Abstract
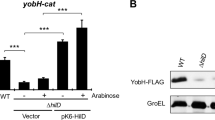
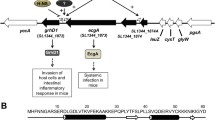
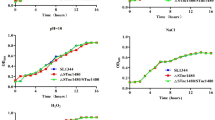
Salmonella enterica serovar Typhimurium (S. Typhimurium) is an important gram-negative intracellular pathogen that infects humans and animals. More than 50 putative regulatory proteins have been identified in the S. Typhimurium genome, but few have been clearly defined. In this study, the physiological function and regulatory role of STM14_3563, which encodes a ParD family putative transcriptional regulator in S. Typhimurium, were investigated. Macrophage replication assays and mice experiments revealed that S. Typhimurium showed reduced growth in murine macrophages and attenuated virulence in mice owing to deletion of STM14_3563 gene. RNA sequencing (RNA-Seq) data showed that STM14_3563 exerts wide-ranging effects on gene expression in S. Typhimurium. STM14_3563 activates the expression of several genes encoded in Salmonella pathogenicity island (SPI)-6, SPI-12, and SPI-13, which are required for intracellular replication of S. Typhimurium. Additionally, the global transcriptional regulator Fis was found to directly activate STM14_3563 expression by binding to the STM14_3563 promoter. These results indicate that STM14_3563 is involved in the regulation of a variety of virulence-related genes in S. Typhimurium that contribute to its growth in macrophages and virulence in mice.




Similar content being viewed by others
References
Blanc-Potard AB (1997) The Salmonella selC locus contains a pathogenicity island mediating intramacrophage survival. EMBO J 16:5376–5385. https://doi.org/10.1093/emboj/16.17.5376
Bustamante VH, Martinez LC, Santana FJ, Knodler LA, Steele-Mortimer O, Puente JL (2008) HilD-mediated transcriptional cross-talk between SPI-1 and SPI-2. Proc Natl Acad Sci U S A 105:14591–14596. https://doi.org/10.1073/pnas.0801205105
Chakraborty S, Chaudhuri D, Balakrishnan A, Chakravortty D (2014) Salmonella methylglyoxal detoxification by STM3117-encoded lactoylglutathione lyase affects virulence in coordination with Salmonella pathogenicity island 2 and phagosomal acidification. Microbiology+ 160:1999–2017. https://doi.org/10.1099/mic.0.078998-0
Cho BK, Knight EM, Barrett CL, Palsson BO (2008) Genome-wide analysis of Fis binding in Escherichia coli indicates a causative role for A-/AT-tracts. Genome Res 18:900–910. https://doi.org/10.1101/gr.070276.107
Coburn B, Grassl GA, Finlay BB (2007) Salmonella, the host and disease: a brief review. Immunol Cell Biol 85:112–118. https://doi.org/10.1038/sj.icb.7100007
Coombes BK, Brown NF, Valdez Y, Brumell JH, Finlay BB (2004) Expression and secretion of Salmonella pathogenicity island-2 virulence genes in response to acidification exhibit differential requirements of a functional type III secretion apparatus and SsaL. J Biol Chem 279:49804–49815. https://doi.org/10.1074/jbc.M404299200
Datsenko KA, Wanner BL (2000) One-step inactivation of chromosomal genes in Escherichia coli K-12 using PCR products. Proc Natl Acad Sci U S A 97:6640–6645. https://doi.org/10.1073/pnas.120163297
De la Cruz MA, Zhao W, Farenc C, Gimenez G, Raoult D, Cambillau C, Gorvel JP, Meresse S (2013) A toxin-antitoxin module of Salmonella promotes virulence in mice. PLoS Pathog 9:e1003827. https://doi.org/10.1371/journal.ppat.1003827
Deininger P (1990) Molecular cloning: a laboratory manual: 2nd ed. Edited by J. Sambrook, E. F. Fritsch, and T. Maniatis. Cold Spring Harbor Laboratory Press, Cold Spring Harbor, NY, 1989 (in 3 volumes). 186:182–183. doi:https://doi.org/10.1016/0003-2697(90)90595-Z
Eriksson S, Lucchini S, Thompson A, Rhen M, Hinton JC (2003) Unravelling the biology of macrophage infection by gene expression profiling of intracellular Salmonella enterica. Mol Microbiol 47:103–118. https://doi.org/10.1046/j.1365-2958.2003.03313.x
Fabrega A, Vila J (2013) Salmonella enterica serovar Typhimurium skills to succeed in the host: virulence and regulation. Clin Microbiol Rev 26:308–341. https://doi.org/10.1128/CMR.00066-12
Folkesson A, Löfdahl S, Normark S (2002) The Salmonella enterica subspecies I specific centisome 7 genomic island encodes novel protein families present in bacteria living in close contact with eukaryotic cells. Res Microbiol 153:537–545
Goeders N, Van Melderen L (2014) Toxin-antitoxin systems as multilevel interaction systems. Toxins 6:304–324
Gunn JS, Alpuche-Aranda CM, Loomis WP, Belden WJ, Miller SI (1995) Characterization of the Salmonella typhimurium pagC/pagD chromosomal region. J Bacteriol 177:5040–5047. https://doi.org/10.1128/jb.177.17.5040-5047.1995
Guo P, Wu CM (2017) Gut microbiota brings a novel way to illuminate mechanisms of natural products in vivo. Chin Herbal Med (CHM) 9(4):301–306
Haneda T, Ishii Y, Danbara H, Okada N (2009) Genome-wide identification of novel genomic islands that contribute to Salmonella virulence in mouse systemic infection. FEMS Microbiol Lett 297:241–249. https://doi.org/10.1111/j.1574-6968.2009.01686.x
Hansen-Wester I, Hensel M (2002) Genome-based identification of chromosomal regions specific for Salmonella spp. Infect Immun 70:2351–2360. https://doi.org/10.1128/iai.70.5.2351-2360.2002
Hautefort I, Thompson A, Eriksson-Ygberg S, Parker ML, Lucchini S, Danino V, Bongaerts RJ, Ahmad N, Rhen M, Hinton JC (2008) During infection of epithelial cells Salmonella enterica serovar Typhimurium undergoes a time-dependent transcriptional adaptation that results in simultaneous expression of three type 3 secretion systems. Cell Microbiol 10:958–984
Hensel M (2004) Evolution of pathogenicity islands of Salmonella enterica. Int J Med Microbiol 294:95–102. https://doi.org/10.1016/j.ijmm.2004.06.025
Jiang L, Feng L, Yang B, Zhang W, Wang P, Jiang X, Wang L (2017) Signal transduction pathway mediated by the novel regulator LoiA for low oxygen tension induced Salmonella Typhimurium invasion. PLoS Pathog 13:e1006429. https://doi.org/10.1371/journal.ppat.1006429
Jiang X, Li X, Sun S, Jiang L (2018) The transcriptional regulator VarN contributes to Salmonella Typhimurium growth in macrophages and virulence in mice. Res Microbiol 169:214–221. https://doi.org/10.1016/j.resmic.2018.03.003
Kelly A (2004) A global role for Fis in the transcriptional control of metabolism and type III secretion in Salmonella enterica serovar Typhimurium. Microbiology+ 150:2037–2053. https://doi.org/10.1099/mic.0.27209-0
Langmead B, Salzberg SL (2012) Fast gapped-read alignment with Bowtie 2. Nat Methods 9:357–359. https://doi.org/10.1038/nmeth.1923
LaRock DL, Chaudhary A, Miller SI (2015) Salmonellae interactions with host processes. Nat Rev Microbiol 13:191–205. https://doi.org/10.1038/nrmicro3420
Li H, Li X, Lv R, Jiang X, Cao H, Du Y, Jiang L, Liu B (2018) Global regulatory function of the low oxygen-induced transcriptional regulator LoiA in Salmonella Typhimurium revealed by RNA sequencing. Biochem Bioph Res Co 503(3):2022–2027
Liu Y, Liu Q, Qi L, Ding T, Wang Z, Fu J, Hu M, Li M, Song J, Liu X (2017) Temporal regulation of a Salmonella Typhimurium virulence factor by the transcriptional regulator YdcR. Mol Cell Proteomics 16:1683–1693. https://doi.org/10.1074/mcp.M117.068296
Martínez LC, Yakhnin H, Camacho MI, Georgellis D, Babitzke P, Puente JL, Bustamante VH (2011) Integration of a complex regulatory cascade involving the SirA/BarA and Csr global regulatory systems that controls expression of the Salmonella SPI-1 and SPI-2 virulence regulons through HilD. Mol Microbiol 80:1637–1656. https://doi.org/10.1111/j.1365-2958.2011.07674.x
Mastroeni P, Grant A, Restif O, Maskell D (2009) A dynamic view of the spread and intracellular distribution of Salmonella enterica. Nat Rev Microbiol 7:73–80. https://doi.org/10.1038/nrmicro2034
Mulder DT, Cooper CA, Coombes BK (2012) Type VI secretion system-associated gene clusters contribute to pathogenesis of Salmonella enterica serovar Typhimurium. Infect Immun 80:1996–2007. https://doi.org/10.1128/IAI.06205-11
O’Cróinín T, Carroll RK, Kelly A, Dorman CJ (2006) Roles for DNA supercoiling and the Fis protein in modulating expression of virulence genes during intracellular growth of Salmonella enterica serovar Typhimurium. Mol Microbiol 62:869–882. https://doi.org/10.1111/j.1365-2958.2006.05416.x
Pezoa D, Yang H, Blondel CJ, Santiviago CA, Andrews-Polymenis HL, Contreras I (2013) The type VI secretion system encoded in SPI-6 plays a role in gastrointestinal colonization and systemic spread of Salmonella enterica serovar Typhimurium in the chicken. PLoS One 8:e63917. https://doi.org/10.1371/journal.pone.0063917
Pontes MH, Groisman EA (2019) Slow growth determines nonheritable antibiotic resistance in Salmonella enterica. Sci Signal 12. https://doi.org/10.1126/SciSignal.aax3938
Ramage HR, Connolly LE, Cox JS (2009) Comprehensive functional analysis of Mycobacterium tuberculosis toxin-antitoxin systems: implications for pathogenesis, stress responses, and evolution. PLoS Genet 5:e1000767. https://doi.org/10.1371/journal.pgen.1000767
Schlumberger MC, Hardt W (2006) Salmonella type III secretion effectors: pulling the host cell’s strings. Curr Opin Microbiol 9:46–54. https://doi.org/10.1016/j.mib.2005.12.006
Shah DH, Lee MJ, Park JH, Lee JH, Eo SK, Kwon JT, Chae JS (2005) Identification of Salmonella gallinarum virulence genes in a chicken infection model using PCR-based signature-tagged mutagenesis. Microbiology+ 151:3957–3968. https://doi.org/10.1099/mic.0.28126-0
Shi L, Adkins JN, Coleman JR, Schepmoes AA, Dohnkova A, Mottaz HM, Norbeck AD, Purvine SO, Manes NP, Smallwood HS, Wang H, Forbes J, Gros P, Uzzau S, Rodland KD, Heffron F, Smith RD, Squier TC (2006) Proteomic analysis of Salmonella enterica serovar Typhimurium isolated from RAW 264.7 macrophages. J Biol Chem 281:29131–29140. https://doi.org/10.1074/jbc.M604640200
Slauch JM, Lee AA, Mahan MJ, Mekalanos JJ (1996) Molecular characterization of the oafA locus responsible for acetylation of Salmonella typhimurium O-antigen: OafA is a member of a family of integral membrane trans-acylases. J Bacteriol 178:5904–5909. https://doi.org/10.1128/jb.178.20.5904-5909
Soo VW, Wood TK (2013) Antitoxin MqsA represses curli formation through the master biofilm regulator CsgD. Sci Rep 3:3186–3187. https://doi.org/10.1038/srep03186
Srikumar S, Kröger C, Hébrard M, Colgan A, Owen SV, Sivasankaran SK, Cameron ADS, Hokamp K, Hinton JCD (2015) RNA-seq brings new insights to the intra-macrophage transcriptome of Salmonella Typhimurium. PLoS Pathog 11:e1005262. https://doi.org/10.1371/journal.ppat.1005262
Stapels DAC, Hill PWS, Westermann AJ, Fisher RA, Thurston TL, Saliba AE, Blommestein I, Vogel J, Helaine S (2018) Salmonella persisters undermine host immune defenses during antibiotic treatment. Science 362:1156–1160. https://doi.org/10.1126/science.aat7148
Valdivia RH, Falkow S (1997) Fluorescence-based isolation of bacterial genes expressed within host cells. Science 277:2007–2011. https://doi.org/10.1126/science.277.5334.2007
Vazquez-Torres A, Jones-Carson J, Mastroeni P, Ischiropoulos H, Fang FC (2000) Antimicrobial actions of the NADPH phagocyte oxidase and inducible nitric oxide synthase in experimental salmonellosis. I. Effects on microbial killing by activated peritoneal macrophages in vitro. J Exp Med 192:227–236
Wang X, Kim Y, Hong SH, Ma Q, Brown BL, Pu M, Tarone AM, Benedik MJ, Peti W, Page R, Wood TK (2011) Antitoxin MqsA helps mediate the bacterial general stress response. Nat Chem Biol 7:359–366. https://doi.org/10.1038/nchembio.560
Wang H, Liu B, Wang Q, Wang L (2013) Genome-wide analysis of the Salmonella Fis regulon and its regulatory mechanism on pathogenicity islands. PLoS One 8:e64688. https://doi.org/10.1371/journal.pone.0064688
Funding
This study was supported by a grant from the National Natural Science Foundation of China (number 31800126).
Author information
Authors and Affiliations
Contributions
LJ conceived and designed the study. XS and HZ performed most of the experiments and wrote the manuscript. XL, JY, PW, and RL participated in the animal experiments and EMSAs. BY and DH prepared the reagents, materials, and analysis tools. LJ revised the manuscript. XS and HZ contributed equally to this work.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethics statement
All animal procedures were performed with the approval of the Institutional Animal Care Committee at Nankai University (Tianjin, China). Every effort was made to minimize animal suffering.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
ESM 1
(DOCX 676 kb)
Rights and permissions
About this article
Cite this article
Song, X., Zhang, H., Liu, X. et al. The putative transcriptional regulator STM14_3563 facilitates Salmonella Typhimurium pathogenicity by activating virulence-related genes. Int Microbiol 23, 381–390 (2020). https://doi.org/10.1007/s10123-019-00110-3
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10123-019-00110-3