Abstract
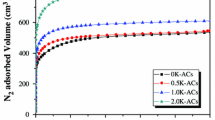
In this work, the correlation between the pore characteristics of activated carbon (AC) and the adsorption/desorption characteristics of evaporated fuel was studied. AC was prepared by various physical re-activation methods using coconut-derived commercial AC. Pore characteristics of the re-activated AC were investigated using N2/77 K adsorption isotherms. The structural characteristics of the AC were observed by X-ray diffraction and Raman spectroscopy. The butane working capacity was observed according to ASTM D5228. From the results, the specific surface area and total pore volume of the ACs were determined to be 1380–2040 m2/g and 0.60–0.96 cm3/g, respectively. It was also observed that various pore size distributions were found to be dependent on the functions of the activation method and time. A close relationship between butane activity/retentivity and micropore/mesopore volumes was found. In addition, it was inferred that the volume fraction of micropores and sub-mesopores with diameters between 1.5 and 3.0 nm primarily controls butane activity.







Similar content being viewed by others
References
Gurjar BR, Molina LT, Ojha CSP (2010) Air pollution: health and environmental impacts. CRC Press, Boca Raton
US-EPA Technical bulletin (1999) Nitrogen oxides (NOx), why and how they are controlled. Clean Air Technology Center, U.S. Environmental Protection Agency. https://www3.epa.gov/ttncatc1/dir1/fnoxdoc.pdf. Accessed 16 Oct 2019
Burchell TD (1999) Carbon materials for advanced technologies. Pergamon, Amsterdam
Allen JL, Gatz JL, Eklund PC (1999) Applications for activated carbons from used tires: butane working capacity. Carbon 37:1485–1489. https://doi.org/10.1016/S0008-6223(99)00011-1
Yenisoy-Karakas S, Aygun A, Gunes M, Tahrasakal E (2004) Physical and chemical characteristics of polymer-based spherical activated carbon and its ability to adsorb organics. Carbon 42:477–484. https://doi.org/10.1016/j.carbon.2003.11.019
Miguel GS, Fowler GD, Sollars CJ (2003) A study of the characteristics of activated carbons produced by steam and carbon dioxide activation of waste tire rubber. Carbon 41:1009–1016. https://doi.org/10.1016/S0008-6223(02)00449-9
Johnson HR, Williams RS (1990) Performance of activated carbon in evaporative loss control systems. SAE Trans 99:753–775. https://doi.org/10.4271/902119
Khosravi R, Azizi A, Ghaedrahmati R, Gupta VK, Agarwal S (2017) Adsorption of gold from cyanide leaching solution onto activated carbon originating from coconut shell-optimization, kinetics and equilibrium studies. J Ind Eng Chem 54:464–471. https://doi.org/10.1016/j.jiec.2017.06.036
Lee SW, Daud WMAW, Lee MG (2010) Adsorption characteristics of methyl mercaptan, dimethyl disulfide, and trimethylamine on coconut-based activated carbons modified with acid and base. J Ind Eng Chem 16:973–977. https://doi.org/10.1016/j.jiec.2010.04.002
Borghei M, Laocharoen N, Kibena-Poldsepp E, Johansson LS, Campbell J, Kauppinen E, Tammeveski K, Rojas OJ (2017) Porous N, P-doped carbon from coconut shells with high electrocatalytic activity for oxygen reduction: alternative to Pt-C for alkaline fuel cells. Appl Catal B Environ 204:394–402. https://doi.org/10.1016/j.apcatb.2016.11.029
Hu Z, Srinivasan MP (1999) Preparation of high-surface-area activated carbons from coconut shell. Microporous Mesoporous Mater 27:11–18. https://doi.org/10.1016/S1387-1811(98)00183-8
Xin-Hui D, Srinivasakannan C, Jin-sheng L (2014) Process optimization of thermal regeneration of spent coal based activated carbon using steam and application to methylene blue dye adsorption. J Taiwan Inst Chem E 45:1618–1627. https://doi.org/10.1016/j.jtice.2013.10.019
Zhang J, Zhang W, Zhang H, Pang J, Cao G, Han M, Yang Y (2017) Facile preparation of water soluble phenol formaldehyde resin-derived activated carbon by Na2CO3 activation for high performance supercapacitors. Mater Lett 206:67–70. https://doi.org/10.1016/j.matlet.2017.06.091
Seo SW, Choi YJ, Kim JH, Cho JH, Lee YS, Im JS (2019) Micropore-structured activated carbon prepared by waste PET/petroleum-based pitch. Carbon Lett 29:385–392. https://doi.org/10.1007/s42823-019-00028-w
Heschel W, Klose E (1995) On the suitability of agricultural by-products for the manufacture of granular activated carbon. Fuel 74:1786–1791. https://doi.org/10.1016/0016-2361(95)80009-7
Dyk JCV, Benson SA, Laumb ML, Waanders B (2009) Coal and coal ash characteristics to understand mineral transformations and slag formation. Fuel 88:1057–1063. https://doi.org/10.1016/j.fuel.2008.11.034
Lee HM, Heo YJ, An KH, Jung SC, Chung DC, Park SJ, Kim BJ (2018) A study on optimal pore range for high pressure hydrogen storage behaviors by porous hard carbon materials prepared from a polymeric precursor. Int J Hydrogen Energy 43:5894–5902. https://doi.org/10.1016/j.ijhydene.2017.09.085
Baek J, An KH, Chung DC, Kim BJ (2018) Correlation studies between pore structure and electrochemical performance of activated polymer-based hard carbon with various organic and aqueous electrolytes. Int J Energy Res 42:2927–2939. https://doi.org/10.1002/er.3939
Beak J, Shin HS, Chung DC, Kim BJ (2017) Studies on the correlation between nanostructure and pore development of polymeric precursor-based activated hard carbons: II. Transmission electron microscopy and Raman spectroscopy studies. J Ind Eng Chem 54:324–331. https://doi.org/10.1016/j.jiec.2017.06.007
Kim JH, Hwang SY, Park JE, Lee GB, Kim H, Kim SK, Hong BU (2019) mpact of the oxygen functional group of nitric acid-treated activated carbon on KOH activation reaction. Carbon Lett 29:281–287. https://doi.org/10.1007/s42823-019-00024-0
Teo EYL, Muniandy L, Ng EP, Adam F, Mohamed AR, Jose R, Chong KF (2016) High surface area activated carbon from rice husk as a high performance supercapacitor electrode. Electrochim Acta 192:110–119. https://doi.org/10.1016/j.electacta.2016.01.140
Coromina HM, Walsh DA, Mokaya R (2016) Biomass-derived activated carbon with simultaneously enhanced CO2 uptake for both pre and post combustion capture applications. J Mater Chem A 4:280–289. https://doi.org/10.1039/C5TA09202G
Baek J, Lee HM, An KH, Kim BJ (2019) Preparation and characterization of highly mesoporous activated short carbon fibers from kenaf precursors. Carbon lett. 29:393–399. https://doi.org/10.1007/s42823-019-00042-y
Brunauer S, Emmett PH, Teller E (1938) Adsorption of gases in multimolecular layers. J Am Chem Soc 60:309–319. https://doi.org/10.1021/ja01269a023
Barrett EP, Joyner LG, Halenda PP (1951) The determination at pore volume and area distributions in porous substances. I. Computation from nitrogen isotherms. J Am Chem Soc 73:373–380. https://doi.org/10.1021/ja01145a126
Kierlik E, Rosinberg ML (1990) Free-energy density functional for the inhomogeneous hard-sphere fluid: application to interfacial adsorption. Phys Rev 42:3382–3387. https://doi.org/10.1103/PhysRevA.42.3382
Savitzky A, Golay MJE (1964) Smoothing and differentiation of data by simplified least squares procedures. Anal Chem 36:1627–1639. https://doi.org/10.1021/ac60214a047
Thommes M, Kaneko K, Neimark AV, Olivier JP, Rodriguez-Reinoso F, Rouquerol J, Sing KSW (2015) Physisorption of gases, with special reference to the evaluation of surface area and pore size distribution (IUPAC technical report). Pure Appl Chem 87:1051–1069. https://doi.org/10.1515/pac-2014-1117
Wigmans T (1989) Industrial aspects of production and use of activated carbons. Carbon 27:13–22. https://doi.org/10.1016/0008-6223(89)90152-8
Rodríguez-Reinoso F, Molina-Sabio M, González T (1995) The use of steam and CO2 as activating agents in the preparation of activated carbons. Carbon 33:15–23. https://doi.org/10.1016/0008-6223(94)00100-E
Sing KSW, Williams RT (2004) The use of molecular probes for the characterization of nanoporous adsorbents. Part Part Syst Charact 21:71–79. https://doi.org/10.1002/ppsc.200400923
Hanamoto T, Abe S, Ishikawa K (2012) Adsorbent, process for producing the same, canister and method for using the same. US Patent 8226747B2
Hiltzik LH, Jagiello JZ, Tolles ED, Williams RS (2003) Method for reducing emissions from evaporative emissions control systems. US Patent 6540815B1
Tai FC, Wei C, Chang SH, Chen WS (2009) Raman and X-ray diffraction analysis on unburned carbon powder refined from fly ash. J Raman Spectrosc 41:933–937. https://doi.org/10.1002/jrs.2532
Warren BE (1934) X-ray diffraction study of carbon black. J Chem Phys 2:551–555. https://doi.org/10.1063/1.1749528
Warren BE (1941) X-ray diffraction in random layer lattices. Phys Rev 59:693–698. https://doi.org/10.1103/PhysRev.59.693
Woodhead AL, Souza ML, Church JS (2017) An investigation into the surface heterogeneity of nitric acid oxidized carbon fiber. Appl Surf Sci 401:79–88. https://doi.org/10.1016/j.apsusc.2016.12.218
Feng D, Zhang Y, Zhao Y, Sun S, Gao J (2018) Improvement and maintenance of biochar catalytic activity for in situ biomass tar reforming during pyrolysis and H2O/CO2 gasification. Fuel Process Technol 172:106–114. https://doi.org/10.1016/j.fuproc.2017.12.011
Acknowledgements
This research was supported by the “A Study on the Design of Protective Woven Fabrics Using ACF Materials” funded by the Agency for Defense Development, Republic of Korea (UD170102ID). This research was supported by Nano Material Technology Development Program through the National Research Foundation of Korea (NRF) funded by Ministry of Science and ICT (Project no. 2019M3A7B9071501).
Author information
Authors and Affiliations
Corresponding authors
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Lee, HM., Lee, BH., An, KH. et al. Facile preparation of activated carbon with optimal pore range for high butane working capacity. Carbon Lett. 30, 297–305 (2020). https://doi.org/10.1007/s42823-019-00098-w
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s42823-019-00098-w