Abstract
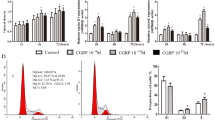
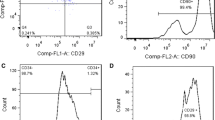
Lentiviral expression vectors for calcitonin gene-related peptide (CGRP) were used to transfect rat bone marrow mesenchymal stem cells (MSCs). After assessing the biological characteristics of proliferation and aging in MSCs transfected with CGRP, we observed the effects of the CGRP-modified rat MSCs on the migration and proliferation of rat vascular smooth muscle cells (VSMCs) in vitro. Rat MSCs were isolated, cultured in vitro, and identified by flow cytometry. A CGRP recombinant lentivirus was transfected into MSCs. The transfection efficiency was determined by fluorescence microscopy and flow cytometry, and CGRP in MSCs was detected by real-time quantitative PCR, ELISA, and immunofluorescence. The proliferation and senescence of CGRP-modified MSCs were evaluated by MTT assay and beta-galactosidase staining. VSMCs were isolated, cultured in vitro, and identified by immunofluorescence. CGRP-modified MSCs and VSMCs were cocultured in a Transwell system. The proliferation and migration of VSMCs were evaluated by scratch testing and the MTT method. Rat bone marrow MSCs showed a spindle-shaped morphology, adherent growth in vitro, positive CD29 and CD90 expression, and negative CD45 expression. CGRP was stably expressed in MSCs after 48 h of recombinant lentivirus transfection. CGRP mRNA and protein secretion in CGRP recombinant lentivirus-transfected MSCs were higher than that in control MSCs. Immunofluorescence showed that CGRP protein could be expressed in CGRP-modified MSCs. The proliferation ability and senescence rates did not differ between lentivirus-transfected MSCs and untransfected MSCs. Rat VSMCs expressed α-SMA protein and exhibited a spindle-shaped morphology and adherent growth in vitro. In Transwell coculture experiments, scratch testing of VSMCs showed that CGRP-modified MSCs could reduce VSMC proliferation and migration. The CGRP gene can be stably expressed in MSCs after CGRP recombinant lentivirus transfection. CGRP recombinant lentivirus transfection has little effect on the proliferation or senescence of MSCs, and CGRP-modified MSCs can inhibit the proliferation and migration of VSMCs. These results lay a foundation for research on the use of CGRP gene-engineered MSCs in restenosis therapy.






Similar content being viewed by others
References
Brozovich FV, Nicholson CJ, Degen CV, Gao YZ, Aggarwal M, Morgan KG (2016) Mechanisms of vascular smooth muscle contraction and the basis for pharmacologic treatment of smooth muscle disorders. Pharmacol Rev 68:476–532. https://doi.org/10.1124/pr.115.010652
Casscells W (1992) Migration of smooth muscle and endothelial cells. Critical events in restenosis. Circulation 86:723–729. https://doi.org/10.1161/01.cir.86.3.723
Correction to "pharmacological discrimination of calcitonin receptor: receptor activity-modifying protein complexes" (2016) Mol Pharmacol 89:484. https://doi.org/10.1124/mol.115.008615err
Daley SJ, Gotlieb AI (1996) Fibroblast growth factor receptor-1 expression is associated with neointimal formation in vitro. Am J Pathol 148:1193–1202
Fang H, Limei Y (2016) Effects of ginsenoside Rg1 on characteristics and functions of adult stem cells. Chin Pharmacol Bull 32:319–322
Guildford AL, Stewart HJ, Morris C, Santin M (2011) Substrate-induced phenotypic switches of human smooth muscle cells: an in vitro study of in-stent restenosis activation pathways. J R Soc Interface 8:641–649. https://doi.org/10.1098/rsif.2010.0532
Haegerstrand A, Dalsgaard CJ, Jonzon B, Larsson O, Nilsson J (1990) Calcitonin gene-related peptide stimulates proliferation of human endothelial cells. Proc Natl Acad Sci U S A 87:3299–3303. https://doi.org/10.1073/pnas.87.9.3299
He F, Zhou A, Feng S (2018a) Use of human amniotic epithelial cells in mouse models of bleomycin-induced lung fibrosis: a systematic review and meta-analysis. PLoS One 13:e0197658. https://doi.org/10.1371/journal.pone.0197658
He F, Zhou A, Feng S, Li Y, Liu T (2018b) Mesenchymal stem cell therapy for paraquat poisoning: a systematic review and meta-analysis of preclinical studies. PLoS One 13:e0194748. https://doi.org/10.1371/journal.pone.0194748
Hirata Y, Takagi Y, Takata S, Fukuda Y, Yoshimi H, Fujita T (1988) Calcitonin gene-related peptide receptor in cultured vascular smooth muscle and endothelial cells. Biochem Biophys Res Commun 151:1113–1121. https://doi.org/10.1016/s0006-291x(88)80481-9
Ito T, Itakura S, Todorov I, Rawson J, Asari S, Shintaku J, Nair I, Ferreri K, Kandeel F, Mullen Y (2010) Mesenchymal stem cell and islet co-transplantation promotes graft revascularization and function. Transplantation 89:1438–1445. https://doi.org/10.1097/tp.0b013e3181db09c4
Li Y, Fiscus RR, Wu J, Yang L, Wang X (1997a) The antiproliferative effects of calcitonin gene-related peptide in different passages of cultured vascular smooth muscle cells. Neuropeptides 31:503–509. https://doi.org/10.1016/S0143-4179(97)90046-9
Li Z, Cheng H, Lederer WJ, Froehlich J, Lakatta EG (1997b) Enhanced proliferation and migration and altered cytoskeletal proteins in early passage smooth muscle cells from young and old rat aortic explants. Exp Mol Pathol 64:1–11. https://doi.org/10.1006/exmp.1997.2204
Liang W, Zhuo X, Tang Z, Wei X, Li B (2015) Calcitonin gene-related peptide stimulates proliferation and osteogenic differentiation of osteoporotic rat-derived bone mesenchymal stem cells. Mol Cell Biochem 402:101–110. https://doi.org/10.1007/s11010-014-2318-6
Liu N, Chen G, Wang X, Yao X, Su J, Li T, Wu X, Zhang Y, Tang J, Tang C (2003) Effects of certain vasoactive peptides on pathogenesis of vascular restenosis. Chin Med Sci J 18:1–8
Mentlein R, Roos T (1996) Proteases involved in the metabolism of angiotensin II, bradykinin, calcitonin gene-related peptide (CGRP), and neuropeptide Y by vascular smooth muscle cells. Peptides 17:709–720. https://doi.org/10.1016/0196-9781(96)00066-6
Morris HR, Panico M, Etienne T, Tippins J, Girgis SI, MacIntyre I (1984) Isolation and characterization of human calcitonin gene-related peptide. Nature 308:746–748. https://doi.org/10.1038/308746a0
Oswald J, Boxberger S, Jorgensen B, Feldmann S, Ehninger G, Bornhauser M, Werner C (2004) Mesenchymal stem cells can be differentiated into endothelial cells in vitro. Stem Cells 22:377–384. https://doi.org/10.1634/stemcells.22-3-377
Owens GK, Kumar MS, Wamhoff BR (2004) Molecular regulation of vascular smooth muscle cell differentiation in development and disease. Physiol Rev 84:767–801. https://doi.org/10.1152/physrev.00041.2003
Potapova IA, Gaudette GR, Brink PR, Robinson RB, Rosen MR, Cohen IS, Doronin SV (2007) Mesenchymal stem cells support migration, extracellular matrix invasion, proliferation, and survival of endothelial cells in vitro. Stem Cells 25:1761–1768. https://doi.org/10.1634/stemcells.2007-0022
Qin XP, Ye F, Hu CP, Liao DF, Deng HW, Li YJ (2004) Effect of calcitonin gene-related peptide on angiotensin II-induced proliferation of rat vascular smooth muscle cells. Eur J Pharmacol 488:45–49. https://doi.org/10.1016/j.ejphar.2004.02.010
Schaeffer C, Vandroux D, Thomassin L, Athias P, Rochette L, Connat JL (2003) Calcitonin gene-related peptide partly protects cultured smooth muscle cells from apoptosis induced by an oxidative stress via activation of ERK1/2 MAPK. Biochim Biophys Acta 1643:65–73. https://doi.org/10.1016/j.bbamcr.2003.09.005
Schuster NM, Rapoport AM (2017) Calcitonin gene-related peptide-targeted therapies for migraine and cluster headache: a review. Clin Neuropharmacol 40:169–174. https://doi.org/10.1097/wnf.0000000000000227
Schwartz SM (1997) Perspectives series: cell adhesion in vascular biology. Smooth muscle migration in atherosclerosis and restenosis. J Clin Invest 99:2814–2816. https://doi.org/10.1172/jci119472
Shi B, Guo Y, Wang Z, Wang D, Shen C (2009) Effects of transplantation of peripheral blood mesenchymal stem cells with hypoxia preconditioning on postangioplasty restenosis in rabbits. Chin J Pathophysiol 25:1686–1691
Teirstein PS, Massullo V, Jani S, Popma JJ, Mintz GS, Russo RJ, Schatz RA, Guarneri EM, Steuterman S, Morris NB, Leon MB, Tripuraneni P (1997) Catheter-based radiotherapy to inhibit restenosis after coronary stenting. N Engl J Med 336:1697–1703. https://doi.org/10.1056/nejm199706123362402
Toutouzas K, Colombo A, Stefanadis C (2004) Inflammation and restenosis after percutaneous coronary interventions. Eur Heart J 25:1679–1687. https://doi.org/10.1016/j.ehj.2004.06.011
Xu G, Xu J, Zhou Q, Wu X (2008) Effect of calcitonin gene relate peptide on proliferation of human mesenchymal stem cells. Mil Med J South China:21–24
Funding
This work was supported by the Science and Technology Fund of Guizhou Province (Qian ke he LH zi [2014] 7576) and the National Natural Science Foundation of China (81060014).
Author information
Authors and Affiliations
Contributions
P.C. and B.S. designed the study; P.C. and S.M. performed the experiments, analyzed the data, and obtained the results; P.C., T.L., and F.H. wrote the manuscript and made the illustrations.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Editor: Tetsuji Okamoto
Rights and permissions
About this article
Cite this article
Chen, P., He, F., Liu, T. et al. Construction of calcitonin gene-related peptide-modified mesenchymal stem cells and analysis of their effects on the migration and proliferation of vascular smooth muscle cells. In Vitro Cell.Dev.Biol.-Animal 56, 181–191 (2020). https://doi.org/10.1007/s11626-019-00429-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11626-019-00429-1