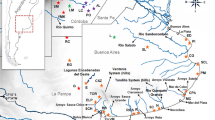
Abstract
The genus Ctenomys comprises approximately 70 recognized living species of subterranean rodents endemic to South America. Phylogenetic studies to date, based on mitochondrial DNA data, place 44 recognized species in eight species groups and provide evidence for a burst of speciation early in the history of the genus. Species from Argentina, Uruguay, Bolivia, and southern Brazil are well studied at the phylogenetic level. However, the taxonomic status of the species inhabiting midwest and northern Brazil remains poorly understood. In this study, we construct phylogenies based on maximum likelihood and Bayesian inference methods with cytochrome b gene haplotypes of Ctenomys from midwest and northern Brazil and with haplotypes representative of the genus Ctenomys to place the sampled haplotypes into a phylogenetic framework; we also evaluate skull geometric morphometrics data among sampling sites to assess whether skull morphology corroborates the phylogenetic patterns observed. The results show that the sampling sites used in this study are represented by two species, namely, Ctenomys bicolor, which is present in the state of Rondônia, and Ctenomys nattereri, which is present in Mato Grosso and Bolivia. The results also reveal two lineages of Ctenomys distinct from C. bicolor and C. nattereri, henceforth called Ctenomys sp. “xingu” and Ctenomys sp. “central.” Both the species and lineages share a most recent common ancestor with C. boliviensis and are part of the boliviensis species group.





Similar content being viewed by others
References
Ackerly DD, Thomas WMW, Ferreira CAC, Pirani JR (1989) The forest – cerrado transition zone in southern Amazonia: results of the 1985 Projeto Flora Amazonica Expedition to Mato Grosso. Brittonia 41(2): 113–128
Adams DC, Otárola-Castillo E (2013) Geomorph: an r package for the collection and analysis of geometric morphometric shape data. Methods Ecol Evol 4:393–399
Anderson S, Yates TL, Cook JA (1987) Notes on Bolivian mammals, 4: the genus Ctenomys (Rodentia: Ctenomyidae) in the eastern lowlands. Am Mus Novitates 2891:1–20
Avise JC (1994) Molecular Markers, Natural History, and Evolution. Chapman & Hall, New York
Avise JC (2000) Phylogeography: The History and Formation of Species. Harvard University Press, Cambridge
Baird NA, Etter PD, Atwood TS, Currey MC, Shiver AL, Lewis ZA, Selker EU, Cresko WA, Johnson EA (2008) Rapid SNP discovery and genetic mapping using sequenced RAD markers. PLoS One 3(10) e3776. https://doi.org/10.1371/journal.pone.0003376
Ballard JWO, Whitlock MC (2004) The incomplete natural history of mitochondria. Mol Ecol 13: 729–744. https://doi.org/10.1046/j.1365-294X.2003.02063.x
Bidau CJ, Ávila-Pires FD (2009) On the type locality of Ctenomys bicolor Miranda Ribeiro, 1914 (Rodentia: Ctenomyidae). Mastozool neotrop 16:445–447.
Bidau CJ (2015) Family Ctenomyidae. In: Patton, JL, Pardiñas, UFJ, D’Elía G (eds) Mammals of South America Vol. 2: Rodents. University of Chicago Press, Chicago
Bookstein FL (1991) Morphometric Tools for Landmark Data: Geometry and Biology. Cambridge University Press, London
Borges LR, Maestri R, Kubiak BB, Galiano D, Fornel R, Freitas TRO (2017) The role of soil features in shaping the bite force and related skull and mandible morphology in the subterranean rodents of genus Ctenomys (Hystricognathi: Ctenomyidae). J Zool 301:108–117. https://doi.org/10.1111/jzo.12398
Bouckaert R, Heled J, Kühnert D, Vaughan T, Wu CH, Xie D, Suchard MA, Rambaut A, Drummond AJ (2014) BEAST 2: a software platform for Bayesian evolutionary analysis. PLoS Comput Biol 10(4): e1003537. https://doi.org/10.1371/jornal.pcbi.1003537
Cariou M, Duret L, Charlat S (2013) Is RAD-seq suitable for phylogenetic inference? An in silico assessment and optimization. Ecol Evol 3(4): 846–852. https://doi.org/10.1002/ece3.512
Castillo AH, Cortinas MN, Lessa EP (2005) Rapid diversification of South American tuco-tucos (Ctenomys; Rodentia, Ctenomyidae): contrasting mitochondrial and nuclear intron suquences. J Mammal 86:170–179
Cheng L, Connor TR, Aanensen, Spratt BG, Corander J (2011) Bayesian semi-supervised classification of bacterial samples using MLST databases. BMC Bioinformatics 12:302
Corander J, Marttinen P, Mäntyniemi S (2006) Bayesian identification of stock mixtures from molecular marker data. Fishery Bull 104: 550–558
Corander J, Marttinen P, Sirén J, Tang J (2008) Enhanced Bayesian modelling in BAPS software for learning genetic structures of populations. BMC Bioinformatics 9:539
Darriba D, Taboada GL, Doallo R, Posada D (2012) jModelTest 2: more models, new heuristics and parallel computing. Nature Methods 9(8): 772
Davey JW, Hohenlohe PA, Etter PD, Boone JQ, Catchen JM, Blaxter ML (2011) Genome-wide genetic marker discovery and genotyping using next-generation sequencing. Nature Reviews Genetics 12(7):499–510. https://doi.org/10.1038/nrg3012
D’Elía G, Lessa EP, Cook JA (1999) Molecular phylogeny of tuco-tucos, genus Ctenomys (Rodentia, Octodontidae): evaluation of the mendocinus species group and the evolution of asymmetric sperm. J Mammal Evol 6: 19–38
Doyle JJ, Doyle JL (1987) A rapid DNA isolation procedure for small quantities of fresh leaf tissue. Phytochem Bull 19:11–15
Dryden IL, Mardia KV (1998) Statistical Shape Analysis. John Wiley & Sons, New York
Drummond AJ, Ho SYW, Phillips MJ, Rambaut A (2006) Relaxed phylogenetics and Dating with Confidence. PLoS Biol 4:e88
Edgar R (2004) MUSCLE: multiple sequence alignment with high accuracy and high throughput. Nucleic Acids Res 32:1792–1797
Emerson KJ, Merz CR, Catchen JM, Hohenlohe PA, Cresko WA, Bradshaw WE, Holzapfel CM (2010) Resolving postglacial phylogeography using high-throughput sequencing. Proc Natl Acad Sci USA 107 (37):16196–16200. https://doi.org/10.1073/pnas.1006538107
Fernandes FA, Fornel R, Cordeiro-Estrela P, Freitas TRO (2009) Intra- and interespecific skull variation in two sister species of the subterranean rodent genus Ctenomys (Rodentia, Ctenomyidae): coupling geometric morphometrics and chromosomal polymorphism. Zool J Linn Soc 155: 220–237
Freitas TRO (2001) Tuco-tucos (Rodentia, Octodontidae) in southern Brazil: Ctenomys lami spec. nov. separated from C. minutus Nehring 1887. Stud Neotrop Fauna Environ 36: 1–8
Freitas TRO (2005) Analysis of skull morphology in 15 species of the genus Ctenomys, including seven karyologically distinct forms of Ctenomys minutus (Rodentia: Ctenomyidae). In: Lacey EA, Myers P (eds) Mammalian Diversification: From Chromosomes to Phylogeography (A Celebration of the Career of James L. Patton). Univ Calif Publ Zool 133:131–154
Freitas TRO (2016) Family Ctenomyidae. In: Wilson DE, Lacher TE Jr, Mittermeier RA (eds) Handbook of the Mammals of the World: Lagomorphs and Rodents I, Vol. 6. Lynx Editions, Barcelona, pp 499–534
Freitas TRO, Fernandes FA, Fornel R, Roratto PA (2012) An endemic new species of tuco-tuco, genus Ctenomys (Rodentia: Ctenomyidae), with a restricted geographic distribution in southern Brazil. J Mammal 93:1355–1367
Gardner SL, Salazar-Bravo J, Cook JA (2014) New species of Ctenomys (Rodentia: Ctenomyidae) from the lowlands and central valleys of Bolivia. Spec Publ Mus Texas Tech Univ 62:1–34
Guindon S, Gascuel O (2003) A simple, fast and accurate method to estimate large phylogenies by maximum-likelihood. Syst Biol 52:696–704
Hasegawa M, Kishino H, Yano T (1985) Dating the human-ape splitting by a molecular clock of mitochondrial DNA. J Mol Evol 22:160–174
Heled J, Drummond AJ (2011) Calibrated tree priors for relaxed phylogenetics and divergence time estimation. Syst Biol 61:138–149
IBGE (2004) Instituto Brasileiro de Geografia e Estatística. Maps of Biomes and Vegetation. Available at: ftp://ftp.ibge.gov.br/Cartas_e_Mapas/Mapas_Murais/
Kent JT, Mardia KV (2001) Shape, Procrustes tangent projections and bilateral symmetry. Biometrika 88 (2):469–485. https://doi.org/10.1093/biomet/88.2.469
Klingenberg CP (2011) MorphoJ: an integrated software package for geometric morphometrics. Mol Ecol Resources 11: 353–357
Klingenberg CP, Barluenga M, Meyer A (2002) Shape analysis of symmetric structures: quantifying variation among individuals and asymmetry. Evolution 56:1909–1920
Lacey EA, Patton JL, Cameron GN (2000) Life Underground: The Biology of Subterranean Rodents. University of Chicago Press, Chicago and London, 449 pp
Lessa EP, Cook JA (1998) The molecular phylogenetics of tuco-tucos (genus Ctenomys, Rodentia: Octodontidae) suggests an early burst of speciation. Mol Phylogenet Evol 9 (1):88–99
Librado P, Rozas J (2009) DnaSP v5: a software for comprehensive analysis of DNA polymorphism data. Bioinformatics 25:1451–1452
Lichtenstein H (1830) Darstellung neuer oder wenig bekannter. Saugethiere in Abbildung und Beschriebungen. Luderitz edit, Berlin (1827–31)
Lopes CM, Ximenes SSF, Gava A, Freitas TRO (2013) The role of chromosomal rearrangements and geographical barriers in the divergence of lineages in a South American subterranean rodent (Rodentia: Ctenomyidae: Ctenomys minutus). Heredity 111:293–305. https://doi.org/10.1038/hdy.2013.49
Maestri R, Fornel R, Gonçalves GL, Geise L, Freitas TRO, Carnaval AC (2016) Predictors of intraspecific morphological variability in a tropical hotspot: comparing the influence of random and non-random factors. J Biogeogr 43:2160–2172
Manthey JD, Moyle RG (2015) Isolation by environment in White-breasted Nuthatches (Sitta carolinensis) of the Madrean Archipelago sky islands: a landscape genomics approach. Mol Ecol 24 (14): 3628–3638. https://doi.org/10.1111/mec.13258
Marcy AE, Hadly EA, Sherratt E, Garland K, Weisbecker V (2016) Getting a head in hard soils: convergent skull evolution and divergent allometric patterns explain shape variation in a highly diverse genus of pocket gophers (Thomomys). BMC Evol Biol 16:207. https://doi.org/10.1186/s12862-016-0782-1
Marimon BS, Marimon-Junior BH, Feldpausch TR, Oliveira-Santos C, Mews HA, Lopez-Gonzalez G, Lloyd J, Franczak DD, Oliveira EA, Maracahipes L, Miguel A, Lenza E, Phillips OL (2013) Disequilibrium and hyperdynamic tree turnover at the forest–cerrado transition zone in southern Amazonia. Plant Ecol Diversity 7(1–2):281–292. https://doi.org/10.1080/17550874.2013.818072.
Marshall LG, Sempere T (1993) Evolution of the Neotropical Cenozoic land mammal fauna in its geochronologic, stratigraphic, and tectonic context. In: Goldblatt P (ed) Biological Relationships Between Africa and South America. Yale University Press, New Haven, pp 329–392
Mascheretti S, Mirol P, Gimenez M, Bidau C, Contreras J, Searle J (2000) Phylogenetics of the speciose and chromosomally variable genus Ctenomys (Ctenomyidae, Octodontoidea), based on mitochondrial cytochrome b sequences. Biol J Linn Soc 70:361–376
Miranda-Ribeiro A De (1914) Zoologia. Commisão de Linhas Telegráphicas Estratégicas de MattoGrosso ao Amazonas. Annexo 5, Historia Natural; publ no 17, Mammíferos. 49 pp + Append, 3 pp + 25 pls
Moritz C (1994) Defining ‘evolutionary significant units’ for conservation. Trends Ecol Evol 9 (10):373–375
Neigel JE, Avise JC (1987) Phylogenetic relationships of mitochondrial DNA under various demographic models of speciation. In: Nevo E, Karlin S (eds) Evolutionary Processes and Theory. Academic Press, New York, pp 515–534. https://doi.org/10.1016/B978-0-12-398760-0.50026-2
Nowak RM (1999) Walker’s Mammals of the World. 6th edition, volume 2. Johns Hopkins University Press, Baltimore
Parada A, D’Elía G, Bidau CJ, Lessa EP (2011) Species groups and the evolutionary diversification of tuco-tucos, genus Ctenomys (Rodentia: Ctenomyidae). J Mammal 92(3):671–682
Patton JL, Pardiñas UFJ, D’Elía, G (2015) Mammals of South America, Vol. 2. University of Chicago Press, Chicago
Peterson BK, Weber JN, Kay EH, Fisher HS, Hoekstra HE (2012) Double digest RADseq: an inexpensive method for de novo SNP discovery and genotyping in model and non-model species. PLoS One 7(5)e37135. doi:https://doi.org/10.1371/journal.pone.0037135
R Core Team (2017) R: a language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria. URL https://www.R-project.org/
Rambaut A, Drummond AJ (2015a) LogCombiner v1.8.2. http://beast.bio.ed.ac.uk
Rambaut A, Drummond AJ (2015b) TreeAnnotator v1.8.2: MCMC Output analysis. http://beast.bio.ed.ac.uk
Rambaut A, Suchard M A, Xie W, Drummond AJ (2013) Tracer v.1.6.0: MCMC Trace Analysis Tool. http://beast.bio.ed.ac.uk
Reig OA, Busch C, Ortells MO, Contreras JR (1990) An overview of evolution, systematics, population biology, cytogenetics, molecular biology and speciation in Ctenomys. In: Nevo E, Reig OA (eds) Evolution of Subterranean Mammals at the Organismal and Molecular Levels. Wiley- Liss, New York, pp 71–96
Rohlf FJ (2010) TPSDig 2.16. Stony Brook New York: Department of Ecology and Evolution, State University of New York at Stony Brook
Sikes RS, the Animal Care and Use Committee of the American Society of Mammalogists (2016) 2016 Guidelines of the American Society of Mammalogists for the use of wild mammals in research and education. J Mammal 97(3):663–688
Samuels JX (2009) Cranial morphology and dietary habits of rodents. Zool J Linn Soc 156:864–888
Slamovits CH, Cook JA, Lessa EP, Rossi MS (2001) Recurrent amplifications and deletions of satellite DNA accompanied chromosomal diversification in South American tuco-tucos (Genus Ctenomys, Rodentia: Octodontidae): a phylogenetic approach. Mol Biol Evol 18:1708–1719
Smith MF, Patton JL (1999) Phylogenetic relationships and the radiation of sigmodontine rodents in South America: evidence from cytochrome b. J Mammal Evol 6(2): 89–128 S
Stein B (2000) Morphology of subterranean rodents. In: Lacey AP, Patton JL, Cameron GN (eds) Life Underground: The Biology of Subterranean Rodents. University of Chicago Press, Chicago, pp 19–60
Stolz JFB, Gonçalves GL, Leipnitz L, Freitas TRO (2013) DNA-based and geometric morphometric analysis to validate species designation: a case study of the subterranean rodent Ctenomys bicolor. Genet Mol Res 12(4): 5023–5037
Tamura K, Stecher G, Peterson D, Filipski A, Kumar S (2013) MEGA6: molecular evolutionary genetics analysis version 6.0. Mol Biol Evol 30:2725–2729
Tobe SS, Kitchener AC, Linacre AMT (2010) Reconstructing mammalian phylogenies: a detailed comparison of the cytochrome b and cytochrome oxidase subunit I mitochondrial genes. PLoS One 5(11): e14156. https://doi.org/10.1371/journal.pone.0014156
Travi VH (1981) Nota prévia sobre nova espécie do gênero Ctenomys Blainville, 1826 (Rodentia: Ctenomyidae). Iheringia 60: 123–124
Upham NS, Patterson BD (2012) Diversification and biogeography of the Neotropical caviomorph lineage Octodontoidea (Rodentia: Hystrichognathi). Mol Phylogenet Evol 60-2: 417–429. https://doi.org/10.1016/j.ympev.2012.01.020
Verzi, DH (2002) Patrones de evolución morfológica en Ctenomyinae (Rodentia, Octodontidae). Mastozool neotrop 9:309–328.
Verzi DH, Olivares AI, Morgan CC (2009) The oldest South American tuco-tuco (late Pliocene, northwestern Argentina) and the boundaries of the genus Ctenomys (Rodentia, Ctenomyidae). Mammal Biol 75:243–252
Vucetich MG, Verzi DH, Hartenberger JL (1999) Review and analysis of the radiation of the South American Hystricognathi (Mammalia, Rodentia). Paleontology 329:763–769
Wagner A (1848) Beiträge zur Kenntniss der Arten von Ctenomys. Archiv für Naturgeschichte 14:72–78
Wlasiuk G, Garza JC, Lessa EP (2003) Genetic and geographic differentiation in the Rio Negro tuco-tuco (Ctenomys rionegrensis): inferring the roles of migration and drift from multiple genetic markers. Evolution 57:913–926
Woods CA, Kilpatrick CW (2005) Infraorder Hystricognathi. In: Wilson DE, Reeder DM (eds) Mammal Species of the World. (3rd ed.). Johns Hopkins University Press, Baltimore, pp 1538–1600
Zenuto RR, Busch C (1998) Population biology of the subterranean rodent Ctenomys australis (tuco-tuco) in a coastal dunefield in Argentina. Z Säugetierk 63:357–367
Acknowledgments
We thank all colleagues from the Laboratory of Cytogenetics and Evolution (LACE) for their invaluable support in reviewing this manuscript. We also thank Professors Andreia Turchetto, Gislene Lopes Gonçalves, and Renan Maestri for reviewing the methods, results, and discussion sections of this paper. L.T.L. and L.E.J.R. received scholarships from the Comissão de Aperfeiçoamento de Pessoal de Nível Técnico e Superior (CAPES), and T.R.O.F. received research grants from CAPES, Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq) and Fundação de Amparo à Pesquisa do Estado do Rio Grande do Sul (FAPERGS).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Leipnitz, L.T., Fornel, R., Ribas, L.E.J. et al. Lineages of Tuco-Tucos (Ctenomyidae: Rodentia) from Midwest and Northern Brazil: Late Irradiations of Subterranean Rodents Towards the Amazon Forest. J Mammal Evol 27, 161–176 (2020). https://doi.org/10.1007/s10914-018-9450-0
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10914-018-9450-0