Abstract
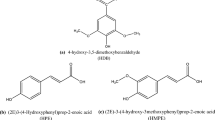
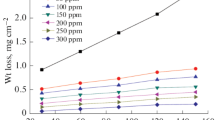
The aim of this research is evaluation of the performance of Pistacia khinjuk aerial part extract (PKAPE) on the corrosion of carbon steel in 2.0 M hydrochloric acid and 1.0 M sulfuric acid solutions and comparing the differences in the behavior of the extract in these two acidic environments. For this study, the classical method of weight loss and electrochemical methods [open-circuit potential (OCP) time, potentiodynamic polarization, and electrochemical impedance spectroscopy (EIS)] were used. Based on weight loss experiments, the maximum inhibition efficiencies of 92.8% and 90.3% were obtained at 2.0 g/L of extract in 2.0 M HCl and 1.0 M H2SO4 solutions, respectively. Potentiodynamic polarization results revealed that PKAPE in both examined acidic solutions act as mixed-type inhibitor. Polarization measurements in the temperature range of 25–65 °C were carried out for different concentration to define the effect of temperature on the behavior of the extract. At all investigated temperatures, adsorption of PKAPE on the surface for both acidic media follows the Langmuir adsorption isotherm. Thermodynamic and activation parameters were calculated, and the results inferred that the adsorption of PKAPE on the surface for both acidic solutions is physisorption. Quantum chemical parameters were calculated using the density functional theory (DFT) method for main components of PKAPE to predict their adsorption on the surface.









Similar content being viewed by others
References
Abdallah M, Kamar EM, Eid S, El-Etre AY (2016) Animal glue as green inhibitor for corrosion of aluminum and aluminum-silicon alloys in sodium hydroxide solutions. J Mol Liq 220:755–761. https://doi.org/10.1016/j.molliq.2016.04.062
Afia L et al (2012) Argan hulls extract: green inhibitor of mild steel corrosion in 1 M HCl solution. Res Chem Intermed 38:1707–1717. https://doi.org/10.1007/s11164-012-0496-y
Ali SA, Saeed M, Rahman S (2003) The isoxazolidines: a new class of corrosion inhibitors of mild steel in acidic medium. Corros Sci 45:253–266. https://doi.org/10.1016/S0010-938X(02)00099-9
Becke AD (1993) A new mixing of Hartree-Fock and local density-functional theories. J Chem Phys 98:1372–1377. https://doi.org/10.1063/1.464304
Behpour M, Ghoreishi S, Khayatkashani M, Soltani N (2011) The effect of two oleo-gum resin exudate from Ferula assa-foetida and Dorema ammoniacum on mild steel corrosion in acidic media. Corros Sci 53:2489–2501. https://doi.org/10.1016/j.corsci.2011.04.005
Bentiss F, Traisnel M, Lagrenee M (2000) The substituted 1, 3, 4-oxadiazoles: a new class of corrosion inhibitors of mild steel in acidic media. Corros Sci 42:127–146. https://doi.org/10.1016/S0010-938X(99)00049-9
Bentiss F, Lebrini M, Traisnel M, Lagrenée M (2009) Synergistic effect of iodide ions on inhibitive performance of 2, 5-bis (4-methoxyphenyl)-1, 3, 4-thiadiazole during corrosion of mild steel in 0.5 M sulfuric acid solution. J Appl Electrochem 39:1399–1407. https://doi.org/10.1007/s10800-009-9810-9
Bozorgi M, Memariani Z, Mobli M, Salehi Surmaghi MH, Shams-Ardekani MR, Rahimi R (2013) Five Pistacia species (P. vera, P. atlantica, P. terebinthus, P. khinjuk, and P. lentiscus): a review of their traditional uses, phytochemistry, and pharmacology. Sci World J. https://doi.org/10.1155/2013/219815
de Souza FS, Giacomelli C, Gonçalves RS, Spinelli A (2012) Adsorption behavior of caffeine as a green corrosion inhibitor for copper. Mater Sci Eng C 32:2436–2444. https://doi.org/10.1016/j.msec.2012.07.019
Deyab MA (2015) Egyptian licorice extract as a green corrosion inhibitor for copper in hydrochloric acid solution. J Ind Eng Chem 22:384–389. https://doi.org/10.1016/j.jiec.2014.07.036
Gaussian09 RA, 1, MJ Frisch, GW Trucks, HB Schlegel, GE Scuseria, MA Robb, JR Cheeseman, G. Scalmani, V. Barone, B. Mennucci, GA Petersson et al., Gaussian Inc, Wallingford CT (2009)
Gerengi H, Sahin HI (2011) Schinopsis lorentzii extract as a green corrosion inhibitor for low carbon steel in 1 M HCl solution. Ind Eng Chem Res 51:780–787. https://doi.org/10.1021/ie201776q
Gopiraman M, Sakunthala P, Kesavan D, Alexramani V, Kim I, Sulochana N (2012) An investigation of mild carbon steel corrosion inhibition in hydrochloric acid medium by environment friendly green inhibitors. J Coat Technol Res 9:15–26. https://doi.org/10.1007/s11998-011-9374-6
Guo L, Kaya S, Obot IB, Zheng X, Qiang Y (2017) Toward understanding the anticorrosive mechanism of some thiourea derivatives for carbon steel corrosion: a combined DFT and molecular dynamics investigation. J Colloid Interface Sci 506:478–485. https://doi.org/10.1016/j.jcis.2017.07.082
Hamani H, Douadi T, Daoud D, Al-Noaimi M, Rikkouh RA, Chafaa S (2017) 1-(4-Nitrophenylo-imino)-1-(phenylhydrazono)-propan-2-one as corrosion inhibitor for mild steel in 1 M HCl solution: weight loss, electrochemical, thermodynamic and quantum chemical studies. J Electroanal Chem 801:425–438. https://doi.org/10.1016/j.jelechem.2017.08.031
Hong S, Chen W, Zhang Y, Luo HQ, Li M, Li NB (2013) Investigation of the inhibition effect of trithiocyanuric acid on corrosion of copper in 3.0 wt% NaCl. Corros Sci 66:308–314. https://doi.org/10.1016/j.corsci.2012.09.034
Ji G, Anjum S, Sundaram S, Prakash R (2015) Musa paradisica peel extract as green corrosion inhibitor for mild steel in HCl solution. Corros Sci 90:107–117. https://doi.org/10.1016/j.corsci.2014.10.002
Jokar M, Farahani TS, Ramezanzadeh B (2016) Electrochemical and surface characterizations of morus alba pendula leaves extract (MAPLE) as a green corrosion inhibitor for steel in 1 M HCl. J Taiwan Inst Chem Eng 63:436–452. https://doi.org/10.1016/j.jtice.2016.02.027
Jr OLR (1973) Corrosion inhibitors, 2nd edn. C.C. Nathan, Houston, TX
Ju H, Kai Z-P, Li Y (2008) Aminic nitrogen-bearing polydentate Schiff base compounds as corrosion inhibitors for iron in acidic media: a quantum chemical calculation. Corros Sci 50:865–871. https://doi.org/10.1016/j.corsci.2007.10.009
Kaczerewska O, Leiva-Garcia R, Akid R, Brycki B, Kowalczyk I, Pospieszny T (2018) Effectiveness of O-bridged cationic gemini surfactants as corrosion inhibitors for stainless steel in 3 M HCl: experimental and theoretical studies. J Mol Liq 249:1113–1124. https://doi.org/10.1016/j.molliq.2017.11.142
Kamal C, Sethuraman M (2013) Hydroclathrus clathratus marine alga as a green inhibitor of acid corrosion of mild steel. Res Chem Intermed 39:3813–3828. https://doi.org/10.1007/s11164-012-0883-4
Kawashty S, Mosharrafa S, El-Gibali M, Saleh N (2000) The flavonoids of four Pistacia species in Egypt. Biochem Syst Ecol 28:915–917. https://doi.org/10.1016/S0305-1978(99)00113-1
Khadraoui A, Khelifa A, Hamitouche H, Mehdaoui R (2014) Inhibitive effect by extract of Mentha rotundifolia leaves on the corrosion of steel in 1 M HCl solution. Res Chem Intermed 40:961–972. https://doi.org/10.1007/s11164-012-1014-y
Kıcır N, Tansuğ G, Erbil M, Tüken T (2016) Investigation of ammonium (2,4-dimethylphenyl)-dithiocarbamate as a new, effective corrosion inhibitor for mild steel. Corros Sci 105:88–99. https://doi.org/10.1016/j.corsci.2016.01.006
Kumar KV, Pillai MSN, Thusnavis GR (2011) Green corrosion inhibitor from seed extract of Areca catechu for mild steel in hydrochloric acid medium. J Mater Sci 46:5208–5215. https://doi.org/10.1007/s10853-011-5457-0
Kumar R, Yadav OS, Singh G (2017) Electrochemical and surface characterization of a new eco-friendly corrosion inhibitor for mild steel in acidic media: a cumulative study. J Mol Liq 237:413–427. https://doi.org/10.1016/j.molliq.2017.04.103
Lee C, Yang W, Parr RG (1988) Development of the Colle-Salvetti correlation-energy formula into a functional of the electron density. Phys Rev B 37:785–789. https://doi.org/10.1103/PhysRevB.37.785
Li W, Hu L, Zhang S, Hou B (2011) Effects of two fungicides on the corrosion resistance of copper in 3.5% NaCl solution under various conditions. Corros Sci 53:735–745. https://doi.org/10.1016/j.corsci.2010.11.006
Li L, Zhang X, Lei J, He J, Zhang S, Pan F (2012a) Adsorption and corrosion inhibition of Osmanthus fragran leaves extract on carbon steel. Corros Sci 63:82–90. https://doi.org/10.1016/j.corsci.2012.05.026
Li X, Deng S, Fu H (2012b) Inhibition of the corrosion of steel in HCl, H2SO4 solutions by bamboo leaf extract. Corros Sci 62:163–175. https://doi.org/10.1016/j.corsci.2012.05.008
Liao LL, Mo S, Lei JL, Luo HQ, Li NB (2016) Application of a cosmetic additive as an eco-friendly inhibitor for mild steel corrosion in HCl solution. J Colloid Interface Sci 474:68–77. https://doi.org/10.1016/j.jcis.2016.04.015
Martinez S, Stern I (2002) Thermodynamic characterization of metal dissolution and inhibitor adsorption processes in the low carbon steel/mimosa tannin/sulfuric acid system. Appl Surf Sci 199:83–89. https://doi.org/10.1016/S0169-4332(02)00546-9
Messali M, Lgaz H, Dassanayake R, Salghi R, Jodeh S, Abidi N, Hamed O (2017) Guar gum as efficient non-toxic inhibitor of carbon steel corrosion in phosphoric acid medium: electrochemical, surface, DFT and MD simulations studies. J Mol Struct 1145:43–54. https://doi.org/10.1016/j.molstruc.2017.05.081
Njoku DI, Onuoha GN, Oguzie EE, Oguzie KL, Egbedina AA, Alshawabkeh AN (n.d.) (2016) Nicotiana tabacum leaf extract protects aluminium alloy AA3003 from acid attack. Arab J Chem. https://doi.org/10.1016/j.arabjc.2016.07.017
Njoku DI, Oguzie EE, Li Y (2017) Characterization, electrochemical and theoretical study of the anticorrosion properties of Moringa oleifera extract. J Mol Liq 237:247–256. https://doi.org/10.1016/j.molliq.2017.04.087
Noor EA (2009) Evaluation of inhibitive action of some quaternary N-heterocyclic compounds on the corrosion of Al–Cu alloy in hydrochloric acid. Mater Chem Phys 114:533–541. https://doi.org/10.1016/j.matchemphys.2008.09.065
Obot I, Obi-Egbedi N (2010) An interesting and efficient green corrosion inhibitor for aluminium from extracts of Chlomolaena odorata L. in acidic solution. J Appl Electrochem 40:1977–1984. https://doi.org/10.1007/s10800-010-0175-x
Odewunmi NA, Umoren SA, Gasem ZM (2015a) Watermelon waste products as green corrosion inhibitors for mild steel in HCl solution. J Environ Chem Eng 3:286–296. https://doi.org/10.1016/j.jece.2014.10.014
Odewunmi NA, Umoren SA, Gasem ZM, Ganiyu SA, Muhammad Q (2015b) L-Citrulline: an active corrosion inhibitor component of watermelon rind extract for mild steel in HCl medium. J Taiwan Inst Chem Eng 51:177–185. https://doi.org/10.1016/j.jtice.2015.01.012
Pavithra M, Venkatesha T, Vathsala K, Nayana K (2010) Synergistic effect of halide ions on improving corrosion inhibition behaviour of benzisothiozole-3-piperizine hydrochloride on mild steel in 0.5 MH2SO4 medium. Corros Sci 52:3811–3819. https://doi.org/10.1016/j.corsci.2010.07.034
Pearson RG (1988) Absolute electronegativity and hardness: application to inorganic chemistry. Inorg Chem 27:734–740. https://doi.org/10.1021/ic00277a030
Prabhu R, Venkatesha T, Shanbhag A (2009) Carmine and fast green as corrosion inhibitors for mild steel in hydrochloric acid solution. J Iran Chem Soc 6:353–363. https://doi.org/10.1007/BF03245845
Prathibha BS, Nagaswarupa HP, Kotteeswaran P, BheemaRaju V (2017) Inhibiting effect of quaternary ammonium compound on the corrosion of mild steel in 1 M hydrochloric acid solution, its adsorption and kinetic characteristics. Mater Today 4:12245–12254. https://doi.org/10.1016/j.matpr.2017.09.156
Quraishi MA (2014) 2-Amino-3, 5-dicarbonitrile-6-thio-pyridines: new and effective corrosion inhibitors for mild steel in 1 M HCl. Ind Eng Chem Res 53:2851–2859. https://doi.org/10.1021/ie401633y
Saha SK, Murmu M, Murmu NC, Obot IB, Banerjee P (2018) Molecular level insights for the corrosion inhibition effectiveness of three amine derivatives on the carbon steel surface in the adverse medium: a combined density functional theory and molecular dynamics simulation study. Surf Interfaces 10:65–73. https://doi.org/10.1016/j.surfin.2017.11.007
Saranya J, Sowmiya M, Sounthari P, Parameswari K, Chitra S, Senthilkumar K (2016) N-heterocycles as corrosion inhibitors for mild steel in acid medium. J Mol Liq 216:42–52. https://doi.org/10.1016/j.molliq.2015.12.096
Solmaz R, Kardaş G, Yazıcı B, Erbil M (2008) Adsorption and corrosion inhibitive properties of 2-amino-5-mercapto-1, 3, 4-thiadiazole on mild steel in hydrochloric acid media. Colloid Surface A 312:7–17
Soltani N, Tavakkoli N, Khayatkashani M, Jalali MR, Mosavizade A (2012) Green approach to corrosion inhibition of 304 stainless steel in hydrochloric acid solution by the extract of Salvia officinalis leaves. Corros Sci 62:122–135. https://doi.org/10.1016/j.corsci.2012.05.003
Soltani N, Tavakkoli N, Kashani MK, Mosavizadeh A, Oguzie E, Jalali M (2014) Silybum marianum extract as a natural source inhibitor for 304 stainless steel corrosion in 1.0 M HCl. J Ind Eng Chem 20:3217–3227. https://doi.org/10.1016/j.jiec.2013.12.002
Taran M, Sharifi M, Azizi E, Khanahmadi M (2010) Antimicrobial activity of the leaves of Pistacia khinjuk. J Med Plants 1:81–85
Umoren S, Eduok U, Israel A, Obot I, Solomon M (2012) Coconut coir dust extract: a novel eco-friendly corrosion inhibitor for Al in HCl solutions. Green Chem Lett Rev 5:303–313. https://doi.org/10.1080/17518253.2011.625980
Umoren S, Obot IB, Gasem Z, Odewunmi NA (2015) Experimental and theoretical studies of red apple fruit extract as green corrosion inhibitor for mild steel in HCl solution. J Dispers Sci Technol 36:789–802. https://doi.org/10.1080/01932691.2014.922887
Verma C, Quraishi M, Ebenso E, Obot I, El Assyry A (2016) 3-Amino alkylated indoles as corrosion inhibitors for mild steel in 1 M HCl: experimental and theoretical studies. J Mol Liq 219:647–660. https://doi.org/10.1016/j.molliq.2016.04.024
Yang W, Mortier WJ (1986) The use of global and local molecular parameters for the analysis of the gas-phase basicity of amines. J Am Chem Soc 108:5708–5711. https://doi.org/10.1021/ja00279a008
Acknowledgements
The financial support of the research council of Payame Noor University of Isfahan and the Payame Noor University of Delijan is gratefully acknowledged.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Soltani, N., Tavakkoli, N., Attaran, A. et al. Inhibitory effect of Pistacia khinjuk aerial part extract for carbon steel corrosion in sulfuric acid and hydrochloric acid solutions. Chem. Pap. 74, 1799–1815 (2020). https://doi.org/10.1007/s11696-019-01026-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11696-019-01026-y