Irani, M. Preparation and characterization of nanosized ZSM-5 zeolite using kaolin and investigation of kaolin content, crystallization time and temperature changes on the size and crystallinity of products. JICS 6, 187–194 (2009). https://doi.org/10.1007/BF03246519
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF03246519