Abstract
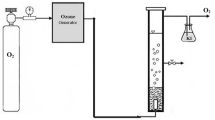
In the present work both FTIR and Flame AAS were jointly used to investigate the uptake of pollutants by the aquatic plant water hyacinth. The plant was immersed alive in synthetic standard solutions containing acetic acid up to 0.40 M for 19.0 h. Then it was immersed in 0.10 M acetic acid for different periods of time up to 210.0 h. Later on, both the acetic acid treated beside non treated ones was immersed separately in 1.0, 3.0 and 5.0 mg l-1 concentrates derived from Cd and Pb up to 240.0 h. The obtained results demonstrate the capability of the plant to remove both organic and inorganic pollutants. It is recommended that the plant be used to remediate Pb and Cd as well as other organic pollutants containing COOH. The results also indicate that the selectivity for Pb is higher than that of Cd. Additionally, pentahydrated divalent metal acetate model is presented to describe the coordination of the divalent metal ion with the acetic acid.
Similar content being viewed by others
References
N. Chand, S.K. Joshi, J. Mat. Sci. Lett. 31 (1994) 156.
K.H. Batanouny, A.M. El-Fiky, Aquatic Botany 1 (1975) 243.
B.C. Wolverton, NASA Technical Memorandum TMX- 7271 Washington, 1975.
C. Wolverton, R.C. Macdonald NASA, Technical Memorandum, TM-X-72731, Washington, 1976.
S. Murmato, Y. Oki Bull. Environ. Contam Toxicology 30 (1983) 170
Y.L. Zhu, A.M. Zayed, J.H. Qian, T.M. de Souza, M. Terry, J. Environ. Qual. 28 (2003) 339.
A.J. England, Municipal Waste Reuse News 33 (1980) 14.
S. Murmato, Y. Oki, Bull. Environ. Contam. Toxicology 33 (1984) 444.
D.C. Mortimar, Environ. Monit Assess. 5 (1985) 311.
M. Ibrahim, MSc thesis, Cairo University, Egypt, 1996.
M. Ibrahim, PhD Thesis, Cairo University, Egypt, 2000.
M.A. Ahmed, G.S. El-Bahy, M. Ibrahim, Bull. NRC Egypt 29 (2004) 523.
M.E. Soltan, M.N. Rashed, Adv. Environ. Res. 7 (2003) 321.
E.A. Ghabbour, G. Davies, Y.Y. Lam, M.E. Vozzella, Environ. Poll. 131 (2004) 445.
H. Xia, X. Ma, Bioresource Techn. 97 (2006) 1050.
M.A. Maine, N. Suñe, H. Hadad, G. Sánchez, C. Bonetto, Ecolog. Engin. 26 (2006) 341.
K. Skinner, N. Wright, E. Porter-Goff, Environ. Pollu. 145 (2007) 234.
E. Mathias, M.W.H. Evangelou, S. Andreas, Chemosphere 68 (2007) 989.
H.E. Piepmeier, L. de Galan, Spectrochim. Acta 31 (1976) 163.
M. Ibrahim, T. Scheytt, 2nd International Congress on Environmental Planning and Management, 5-10 Aug., 2007 TU-Berlin, Berlin, Germany, 2007, 231.
P. Scharmel, Anal. Chim. Acta 72 (1974) 414.
R.E. Sturgeon, C.L. Chakrabarti, I.S. Maines, P. C. Bertels, Anal. Chem. 47 (1975) 1240.
A.A. Shaltout, M. Ibrahim, Cand. J. Anal. Sci. and Spectro. 52 (2007) 280.
D. Beaty, Concepts, Instrumentation and Techniques in Atomic Absorption Spectrometry, Perkin-Elmer Corporation, 29–31, USA, 1978.
R.R. Röle, Anal. Chem. 58 (1986) 2891.
M. Ibrahim, E. Koglin, Acta Chim. Slov. 51 (2004) 453.
M. Ibrahim, H. El-Haes, Int. J. Environ. Poll. 23 (2005) 417.
A.D. Becke, J. Chem. Phys. 98 (1993) 5648.
C. Lee, W. Yang, R.G. Parr, Phys. Rev. B37 (1988) 785.
B. Miehlich, A. Savin, H. Stoll, H. Preuss, Chem. Phys. Lett. 157 (1989) 200.
B.C. Smith, Infrared Spectral Interpretation, A Systematic Approach, CRC Press LLC, 1999.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Ibrahim, M., Shaltout, A.A., Atta, D.E. et al. Removal of COOH, Cd and Pb using water hyacinth: FTIR and flame atomic absorption study. JICS 6, 364–372 (2009). https://doi.org/10.1007/BF03245846
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF03245846