Abstract
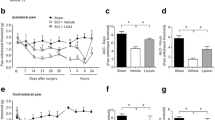
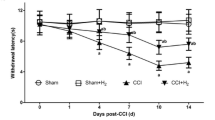
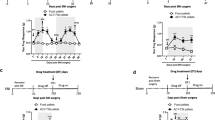
Spinal cord injury (SCI) is a deliberating disorder with impairments in locomotor deficits and incapacitating sensory abnormalities. Harpagophytum procumbens (Hp) is a botanical widely used for treating inflammation and pain related to various inflammatory and musculoskeletal conditions. Using a modified rodent contusion model of SCI, we explored the effects of this botanical on locomotor function and responses to mechanical stimuli, and examined possible neurochemical changes associated with SCI-induced allodynia. Following spinal cord contusion at T10 level, Hp (300 mg/kg, p.o.) or vehicle (water) was administered daily starting 24 h post-surgery, and behavioral measurements made every-other day until sacrifice (Day 21). Hp treatment markedly ameliorated the contusion-induced decrease in locomotor function and increased sensitivity to mechanical stimuli. Determination of Iba1 expression in spinal cord tissues indicated microglial infiltration starting 3 days post-injury. SCI results in increased levels of 4-hydroxynonenal, an oxidative stress product and proalgesic, which was diminished at 7 days by treatment with Hp. SCI also enhanced antioxidant heme oxygenase-1 (HO-1) expression. Concurrent studies of cultured murine BV-2 microglial cells revealed that Hp suppressed oxidative/nitrosative stress and inflammatory responses, including production of nitric oxide and reactive oxygen species, phosphorylation of cytosolic phospholipases A2, and upregulation of the antioxidative stress pathway involving the nuclear factor erythroid 2-related factor 2 and HO-1. These results support the use of Hp for management of allodynia by providing resilience against the neuroinflammation and pain associated with SCI and other neuropathological conditions.










Similar content being viewed by others
Abbreviations
- 4-HHE:
-
4-Hydroxyhexenal
- 4-HNE:
-
4-Hydroxynonenal
- ARA:
-
Arachidonic acid
- CHD:
-
1,3-Cyclohexanedione
- COX-1/2:
-
Cyclooxygenase-1/2
- cPLA2 :
-
Cytosolic phospholipases A2
- DHA:
-
Docosahexaenoic acid
- DCF:
-
ROS detection reagent CM-H2DCFDA
- DMEM:
-
Dulbecco’s modified Eagle’s medium
- ESI:
-
Electrospray ionization
- FBS:
-
Fetal bovine albumin
- HO-1:
-
Heme oxygenase-1
- Hp:
-
Harpagophytum procumbens, or Devil’s Claw
- HPLC:
-
Ammonium acetate
- Ibu:
-
Ibuprofen
- iNOS:
-
Nitric oxide synthase
- LPS:
-
Lipopolysaccharide
- Nrf2:
-
Erythroid 2-related factor 2
- NQO1:
-
NAD(P)H quinone dehydrogenase 1
- ROS:
-
Reactive oxygen species
- SCI:
-
Spinal cord injury
- SPE:
-
Solid phase extraction
- TBS-T:
-
Tween 20
- TOF–MS/MS:
-
Time-of-flight tandem mass spectrometry
- UHPLC:
-
Ultra-high performance liquid chromatography
References
Abdelouahab, N., & Heard, C. (2008). Effect of the major glycosides of Harpagophytum procumbens (Devil’s Claw) on epidermal cyclooxygenase-2 (COX-2) in vitro. Journal of Natural Products,71(5), 746–749.
Ajit, D., Simonyi, A., Li, R., Chen, Z., Hannink, M., Fritsche, K. L., et al. (2016). Phytochemicals and botanical extracts regulate NF-kappaB and Nrf2/ARE reporter activities in DI TNC1 astrocytes. Neurochemistry International,97, 49–56. https://doi.org/10.1016/j.neuint.2016.05.004.
Anthonymuthu, T. S., Kenny, E. M., & Bayir, H. (2016). Therapies targeting lipid peroxidation in traumatic brain injury. Brain Research,1640(Pt A), 57–76. https://doi.org/10.1016/j.brainres.2016.02.006.
Baastrup, C., & Finnerup, N. B. (2012). Pain in spinal cord injury. Pain Management,2(1), 87–94. https://doi.org/10.2217/pmt.11.70.
Basso, D. M., Beattie, M. S., & Bresnahan, J. C. (2002). Descending systems contributing to locomotor recovery after mild or moderate spinal cord injury in rats: Experimental evidence and a review of literature. Restorative Neurology and Neuroscience,20(5), 189–218.
Chuang, D. Y., Chan, M. H., Zong, Y., Sheng, W., He, Y., Jiang, J. H., et al. (2013). Magnolia polyphenols attenuate oxidative and inflammatory responses in neurons and microglial cells. Journal of Neuroinflammation,10, 15. https://doi.org/10.1186/1742-2094-10-15.
Chuang, D. Y., Cui, J., Simonyi, A., Engel, V. A., Chen, S., Fritsche, K. L., et al. (2014). Dietary Sutherlandia and elderberry mitigate cerebral ischemia-induced neuronal damage and attenuate p47phox and phospho-ERK1/2 expression in microglial cells. ASN Neuro. https://doi.org/10.1177/1759091414554946.
Chuang, D. Y., Simonyi, A., Cui, J., Lubahn, D. B., Gu, Z., & Sun, G. Y. (2016). Botanical polyphenols mitigate microglial activation and microglia-induced neurotoxicity: Role of cytosolic phospholipase A2. NeuroMolecular Medicine,18(3), 415–425. https://doi.org/10.1007/s12017-016-8419-5.
Chuang, D. Y., Simonyi, A., Kotzbauer, P. T., Gu, Z., & Sun, G. Y. (2015). Cytosolic phospholipase A2 plays a crucial role in ROS/NO signaling during microglial activation through the lipoxygenase pathway. Journal of Neuroinflammation,12, 199. https://doi.org/10.1186/s12974-015-0419-0.
Csala, M., Kardon, T., Legeza, B., Lizak, B., Mandl, J., Margittai, E., et al. (2015). On the role of 4-hydroxynonenal in health and disease. Biochimica et Biophysica Acta,1852(5), 826–838. https://doi.org/10.1016/j.bbadis.2015.01.015.
Dai, W., Wang, X., Teng, H., Li, C., Wang, B., & Wang, J. (2019). Celastrol inhibits microglial pyroptosis and attenuates inflammatory reaction in acute spinal cord injury rats. International Immunopharmacology,66, 215–223. https://doi.org/10.1016/j.intimp.2018.11.029.
D’Angelo, R., Morreale, A., Donadio, V., Boriani, S., Maraldi, N., Plazzi, G., et al. (2013). Neuropathic pain following spinal cord injury: What we know about mechanisms, assessment and management. European Review for Medical and Pharmacological Sciences,17(23), 3257–3261.
De Logu, F., Nassini, R., Materazzi, S., Carvalho Goncalves, M., Nosi, D., Rossi Degl’Innocenti, D., et al. (2017). Schwann cell TRPA1 mediates neuroinflammation that sustains macrophage-dependent neuropathic pain in mice. Nature Communications,8(1), 1887. https://doi.org/10.1038/s41467-017-01739-2.
Diaz, A. F., Polo, S., Gallardo, N., Leanez, S., & Pol, O. (2019). Analgesic and antidepressant effects of Oltipraz on neuropathic pain in mice by modulating microglial activation. Journal of Clinical Medicine,8(6), 1. https://doi.org/10.3390/jcm8060890.
Fiebich, B. L., Munoz, E., Rose, T., Weiss, G., & McGregor, G. P. (2012). Molecular targets of the antiinflammatory Harpagophytum procumbens (devil’s claw): Inhibition of TNFalpha and COX-2 gene expression by preventing activation of AP-1. Phytotherapy Research,26(6), 806–811. https://doi.org/10.1002/ptr.3636.
Finnerup, N. B. (2013). Pain in patients with spinal cord injury. Pain,154(Suppl 1), S71–S76. https://doi.org/10.1016/j.pain.2012.12.007.
Gaskell, H., Derry, S., Stannard, C., & Moore, R. A. (2016). Oxycodone for neuropathic pain in adults. Cochrane Database of Systematic Reviews,7, CD010692. https://doi.org/10.1002/14651858.cd010692.pub3.
Georgiev, M. I., Ivanovska, N., Alipieva, K., Dimitrova, P., & Verpoorte, R. (2013). Harpagoside: From Kalahari Desert to pharmacy shelf. Phytochemistry,92, 8–15.
Gosselin, R. D., Suter, M. R., Ji, R. R., & Decosterd, I. (2010). Glial cells and chronic pain. Neuroscientist,16(5), 519–531. https://doi.org/10.1177/1073858409360822.
Grace, P. M., Gaudet, A. D., Staikopoulos, V., Maier, S. F., Hutchinson, M. R., Salvemini, D., et al. (2016). Nitroxidative signaling mechanisms in pathological pain. Trends in Neurosciences,39(12), 862–879. https://doi.org/10.1016/j.tins.2016.10.003.
Hackel, D., Pflucke, D., Neumann, A., Viebahn, J., Mousa, S., Wischmeyer, E., et al. (2013). The connection of monocytes and reactive oxygen species in pain. PLoS ONE,8(5), e63564. https://doi.org/10.1371/journal.pone.0063564.
Huang, T. H., Tran, V. H., Duke, R. K., Tan, S., Chrubasik, S., Roufogalis, B. D., et al. (2006). Harpagoside suppresses lipopolysaccharide-induced iNOS and COX-2 expression through inhibition of NF-kappa B activation. Journal of Ethnopharmacology,104(1–2), 149–155. https://doi.org/10.1016/j.jep.2005.08.055.
Jensen, T. S., Baron, R., Haanpaa, M., Kalso, E., Loeser, J. D., Rice, A. S., et al. (2011). A new definition of neuropathic pain. Pain,152(10), 2204–2205. https://doi.org/10.1016/j.pain.2011.06.017.
Jensen, M. P., Kuehn, C. M., Amtmann, D., & Cardenas, D. D. (2007). Symptom burden in persons with spinal cord injury. Archives of Physical Medicine and Rehabilitation,88(5), 638–645. https://doi.org/10.1016/j.apmr.2007.02.002.
Jia, Z., Zhu, H., Li, J., Wang, X., Misra, H., & Li, Y. (2012). Oxidative stress in spinal cord injury and antioxidant-based intervention. Spinal Cord,50(4), 264–274. https://doi.org/10.1038/sc.2011.111.
Kartha, S., Weisshaar, C. L., Philips, B. H., & Winkelstein, B. A. (2018). Pre-treatment with Meloxicam prevents the spinal inflammation and oxidative stress in DRG neurons that accompany painful cervical radiculopathy. Neuroscience,388, 393–404. https://doi.org/10.1016/j.neuroscience.2018.07.054.
Kaszkin, M., Beck, K. F., Koch, E., Erdelmeier, C., Kusch, S., Pfeilschifter, J., et al. (2004). Downregulation of iNOS expression in rat mesangial cells by special extracts of Harpagophytum procumbens derives from harpagoside-dependent and independent effects. Phytomedicine,11(7–8), 585–595. https://doi.org/10.1016/j.phymed.2004.02.003.
Krishna, V., Andrews, H., Jin, X., Yu, J., Varma, A., Wen, X., et al. (2013). A contusion model of severe spinal cord injury in rats. Journal of Visualized Experiments. https://doi.org/10.3791/50111.
Kwon, B. K., Okon, E., Hillyer, J., Mann, C., Baptiste, D., Weaver, L. C., et al. (2011). A systematic review of non-invasive pharmacologic neuroprotective treatments for acute spinal cord injury. Journal of Neurotrauma,28(8), 1545–1588. https://doi.org/10.1089/neu.2009.1149.
Lee, B. B., Cripps, R. A., Fitzharris, M., & Wing, P. C. (2014). The global map for traumatic spinal cord injury epidemiology: Update 2011, global incidence rate. Spinal Cord,52(2), 110–116. https://doi.org/10.1038/sc.2012.158.
Li, W., Jiang, D., Li, Q., Yao, S., Sun, X., Yang, Y., et al. (2016). Lipopolysaccharide-induced preconditioning protects against traumatic spinal cord injury by upregulating Nrf2 expression in rats. Life Sciences,162, 14–20. https://doi.org/10.1016/j.lfs.2016.08.008.
Lim, D. W., Kim, J. G., Han, D., & Kim, Y. T. (2014). Analgesic effect of Harpagophytum procumbens on postoperative and neuropathic pain in rats. Molecules,19(1), 1060–1068. https://doi.org/10.3390/molecules19011060.
Liu, N. K., Byers, J. S., Lam, T., Lu, Q. B., Sengelaub, D. R., & Xu, X. M. (2014a). Inhibition of cPLA2 has neuroprotective effects on motoneuron and muscle atrophy following spinal cord injury. Journal of Neurotrauma. https://doi.org/10.1089/neu.2014.3690.
Liu, N. K., Deng, L. X., Zhang, Y. P., Lu, Q. B., Wang, X. F., Hu, J. G., et al. (2014b). Cytosolic phospholipase A2 protein as a novel therapeutic target for spinal cord injury (Research Support, N.I.H., Extramural Research Support, Non-U.S. Gov’t). Annals of Neurology,75(5), 644–658. https://doi.org/10.1002/ana.24134.
Lv, R., Du, L., Zhang, L., & Zhang, Z. (2019). Polydatin attenuates spinal cord injury in rats by inhibiting oxidative stress and microglia apoptosis via Nrf2/HO-1 pathway. Life Sciences,217, 119–127. https://doi.org/10.1016/j.lfs.2018.11.053.
Ma, L., Liu, H., Chen, G., Chen, M., Wang, L., Zhang, X., et al. (2018). Sulfasalazine attenuates chronic constriction injury-induced neuroinflammation and mechanical hypersensitivity in rats. Neuroscience Letters,683, 174–180. https://doi.org/10.1016/j.neulet.2018.07.042.
Mao, L., Wang, H., Wang, X., Liao, H., & Zhao, X. (2011). Transcription factor Nrf2 protects the spinal cord from inflammation produced by spinal cord injury. Journal of Surgical Research,170(1), e105–e115. https://doi.org/10.1016/j.jss.2011.05.049.
Mncwangi, N., Chen, W., Vermaak, I., Viljoen, A. M., & Gericke, N. (2012). Devil’s Claw-a review of the ethnobotany, phytochemistry and biological activity of Harpagophytum procumbens. Journal of Ethnopharmacology,143(3), 755–771. https://doi.org/10.1016/j.jep.2012.08.013.
Oehler, B., Kistner, K., Martin, C., Schiller, J., Mayer, R., Mohammadi, M., et al. (2017). Inflammatory pain control by blocking oxidized phospholipid-mediated TRP channel activation. Scientific Reports,7(1), 5447. https://doi.org/10.1038/s41598-017-05348-3.
Parenti, C., Arico, G., Chiechio, S., Di Benedetto, G., Parenti, R., & Scoto, G. M. (2015). Involvement of the heme-oxygenase pathway in the antiallodynic and antihyperalgesic activity of Harpagophytum procumbens in rats. Molecules,20(9), 16758–16769. https://doi.org/10.3390/molecules200916758.
Qu, Z., Meng, F., Zhou, H., Li, J., Wang, Q., Wei, F., et al. (2014). NitroDIGE analysis reveals inhibition of protein S-nitrosylation by epigallocatechin gallates in lipopolysaccharide-stimulated microglial cells. Journal of Neuroinflammation,11, 17. https://doi.org/10.1186/1742-2094-11-17.
Quintans, J. S., Antoniolli, A. R., Almeida, J. R., Santana-Filho, V. J., & Quintans-Junior, L. J. (2014). Natural products evaluated in neuropathic pain models—A systematic review. Basic & Clinical Pharmacology & Toxicology,114(6), 442–450. https://doi.org/10.1111/bcpt.12178.
Rahbardar, M. G., Amin, B., Mehri, S., Mirnajafi-Zadeh, S. J., & Hosseinzadeh, H. (2018). Rosmarinic acid attenuates development and existing pain in a rat model of neuropathic pain: An evidence of anti-oxidative and anti-inflammatory effects. Phytomedicine,40, 59–67. https://doi.org/10.1016/j.phymed.2018.01.001.
Rahimi-Movaghar, V., Sayyah, M. K., Akbari, H., Khorramirouz, R., Rasouli, M. R., Moradi-Lakeh, M., et al. (2013). Epidemiology of traumatic spinal cord injury in developing countries: A systematic review. Neuroepidemiology,41(2), 65–85. https://doi.org/10.1159/000350710.
Rao, P., & Knaus, E. E. (2008). Evolution of nonsteroidal anti-inflammatory drugs (NSAIDs): Cyclooxygenase (COX) inhibition and beyond. Journal of Pharmacy and Pharmaceutical Sciences,11(2), 81s–110s.
Raoof, R., Willemen, H., & Eijkelkamp, N. (2018). Divergent roles of immune cells and their mediators in pain. Rheumatology (Oxford),57(3), 429–440. https://doi.org/10.1093/rheumatology/kex308.
Riego, G., Redondo, A., Leanez, S., & Pol, O. (2018). Mechanism implicated in the anti-allodynic and anti-hyperalgesic effects induced by the activation of heme oxygenase 1/carbon monoxide signaling pathway in the central nervous system of mice with neuropathic pain. Biochemical Pharmacology,148, 52–63. https://doi.org/10.1016/j.bcp.2017.12.007.
Shen, S., Yu, S., Binek, J., Chalimoniuk, M., Zhang, X., Lo, S. C., et al. (2005). Distinct signaling pathways for induction of type II NOS by IFNgamma and LPS in BV-2 microglial cells. Neurochemistry International,47(4), 298–307. https://doi.org/10.1016/j.neuint.2005.03.007.
Sheng, W., Zong, Y., Mohammad, A., Ajit, D., Cui, J., Han, D., et al. (2011). Pro-inflammatory cytokines and lipopolysaccharide induce changes in cell morphology, and upregulation of ERK1/2, iNOS and sPLA(2)-IIA expression in astrocytes and microglia. Journal of Neuroinflammation,8, 121. https://doi.org/10.1186/1742-2094-8-121.
Song, H., Lu, Y., Qu, Z., Mossine, V. V., Martin, M. B., Hou, J., et al. (2016). Effects of aged garlic extract and FruArg on gene expression and signaling pathways in lipopolysaccharide-activated microglial cells. Scientific Reports,6, 35323. https://doi.org/10.1038/srep35323.
Sun, G. Y., Chen, Z., Jasmer, K. J., Chuang, D. Y., Gu, Z., Hannink, M., et al. (2015). Quercetin attenuates inflammatory responses in BV-2 microglial cells: Role of MAPKs on the Nrf2 pathway and induction of heme oxygenase-1. PLoS ONE,10(10), e0141509. https://doi.org/10.1371/journal.pone.0141509.
Sun, G. Y., Chuang, D. Y., Zong, Y., Jiang, J., Lee, J. C., Gu, Z., et al. (2014). Role of cytosolic phospholipase A2 in oxidative and inflammatory signaling pathways in different cell types in the central nervous system. Molecular Neurobiology,50(1), 6–14. https://doi.org/10.1007/s12035-014-8662-4.
Sun, G. Y., Simonyi, A., Fritsche, K. L., Chuang, D. Y., Hannink, M., Gu, Z., et al. (2018). Docosahexaenoic acid (DHA): An essential nutrient and a nutraceutical for brain health and diseases. Prostaglandins Leukotrienes and Essential Fatty Acids,136, 3–13. https://doi.org/10.1016/j.plefa.2017.03.006.
Tator, C. H., & Fehlings, M. G. (1991). Review of the secondary injury theory of acute spinal cord trauma with emphasis on vascular mechanisms. Journal of Neurosurgery,75(1), 15–26. https://doi.org/10.3171/jns.1991.75.1.0015.
Visavadiya, N. P., Patel, S. P., VanRooyen, J. L., Sullivan, P. G., & Rabchevsky, A. G. (2016). Cellular and subcellular oxidative stress parameters following severe spinal cord injury. Redox Biology,8, 59–67. https://doi.org/10.1016/j.redox.2015.12.011.
Vlachojannis, J., Roufogalis, B. D., & Chrubasik, S. (2008). Systematic review on the safety of Harpagophytum preparations for osteoarthritic and low back pain. Phytotherapy Research,22(2), 149–152. https://doi.org/10.1002/ptr.2314.
Widerstrom-Noga, E. (2012). Multidimensional clinical pain phenotypes after spinal cord injury. Pain Management,2(5), 467–478. https://doi.org/10.2217/pmt.12.44.
Yadav, R. K., Singh, M., Roy, S., Ansari, M. N., Saeedan, A. S., & Kaithwas, G. (2018). Modulation of oxidative stress response by flaxseed oil: Role of lipid peroxidation and underlying mechanisms. Prostaglandins & Other Lipid Mediators,135, 21–26. https://doi.org/10.1016/j.prostaglandins.2018.02.003.
Yang, B., Fritsche, K. L., Beversdorf, D. Q., Gu, Z., Lee, J. C., Folk, W. R., et al. (2019a). Yin-Yang mechanisms regulating lipid peroxidation of docosahexaenoic acid and arachidonic acid in the central nervous system. Frontiers in Neurology,10, 642. https://doi.org/10.3389/fneur.2019.00642.
Yang, B., Li, R., Greenlief, C. M., Fritsche, K. L., Gu, Z., Cui, J., et al. (2018). Unveiling anti-oxidative and anti-inflammatory effects of docosahexaenoic acid and its lipid peroxidation product on lipopolysaccharide-stimulated BV-2 microglial cells. Journal of Neuroinflammation,15(1), 202. https://doi.org/10.1186/s12974-018-1232-3.
Yang, B., Li, R., Woo, T., Browning, J. D., Jr., Song, H., Gu, Z., et al. (2019b). Maternal dietary docosahexaenoic acid alters lipid peroxidation products and (n − 3)/(n − 6) fatty acid balance in offspring mice. Metabolites,9(3), 1. https://doi.org/10.3390/metabo9030040.
Zhang, L., Feng, L., Jia, Q., Xu, J., Wang, R., Wang, Z., et al. (2011). Effects of beta-glucosidase hydrolyzed products of harpagide and harpagoside on cyclooxygenase-2 (COX-2) in vitro. Bioorganic & Medicinal Chemistry,19(16), 4882–4886. https://doi.org/10.1016/j.bmc.2011.06.069.
Acknowledgements
We thank Dr. Brian Mooney, associate director of the Charles W. Gehrke Proteomics Center at the University of Missouri, for providing assistance with the LC–MS/MS, and Dr. Lloyd W. Sumner, Professor of Biochemistry and Director of MU Metabolomics Center for UHPLC analysis of H. procumbens extract. Thanks are due to Professor Xiaoming Xu at Department of Neurosurgery, Stark Neurosciences Research Institute, Indiana University for providing advise for Western blot analysis of p-cPLA2 and cPLA2 in spinal cord tissue.
Funding
This work was supported in part by the funding from the Missouri Spinal Cord Injury/Disease Research Program (SCIDRP), MIZZOU Advantage grant and research funds of the University of Missouri (ZG), and in part, support by the NIH/NCCIH (R21 AT009086). The contents are solely the responsibility of the authors and do not necessarily represent the official views of the sponsors.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Ungerer, G., Cui, J., Ndam, T. et al. Harpagophytum procumbens Extract Ameliorates Allodynia and Modulates Oxidative and Antioxidant Stress Pathways in a Rat Model of Spinal Cord Injury. Neuromol Med 22, 278–292 (2020). https://doi.org/10.1007/s12017-019-08585-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12017-019-08585-z