Abstract
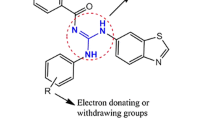
Gallic acid (GA) and its benzamide derivatives have a wide variety of biological activities, such as antimicrobial, antioxidant, anticancer. In this study, we have reported the synthesis of some new hybrid compounds comprised of the 2-aminothiophene and GA moieties and evaluation of their cytotoxic activities against HeLa (cervical cancer), HCT116 (human colon cancer), and FT (fibroblast) cell lines as well as antimicrobial activities against some Gram-positive and Gram-negative bacteria. The reaction of some 2-aminothiophene derivatives (previously prepared from the Gewald reaction) with galloyl chloride having the acetylated hydroxyl groups and the subsequent deprotection of the hydroxyl groups gave the desired hybrid compounds. Then, the antimicrobial activity of the compounds was evaluated using disc diffusion and minimum inhibitory concentration assays. Finally, the MTT assay was carried out to evaluate the cytotoxicity of the synthesized compounds on the mentioned cell lines. The structure of the synthesized compounds was elucidated by conventional spectroscopic methods such as NMR, FT-IR, and UV–Vis spectroscopy. All compounds prevented the growth of Staphylococcus coagulase more than the positive control of chloramphenicol, and one compound was more sensitive to the growth of Klebsiella pneumonia compared to the standard antibiotic. All compounds showed acceptable activity against cancer cells. The highest activity was observed against HeLa with an IC50 value of 3.2 μg/mL for compound 3d and against HCT116 with IC50 of 59.4 μg/mL for 3b. The high anticancer activity of compound 3d against HeLa allows us to consider it as a good lead compound for the development of new potent anticancer agents for the treatment of cervical cancer.
Graphic abstract





Similar content being viewed by others
References
K. Khanbabaee, T. van Ree, Nat. Prod. Rep. 18, 641 (2001)
A. Borges, M.J. Saavedra, M. Simõcs, Biofouling 28, 755 (2012)
H. Masaki, N. Okamoto, S. Sakaki, H. Sakurai, Biol. Pharm. Bull. 20, 304 (1997)
E. Klein, N. Weber, J. Agric. Food Chem. 49, 1224 (2001)
A. Serrano, C. Palacios, G. Roy, C. Cespón, M.L. Villar, M. Nocito, P. González-Porqué, Arch. Biochem. Biophys. 350, 49 (1998)
K. Isuzugawa, Y. Ogihara, M. Inoue, Biol. Pharm. Bull. 24, 249 (2001)
M. Inoue, R. Suzuki, T. Koide, N. Sakaguchi, Y. Ogihara, Y. Yabu, Biochem. Biophys. Res. Commun. 204, 898 (1994)
M. Inoue, N. Sakaguchi, K. Isuzugawa, H. Tani, Y. Ogihara, Biol. Pharm. Bull. 23, 1153 (2000)
S.M. Fiuza, C. Gomes, L.J. Teixeira, M.T. Girão da Cruz, M.N.D.S. Cordeiro, N. Milhazes, F. Borgesa, M.P.M. Marques, Bioorg. Med. Chem. 12, 3581 (2004)
C. Palacios, C. Cespón, C. MartÍn de la Vega, G. Roy, A. Serrano, M. Salinas, P. González-Porqué, J. Enzyme Inhib. 16, 527 (2001)
R.W. Sabnis, D.W. Rangwekar, N.D. Sonawane, J. Heterocycl. Chem. 36, 333 (1999)
K. Bozorov, L.F. Nie, J. Zhao, H.A. Aisa, Aisa Eur. J. Med. Chem. 140, 465 (2017)
H.Y. Meltzer, H.C. Fibiger, Neuropsychopharmacology 14, 83 (1996)
K. Bozorov, H.R. Ma, J.Y. Zhao, H.Q. Zhao, H. Chen, K. Bobakulov, X.L. Xin, B. Elmuradov, K. Shakhidoyatov, H.A. Aisa, Eur. J. Med. Chem. 84, 739 (2014)
K. Bozorov, L.F. Zhao Jy Nie, H.R. Ma, K. Bobakulov, R. Hu, N. Rustamova, G. Huang, T. Efferth, H.A. Aisa, RSC Adv. 7, 31417 (2017)
S. Al-Mousawi, M. El-Apasery, H. Mahmoud, Molecules 18, 7081 (2013)
L. Scotti, M.T. Scotti, E.D.O. Lima, M.S.D. Silva, M.D.C.A.D. Lima, I.D.R. Pitta, R.O.D. Moura, J.G.B.D. Oliveira, R.M.D.D. Cruz, F.J.B. Mendonça-Junior, Molecules 17, 2298 (2012)
J. Desantis, G. Nannetti, S. Massari, M.L. Barreca, G. Manfroni, V. Cecchetti, G. Palù, L. Goracci, A. Loregian, O. Tabarrini, Eur. J. Med. Chem. 138, 128 (2017)
P. Seck, D. Thomae, G. Kirsch, J. Heterocycl. Chem. 45, 853 (2008)
K. Gewald, E. Schinke, H. Böttcher, Chem. Ber. 99, 94 (1966)
E. Rezaei-Seresht, R. Tayebee, M. Yasemi, Synth. Commun. 43, 1859 (2013)
F. Javadi, R. Tayebee, Microporous Mesoporous Mater. 231, 100 (2016)
B. Mahdavi, W.A. Yaacob, L.B. Din, I. Nazlina, Sains Malays. 41, 1233 (2012)
M. Baghayeri, B. Mahdavi, Z. Hosseinpor-Mohsen Abadi, S. Farhadi, Appl. Organomet. Chem. 32, 4057 (2018)
R. Tayebee, S.J. Ahmadi, E. Rezaei-Seresht, F. Javadi, M.A. Yasemi, M. Hosseinpour, B. Maleki, Ind. Eng. Chem. Res. 51, 14577 (2012)
K. Bozorov, L.F. Nie, J. Zhao, H.A. Aisa, Eur. J. Med. Chem. 140, 465 (2017)
H. Behbehani, H.M. Ibrahim, S. Makhseed, M.H. Elnagdi, H. Mahmoud, Eur. J. Med. Chem. 52, 51 (2012)
E.R. Wu, C. Decker, N. Blok, H. Bui, T.J. You, J. Wang, A.R. Bourgoyne, V. Knowles, K.L. Berens, G.W. Holland, T.A. Brock, J. Med. Chem. 47, 1969 (2004)
M.S. Tsang, D. Jiao, B.C. Chan, K.L. Hon, P.C. Leung, C. Lau, E.C. Wong, L. Cheng, C.K. Chan, C.W. Lam, C.K. Wong, Molecules 21, 519 (2016)
G. Oboh, O.B. Ogunsuyi, M.D. Ogunbadejo, S.A. Adefegha, J. Food Drug Anal. 24, 627 (2016)
G.H. Chen, Y.L. Lin, W.L. Hsu, S.K. Hsieh, J.T. Tzen, J. Food Drug Anal. 23, 116 (2015)
M. Arabi, M. Ghaedi, A. Ostovan, J. Chromatogr. B 1048, 102 (2017)
A.C.V. of Aguiar, R.O. of Moura, J.F.B.M. Junior, H.A. de Oliveira Rocha, R.B.G. Câmara, M.D.S.C. Schiavon, Biomed. Pharmacother. 84, 403 (2016)
A. Subramanian, A. John, M. Vellayappan, A. Balaji, S. Jaganathan, E. Supriyanto, M. Yusof, RSC Adv. 5, 35608 (2015)
B.R. You, J.H. Moon, H.Y. Han, W.H. Park, Food Chem. Toxicol. 48, 1334 (2010)
Acknowledgements
We are grateful to Hakim Sabzevari University and Sabzevar University of Medical Sciences for the support of the present study.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Mahdavi, B., Hosseyni-Tabar, S.M., Rezaei-Seresht, E. et al. Synthesis and biological evaluation of novel hybrid compounds derived from gallic acid and the 2-aminothiophene derivatives. J IRAN CHEM SOC 17, 809–815 (2020). https://doi.org/10.1007/s13738-019-01813-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13738-019-01813-0