Abstract
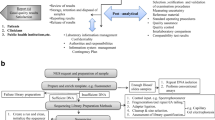
The Reference Health Laboratory (RHL) of Iran is the national authority responsible for making policies and plans for providing quality laboratory services throughout the country. RHL composed a national laboratory standard in 2007 as the minimum mandatory quality requirements for clinical laboratories. The reference of that standard was ISO 15189:2007, with considerable modifications. It also contained many technical details from other credible references to become applicable for Iranian laboratories. Shortly afterward, complying with the national standard became a condition for renewing laboratory licenses. The success rate of license renewal in 2016 revealed that the majority of laboratories had implemented the main requirements of the standard. Therefore, we decided to upgrade the national standard and make it more compatible with ISO 15189:2012. Nevertheless, we tried to keep it comprehensible and user friendly, and adjusted it to the national regulations. To ensure that the new edition matches the local needs, all laboratory stakeholders were involved in revising the related sections. The second edition of the national laboratory standard is used as a scale for competency assessment. It follows the structure and format of the ISO 15189:2012. The content is also similar, although some explanations, examples and regulatory requirements are added to clarify or adapt it to the local condition. Here, we reviewed the process of upgrading the standard and discussed why and how we elaborated on certain sections of the international standard to make it easily understandable and facilitate its application while preserving the format and structure of the ISO 15189.
Similar content being viewed by others
References
WHO (2007) International health regulations (2005): areas of work for implementation. WHO/CDS/EPR/IHR:5–6. http://www.who.int/iris/handle/10665/69770
Safadel N, Anjarani S et al (2014) Establishing an Iranian medical laboratory standard based on ISO 15189. Accred Qual Assur 19:473–476
Dahim P, Amini R et al (2009) Implementation of quality management system in Iranian medical laboratories. Iran J Pub Health 38(Supp 1):150–152
Safadel N, Dahim P et al (2013) Challenges of implementing Iranian national laboratory standards. Iran J Pub Health 42(Supp 1):125–128
Datema Tjeerd AM, Linda Oskam et al (2012) Review and comparison of quality standards, guidelines and regulations for laboratories. Afr J Lab Med 1(1):3
United Nations (2000) General assembly United Nations millennium declaration, NewYork City. http://www.un.org/millennium/declaration/ares552e.pdf. Accessed 8 Sep 2000
World Health Organization (2008) Regional Office for Africa. The Maputo declaration on strengthening of laboratory systems Jan 24, Maputo, Mozambique
United Nations (2010) Millennium development goals Genev. http://www.un.org/millenniumgoals. Accessed 11 Nov 2011
WHO (2011) WHO guide for the stepwise laboratory improvement process towards accreditation in the African Region. WHO Regional office of Africa, Brazzaville Congo
WHO (2011) Laboratory quality management system hand book. World Health Organization, Geneva
Foundation to promote the quality of the laboratory and the accreditation of laboratories in health care (CCKL) (2005) Fourth practical guideline for a quality system for a laboratory in the health care system, 4th edn. Dutch, Bilthoven
Yang Zh (2009) The laboratory accreditation and regulations of clinical laboratory in China. Clin Biochem 42(4–5):310
Mazziotta D (2009) Accreditation of clinical laboratories in the Latin-American region. Clin Biochem 42(4–5):309
Sierra-Amor RI, López-Martinez M (2007) Medical laboratory accreditation according to ISO 15189:2003. The Mexican experience. Biochem Med 17(188–192):2
Sierra-Amor RI (2009) Mexican experience on laboratory accreditation according to ISO 15189:2003. Clin Biochem 42(4–5):318
Wattanasri N, Manoroma W (2010) Laboratory accreditation in Thailand: a systemic approach. Am J Clin Pathol 134(534–540):4
Kusum M, Silva P (2005) Quality standards in health laboratories implementation in Thailand: a novel approach. WHO Regional Office for South-East Asia, New Delhi
Kieviet W, Frank E (2009) Essentials of Clinical Laboratory Management in Developing Regions-IFCC Series
Gyani GJ, Krishnamurthy B (2014) The national accreditation board for hospital and health care providers accreditation programme in India. World Hosp Health Serv 50(9–12):1
Handoo A, Sood SK (2012) Clinical laboratory accreditation in India. Clin Lab Med 32(281–292):2
WHO (2004) Laboratory biosafety manual, 3rd edn. World Health Organization, Geneva
CDC (2009) Biosafety in microbiological and biomedical laboratories, 5th edn. HHS Publication No. (CDC) 21-1112, New York
WHO (2015–2016) Guidance on regulations for the transport of infectious substances. World Health Organization
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Safadel, N., Dahim, P., Anjarani, S. et al. Upgrading the Iranian national laboratory standard to conform to ISO 15189:2012. Accred Qual Assur 24, 463–470 (2019). https://doi.org/10.1007/s00769-019-01401-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00769-019-01401-y