Abstract
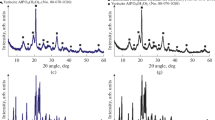
The effect of the temperature and exposure time of the starting aluminophosphate gels on their chemical and phase composition was studied. When the source of aluminum is its hydrated oxide (boehmite), gels with different contents of ammonium phosphate, undissolved boehmite, and amorphous aluminophosphate can be formed. The formation of a gel with dominant amorphous aluminophosphate allows selective crystallization of AlPO4-11 of high phase purity and ~100% crystallinity. The developed procedure for the preparation of highly dispersed aluminophosphate AlPO4-11 molecular sieves with these properties allows a transition to crystallization of silicoaluminophosphates SAPO-11—promising domestic catalysts for industrial hydroisomerization of n-paraffins in the production of Arctic diesel fuels and group III oils and isomerization of butenes.



Similar content being viewed by others
REFERENCES
Cejka, J., Corma, A., and Zones, S., Zeolites and Catalysis: Synthesis, Reactions, and Applications, Weinheim: Wiley-VCH, 2010.
Degnan, T.F., Stud. Surf. Sci. Catal., 2007, vol. 170, pp. 54–65.
Moliner, M., Martínez, C., and Corma, A., Angew. Chem., Int. Ed. Engl., 2015, vol. 54, no. 12, pp. 3560–3579.
Web of Science. https://apps.webofknowledge.com. Cited November 25, 2017.
US Patent 4440871, 1984.
Flanigen, E.M., Patton, R.L., and Wilson, S.T., Stud. Surf. Sci. Catal., 1988, vol. 37, pp. 13–37.
Pastore, H.O., Coluccia, S., and Marchese, L., Annu. Rev. Mater. Res., 2005, vol. 35, pp. 351–395.
Tian, P., Wei, Y., Ye., M., and Liu, Zh., ACS Catal., 2015, vol. 5, no. 3, pp. 1922–1938.
Bértolo, R., Silva, J.M., Riberio, F., Maldonado-Hódar, F.J., Fernandes, F., and Martins, A., Appl. Catal., A, 2014, vol. 485, pp. 230–237.
Baerlocher, C., McCusker, L.B., and Olson, D.H., Atlas of Zeolite Framework Types, Amsterdam: Elsevier, 2007.
Deldari, H., Appl. Catal., A, 2005, vol. 293, pp. 1–10.
Wang, X., Gou, F., Wei, X., Liu, Z., Wei, Z., Guo, S., and Zhao, L., Korean J. Chem. Eng., 2016, vol. 33, no. 7, pp. 2034–2041.
Zhu, Z., Chen, Q., Xie, Z., Yang, W., and Li, C., Microporous Mesoporous Mater., 2006, vol. 88, nos. 1–3, pp. 16–21.
Yang, S.-M., Lin, J.-Y., Guo, D.-H., and Liaw, S.-G., Appl. Catal., A, 1999, vol. 181, no. 1, pp. 113–122.
Yang, Z, Li, J., Liu, Y., and Liu, C., J. Energy Chem., 2017, vol. 26, no. 4, pp. 688–694.
Chen, Z., Dong, Y., Jiang, S., Song, W., Lai, W., Yi, K., and Fang, W., J. Mater. Sci., 2017, vol. 52, no. 8, pp. 4460–4471.
Huo, Q. and Xu, R., J. Chem. Soc., Chem. Commun., 1990, no. 10, pp. 783–784.
Ojo, A.F. and McCusker, L.B., Zeolites, 1991, vol. 11, no. 5, pp. 460–465.
Park, M. and Komarneni, S., Microporous Mesoporous Mater., 1998, vol. 20, nos. 1–3, pp. 39–44.
Bandyopadhyay, M., Bandyopadhyay, R., Kubota, Y., and Sugi, Y., Chem. Lett., 2000, vol. 29, no. 9, pp. 1024–1025.
Tapp, N.J., Milestone, N.B., and Bibby, D.M., Appl. Spectrosc., 1988, vol. 8, no. 3, pp. 183–188.
Kel’tsev, N.V., Osnovy adsorbtsionnoi tekhniki (Fundamentals of Adsorption Engineering), Moscow: Khimiya, 1984.
Gregg, S.J., Sing, K.S.W., and Salzberg, H.W., J. Electrochem. Soc., 1967, vol. 114, no. 11, p. 279C.
Alfonzo, M., Goldwasser, J., López, C.M., Machado, F.J., Matjushin, M., Méndez, B., and Ramírez de Agudelo, M.M., J. Mol. Catal. A: Chem., 1995, vol. 98, no. 1, pp. 35–48.
Huang, Y., Richer, R. and Kirby, C.W., J. Phys. Chem. B, 2003, vol. 107, no. 6, pp. 1326–1337.
Balakrishnan, I. and Prasad, S., Appl. Catal., 1990, vol. 62, no. 1 pp. L7–L11.
Zhang, B., Xu, J., Fan, F., Gou, Q., Tong, X., Yan, W., Yu, J., Deng, F., Li, C., and Xu, R., Microporous Mesoporous Mater., 2012, vol, 147, no. 1, pp. 212–221.
Tong, X., Xu, J., Xin, L., Huang, P., Lu, H., Wang, C., Yan, W., Yu, J., Deng, F., Sun, H., and Xu, R., Microporous Mesoporous Mater., 2012, vol. 164, pp. 56–66.
Cheng, T., Xu, J., Li, X., Li, Y., Zhang, B., Yan, W., Yu, J., Sun, H., Deng, F., and Xu, R., Microporous Mesoporous Mater., 2012, vol. 152, pp. 190–207.
ACKNOWLEDGMENTS
The reported study was funded by RFBR according to the research project no. 18-33-00077 mol_a.
The obtained materials were analyzed using the equipment of the Agidel Multiaccess Center at the Institute of Petrochemistry and Catalysis, Russian Academy of Sciences.
Author information
Authors and Affiliations
Corresponding author
Additional information
Translated by L. Smolina
Rights and permissions
About this article
Cite this article
Agliullin, M.R., Khairullina, Z.R., Faizullin, A.V. et al. Selective Crystallization of Aluminophosphate Molecular Sieves with an AEL Structure. Catal. Ind. 11, 1–6 (2019). https://doi.org/10.1134/S2070050419010021
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S2070050419010021