Abstract
Key message
Three pleiotropic QTL regions associated with spikelet number and heading date were identified, with FT-A1 considered the candidate gene for QTspn/Hd.cau-7A.
Abstract
Spikelet number traits and heading date (HD) play key roles in yield improvement of wheat and its wide adaptation to different environments. Here, we used a Recombinant Inbred Lines population derived from a cross between Yi5029 (5029) and Nongda4332 (4332) to construct a high-density genetic linkage map and identify quantitative trait loci (QTL) associated with total spikelet number per spike (TSPN), fertile spikelet number per spike (FSPN), sterile spikelet number per spike (SSPN) and HD. A total of 22 environmentally stable QTL for TSPN, FSPN, SSPN and HD were identified. Notably, three pleiotropic QTL regions for TSPN and HD were detected on chromosomes 2A, 7A and 7D. The QTL associated with TSPN and HD on chromosome 7AS was designated QTspn/Hd.cau-7A. Furthermore, the candidate gene FT-A1 located in the region of QTspn/Hd.cau-7A had a single-nucleotide polymorphism (T–G) within the third exon, which might be the cause of diversity in spikelet number and HD between the two parents. Additionally, we developed a semi-thermal asymmetric reverse PCR (STARP) marker to analyze the geographical distribution and evolution of FT-A1 (T or G) alleles. This study contributes to our understanding of the molecular mechanisms of the four traits (TSPN, FSPN, SSPN and HD) and provides further insights into the genetic relationship between spikelet number traits and HD in wheat.




Similar content being viewed by others
References
Allen GC, Flores-Vergara MA, Krasynanski S, Kumar S, Thompson WF (2006) A modified protocol for rapid DNA isolation from plant tissues using cetyltrimethylammonium bromide. Nat Protoc 1:2320–2325. https://doi.org/10.1038/nprot.2006.384
Alvarez MA, Tranquilli G, Lewis S, Kippes N, Dubcovsky J (2016) Genetic and physical mapping of the earliness per se locus Eps-A (m) 1 in Triticum monococcum identifies EARLY FLOWERING 3 (ELF3) as a candidate gene. Funct Integr Genomics 16:365–382. https://doi.org/10.1007/s10142-016-0490-3
Barnes MR (2010) Genetic variation analysis for biomedical researchers: a primer. In: Barnes MR, Breen G (eds) Genetic variation: methods and protocols. Humana Press, Totowa, pp 1–20
Basten CJ, Weir BS, Zeng Z-B (1997) QTL cartographer: a reference manual and tutorial for QTL mapping. Department of Statistics, North Carolina State University, Raleigh
Boden SA, Cavanagh C, Cullis BR, Ramm K, Greenwood J, Jean Finnegan E, Trevaskis B, Swain SM (2015) Ppd-1 is a key regulator of inflorescence architecture and paired spikelet development in wheat. Nat Plants 1:14016. https://doi.org/10.1038/nplants.2014.16
Bogard M, Jourdan M, Allard V, Martre P, Perretant MR, Ravel C, Heumez E, Orford S, Snape J, Griffiths S, Gaju O, Foulkes J, Le Gouis J (2011) Anthesis date mainly explained correlations between post-anthesis leaf senescence, grain yield, and grain protein concentration in a winter wheat population segregating for flowering time QTLs. J Exp Bot 62:3621–3636. https://doi.org/10.1093/jxb/err061
Bolser DM, Kerhornou A, Walts B, Kersey P (2015) Triticeae resources in ensembl plants. Plant Cell Physiol 56:e3. https://doi.org/10.1093/pcp/pcu183
Bonnin I, Rousset M, Madur D, Sourdille P, Dupuits C, Brunel D, Goldringer I (2008) FT genome A and D polymorphisms are associated with the variation of earliness components in hexaploid wheat. Theor Appl Genet 116:383–394. https://doi.org/10.1007/s00122-007-0676-0
Brancourt-Hulmel M, Doussinault G, Lecomte C, Berard P, Le Buanec B, Trottet M (2003) Genetic improvement of agronomic traits of winter wheat cultivars released in France from 1946 to 1992. Crop Sci 43:37–45. https://doi.org/10.2135/cropsci2003.3700
Chai L, Chen Z, Bian R, Zhai H, Cheng X, Peng H, Yao Y, Hu Z, Xin M, Guo W, Sun Q, Zhao A, Ni Z (2018) Dissection of two quantitative trait loci with pleiotropic effects on plant height and spike length linked in coupling phase on the short arm of chromosome 2D of common wheat (Triticum aestivum L.). Theor Appl Genet 131:2621–2637. https://doi.org/10.1007/s00122-018-3177-4
Chen A, Dubcovsky J (2012) Wheat TILLING mutants show that the vernalization gene VRN1 down-regulates the flowering repressor VRN2 in leaves but is not essential for flowering. PLoS Genet 8:e1003134. https://doi.org/10.1371/journal.pgen.1003134
Chen Z, Cheng X, Chai L, Wang Z, Bian R, Li J, Zhao A, Xin M, Guo W, Hu Z, Peng H, Yao Y, Sun Q, Ni Z (2020) Dissection of genetic factors underlying grain size and fine mapping of QTgw.cau-7D in common wheat (Triticum aestivum L.). Theor Appl Genet 133(1):149–162. https://doi.org/10.1007/s00122-019-03447-5
Cheng X, Chai L, Chen Z, Xu L, Zhai H, Zhao A, Peng H, Yao Y, You M, Sun Q, Ni Z (2015) Identification and characterization of a high kernel weight mutant induced by gamma radiation in wheat (Triticum aestivum L.). BMC Genet 16:127. https://doi.org/10.1186/s12863-015-0285-x
Chu CG, Xu SS, Friesen TL, Faris JD (2008) Whole genome mapping in a wheat doubled haploid population using SSRs and TRAPs and the identification of QTL for agronomic traits. Mol Breed 22:251–266. https://doi.org/10.1007/s11032-008-9171-9
Cui F, Ding AM, Li J, Zhao CH, Wang L, Wang XQ, Qi XL, Li XF, Li GY, Gao JR, Wang HG (2012) QTL detection of seven spike-related traits and their genetic correlations in wheat using two related RIL populations. Euphytica 186:177–192. https://doi.org/10.1007/s10681-011-0550-7
Debernardi JM, Lin H, Chuck G, Faris JD, Dubcovsky J (2017) microRNA172 plays a crucial role in wheat spike morphogenesis and grain threshability. Development 144:1966–1975. https://doi.org/10.1242/dev.146399
Deng S, Wu X, Wu Y, Zhou R, Wang H, Jia J, Liu S (2011) Characterization and precise mapping of a QTL increasing spike number with pleiotropic effects in wheat. Theor Appl Genet 122:281–289. https://doi.org/10.1007/s00122-010-1443-1
Dixon LE, Farre A, Finnegan EJ, Orford S, Griffiths S, Boden SA (2018a) Developmental responses of bread wheat to changes in ambient temperature following deletion of a locus that includes FLOWERING LOCUS T1. Plant Cell Environ 41:1715–1725. https://doi.org/10.1111/pce.13130
Dixon LE, Greenwood JR, Bencivenga S, Zhang P, Cockram J, Mellers G, Ramm K, Cavanagh C, Swain SM, Boden SA (2018b) TEOSINTE BRANCHED1 regulates inflorescence architecture and development in bread wheat (Triticum aestivum). Plant Cell 30:563–581. https://doi.org/10.1105/tpc.17.00961
Dubcovsky J, Loukoianov A, Fu D, Valarik M, Sanchez A, Yan L (2006) Effect of photoperiod on the regulation of wheat vernalization genes VRN1 and VRN2. Plant Mol Biol 60:469–480. https://doi.org/10.1007/s11103-005-4814-2
Faris J, Simons K, Zhang Z, Gill B (2005) The wheat super domestication gene Q. Wheat Info Serv 100:129–148
Finnegan EJ, Ford B, Wallace X, Pettolino F, Griffin PT, Schmitz RJ, Zhang P, Barrero JM, Hayden MJ, Boden SA, Cavanagh CA, Swain SM, Trevaskis B (2018) Zebularine treatment is associated with deletion of FT-B1 leading to an increase in spikelet number in bread wheat. Plant Cell Environ 41:1346–1360. https://doi.org/10.1111/pce.13164
Foulkes MJ, Slafer GA, Davies WJ, Berry PM, Sylvester-Bradley R, Martre P, Calderini DF, Griffiths S, Reynolds MP (2010) Raising yield potential of wheat. III. Optimizing partitioning to grain while maintaining lodging resistance. J Exp Bot 62:469–486. https://doi.org/10.1093/jxb/erq300
Fu D, Szűcs P, Yan L, Helguera M, Skinner JS, Von Zitzewitz J, Hayes PM, Dubcovsky J (2005) Large deletions within the first intron in VRN-1 are associated with spring growth habit in barley and wheat. Mol Genet Genomics 273:54–65. https://doi.org/10.1007/s00438-005-0045-0
Gaju O, Reynolds MP, Sparkes DL, Foulkes MJ (2009) Relationships between large-spike phenotype, grain number, and yield potential in spring wheat. Crop Sci 49:961–973. https://doi.org/10.2135/cropsci2008.05.0285
Gawroński P, Ariyadasa R, Himmelbach A, Poursarebani N, Kilian B, Stein N, Steuernagel B, Hensel G, Kumlehn J, Sehgal SK (2014) A distorted circadian clock causes early flowering and temperature-dependent variation in spike development in the Eps-3Am mutant of einkorn wheat. Genetics 196:1253–1261. https://doi.org/10.1534/genetics.113.158444
Gomez D, Vanzetti L, Helguera M, Lombardo L, Fraschina J, Miralles DJ (2014) Effect of Vrn-1, Ppd-1 genes and earliness per se on heading time in Argentinean bread wheat cultivars. Field Crops Res 158:73–81. https://doi.org/10.1016/j.fcr.2013.12.023
Griffiths S, Simmonds J, Leverington M, Wang Y, Fish L, Sayers L, Alibert L, Orford S, Wingen L, Herry L, Faure S, Laurie D, Bilham L, Snape J (2009) Meta-QTL analysis of the genetic control of ear emergence in elite European winter wheat germplasm. Theor Appl Genet 119:383–395. https://doi.org/10.1007/s00122-009-1046-x
Guo Z, Chen D, Roder MS, Ganal MW, Schnurbusch T (2018) Genetic dissection of pre-anthesis sub-phase durations during the reproductive spike development of wheat. Plant J. https://doi.org/10.1111/tpj.13998
International Wheat Genome Sequencing C (2018) Shifting the limits in wheat research and breeding using a fully annotated reference genome. Science. https://doi.org/10.1126/science.aar7191
Lewis S, Faricelli ME, Appendino ML, Valarik M, Dubcovsky J (2008) The chromosome region including the earliness per se locus Eps-Am 1 affects the duration of early developmental phases and spikelet number in diploid wheat. J Exp Bot 59:3595–3607. https://doi.org/10.1093/jxb/ern209
Li SS, Jia JZ, Wei XY, Zhang XC, Li LZ, Chen HM, Fan YD, Sun HY, Zhao XH, Lei TD, Xu YF, Jiang FS, Wang HG, Li LH (2007) A intervarietal genetic map and QTL analysis for yield traits in wheat. Mol Breed 20:167–178. https://doi.org/10.1007/s11032-007-9080-3
Li C, Distelfeld A, Comis A, Dubcovsky J (2011) Wheat flowering repressor VRN2 and promoter CO2 compete for interactions with NUCLEAR FACTOR-Y complexes. Plant J 67:763–773. https://doi.org/10.1111/j.1365-313X.2011.04630.x
Li C, Lin H, Chen A, Lau M, Jernstedt J, Dubcovsky J (2019) Wheat VRN1 and FUL2 play critical and redundant roles in spikelet meristem identity and spike determinacy. Development 146:dev175398. https://doi.org/10.1242/dev.175398
Liu H, Li H, Hao C, Wang K, Wang Y, Qin L, An D, Li T, Zhang X (2019a) TaDA1, a conserved negative regulator of kernel size, has an additive effect with TaGW2 in common wheat (Triticum aestivum L.). Plant Biotechnol J. https://doi.org/10.1111/pbi.13298
Liu H, Song S, Xing Y (2019b) Beyond heading time: FT-like genes and spike development in cereals. J Exp Bot 70:1–3. https://doi.org/10.1093/jxb/ery408
Long YM, Chao WS, Ma GJ, Xu SS, Qi LL (2017) An innovative SNP genotyping method adapting to multiple platforms and throughputs. Theor Appl Genet 130:597–607. https://doi.org/10.1007/s00122-016-2838-4
Ma Z, Zhao D, Zhang C, Zhang Z, Xue S, Lin F, Kong Z, Tian D, Luo Q (2007) Molecular genetic analysis of five spike-related traits in wheat using RIL and immortalized F2 populations. Mol Genet Genomics 277:31–42. https://doi.org/10.1007/s00438-006-0166-0
Ma J, Ding P, Liu J, Li T, Zou Y, Habib A, Mu Y, Tang H, Jiang Q, Liu Y, Chen G, Wang J, Deng M, Qi P, Li W, Pu Z, Zheng Y, Wei Y, Lan X (2019) Identification and validation of a major and stably expressed QTL for spikelet number per spike in bread wheat. Theor Appl Genet. https://doi.org/10.1007/s00122-019-03415-z
McIntosh RADJ, Rogers WJ, Morris C, Xia XC (2017) Catalogue of gene symbols for wheat: 2017 supplement. Available online at: https://shigen.nig.ac.jp/wheat/komugi/genes/macgene/supplement2017.pdf
Millet E (1986) Genetic control of heading date and spikelet number in common wheat (T. aestivum L.) line ‘Noa’. Theor Appl Genet 72:105–107
Mulki MA, Bi X, von Korff M (2018) FLOWERING LOCUS T3 controls spikelet initiation but not floral development. Plant Physiol 178:1170–1186. https://doi.org/10.1104/pp.18.00236
Muqaddasi QH, Brassac J, Koppolu R, Plieske J, Ganal MW, Roder MS (2019) TaAPO-A1, an ortholog of rice ABERRANT PANICLE ORGANIZATION 1, is associated with total spikelet number per spike in elite European hexaploid winter wheat (Triticum aestivum L.) varieties. Sci Rep 9:13853. https://doi.org/10.1038/s41598-019-50331-9
Nishimura K, Moriyama R, Katsura K, Saito H, Takisawa R, Kitajima A, Nakazaki T (2018) The early flowering trait of an emmer wheat accession (Triticum turgidum L. ssp. dicoccum) is associated with the cis-element of the Vrn-A3 locus. Theor Appl Genet 131:2037–2053. https://doi.org/10.1007/s00122-018-3131-5
Ochagavia H, Prieto P, Savin R, Griffiths S, Slafer G (2018) Dynamics of leaf and spikelet primordia initiation in wheat as affected by Ppd-1a alleles under field conditions. J Exp Bot 69:2621–2631. https://doi.org/10.1093/jxb/ery104
Ortiz-Ferrara G, Mosaad M, Mahalakshmi V, Fischer R (1995) Photoperiod and vernalization response of wheat under controlled environment and field conditions. Plant Breed 114:505–509. https://doi.org/10.1111/j.1439-0523.1995.tb00845.x
Reynolds M, Rajaram S, Sayre K (1999) Physiological and genetic changes of irrigated wheat in the post–green revolution period and approaches for meeting projected global demand. Crop Sci 39:1611–1621. https://doi.org/10.2135/cropsci1999.3961611x
Shaw LM, Turner AS, Laurie DA (2012) The impact of photoperiod insensitive Ppd-1a mutations on the photoperiod pathway across the three genomes of hexaploid wheat (Triticum aestivum). Plant J 71:71–84
Shaw LM, Lyu B, Turner R, Li C, Chen F, Han X, Fu D, Dubcovsky J (2018) FLOWERING LOCUS T2 regulates spike development and fertility in temperate cereals. J Exp Bot 70:193–204. https://doi.org/10.1093/jxb/ery350
Sreenivasulu N, Schnurbusch T (2012) A genetic playground for enhancing grain number in cereals. Trends Plant Sci 17:91–101. https://doi.org/10.1016/j.tplants.2011.11.003
van Oj W (2006) JoinMap 4.0: Software for the calculation of genetic linkage maps in experimental populations. Kyazma BV, Wageningen
Van Os H, Stam P, Visser RG, Van Eck HJ (2005) RECORD: a novel method for ordering loci on a genetic linkage map. Theor Appl Genet 112:30–40. https://doi.org/10.1007/s00122-005-0097-x
Wang JS, Liu WH, Wang H, Li LH, Wu J, Yang XM, Li XQ, Gao AN (2011) QTL mapping of yield-related traits in the wheat germplasm 3228. Euphytica 177:277–292. https://doi.org/10.1007/s10681-010-0267-z
Wang S, Basten C, Zeng Z (2012) Windows QTL cartographer 2.5. Department of Statistics, North Carolina State University, Raleigh
Wang S, Wong D, Forrest K, Allen A, Chao S, Huang BE, Maccaferri M, Salvi S, Milner SG, Cattivelli L (2014) Characterization of polyploid wheat genomic diversity using a high-density 90,000 single nucleotide polymorphism array. Plant Biotechnol J 12:787–796. https://doi.org/10.1111/pbi.12183
Xu Y, Wang R, Tong Y, Zhao H, Xie Q, Liu D, Zhang A, Li B, Xu H, An D (2014) Mapping QTLs for yield and nitrogen-related traits in wheat: influence of nitrogen and phosphorus fertilization on QTL expression. Theor Appl Genet 127:59–72. https://doi.org/10.1007/s00122-013-2201-y
Yan L, Loukoianov A, Tranquilli G, Helguera M, Fahima T, Dubcovsky J (2003) Positional cloning of the wheat vernalization gene VRN1. Proc Natl Acad Sci USA 100:6263–6268. https://doi.org/10.1073/pnas.0937399100
Yan LL, Loukoianov A, Blechl A, Tranquilli G, Ramakrishna W, SanMiguel P, Bennetzen JL, Echenique V, Dubcovsky J (2004) The wheat VRN2 gene is a flowering repressor down-regulated by vernalization. Science 303:1640–1644. https://doi.org/10.1126/science.1094305
Yan L, Fu D, Li C, Blechl A, Tranquilli G, Bonafede M, Sanchez A, Valarik M, Yasuda S, Dubcovsky J (2006) The wheat and barley vernalization gene VRN3 is an orthologue of FT. Proc Natl Acad Sci USA 103:19581–19586. https://doi.org/10.1073/pnas.0607142103
Yao HN, Xie Q, Xue SL, Luo J, Lu JK, Kong ZX, Wang YP, Zhai WL, Lu N, Wei R, Yang Y, Han YZ, Zhang Y, Jia HY, Ma ZQ (2019) HL2 on chromosome 7D of wheat (Triticum aestivum L.) regulates both head length and spikelet number. Theor Appl Genet 132:1789–1797. https://doi.org/10.1007/s00122-019-03315-2
Yu K, Liu D, Wu W, Yang W, Sun J, Li X, Zhan K, Cui D, Ling H, Liu C, Zhang A (2017) Development of an integrated linkage map of einkorn wheat and its application for QTL mapping and genome sequence anchoring. Theor Appl Genet 130:53–70. https://doi.org/10.1007/s00122-016-2791-2
Zeng ZB (1994) Precision mapping of quantitative trait loci. Genetics 136:1457–1468
Zhai H, Feng Z, Li J, Liu X, Xiao S, Ni Z, Sun Q (2016) QTL analysis of spike morphological traits and plant height in winter wheat (Triticum aestivum L.) using a high-density SNP and SSR-based linkage map. Front Plant Sci 7:1617. https://doi.org/10.3389/fpls.2016.01617
Zhai H, Feng Z, Du X, Song Y, Liu X, Qi Z, Song L, Li J, Li L, Peng H, Hu Z, Yao Y, Xin M, Xiao S, Sun Q, Ni Z (2018) A novel allele of TaGW2-A1 is located in a finely mapped QTL that increases grain weight but decreases grain number in wheat (Triticum aestivum L.). Theor Appl Genet 131:539–553. https://doi.org/10.1007/s00122-017-3017-y
Zhang B, Liu X, Xu W, Chang J, Li A, Mao X, Zhang X, Jing R (2015) Novel function of a putative MOC1 ortholog associated with spikelet number per spike in common wheat. Sci Rep 5:12211. https://doi.org/10.1038/srep12211
Zhou Y, Conway B, Miller D, Marshall D, Cooper A, Murphy P, Chao S, Brown-Guedira G, Costa J (2017) Quantitative trait loci mapping for spike characteristics in hexaploid wheat. Plant Genome. https://doi.org/10.3835/plantgenome2016.10.0101
Zikhali M, Wingen LU, Leverington-Waite M, Specel S, Griffiths S (2017) The identification of new candidate genes Triticum aestivum FLOWERING LOCUS T3–B1 (TaFT3-B1) and TARGET OF EAT1 (TaTOE1-B1) controlling the short-day photoperiod response in bread wheat. Plant Cell Environ 40:2678–2690. https://doi.org/10.1111/pce.13018
Acknowledgements
This work was financially supported by the Major Program of the National Natural Science Foundation of China (Grant No. 91935304 and 31991210) and the National Key Research and Development Program of China (Grant No. 2017YFD0101004). The authors would like to thank Yanming Ma (Institute of Crop Germplasm Resource, Xinjiang Academy of Agricultural Sciences) for kindly provided the data of 152 wheat accessions from Xinjiang, China used for analyzing the effects of FT-A1.
Author information
Authors and Affiliations
Contributions
ZN conceived the project; XC developed the RIL population; ZC constructed the linkage map. ZC, XC, ZW, RB and DD collected data for the RIL population under six environments; ZC developed markers for the QTL region of interest; ZC and LC developed the STARP marker to genotype hexaploid and tetraploid wheat; ZW helped to analysis the results of re-sequencing; QS, AZ, MX, WG, ZH, HP and YY assisted in revising the manuscript; ZC analyzed experimental results and wrote the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Communicated by Albrecht E. Melchinger.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
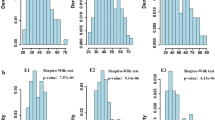
Figure S1
Frequency distribution histograms of the 5029/4332 RIL population for total spikelet number (TSPN), fertile spikelet number (FSPN), sterile spikelet number (SSPN) and heading date (HD) based on the BLUP value. The y-axis represents the density of each trait. P < 0.01 indicates a significant departure from the normal distribution (Shapiro–Wilk test). (TIFF 426 kb)
Figure S2
Comparation of total spikelet number per spike, floret number per spikelet and grain number per spike between FT-A1(T) and FT-A1(G) alleles of 152 accessions from Xinjiang from China. (TIFF 186 kb)
Figure S3
Relative expression levels of FT-A1 with different haplotypes. 6554 represents Jingdong6, JM22 represents Jimai22, BN207 represents Bainong207 and 3338 represents Nongda3338. (TIFF 166 kb)
Rights and permissions
About this article
Cite this article
Chen, Z., Cheng, X., Chai, L. et al. Pleiotropic QTL influencing spikelet number and heading date in common wheat (Triticum aestivum L.). Theor Appl Genet 133, 1825–1838 (2020). https://doi.org/10.1007/s00122-020-03556-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00122-020-03556-6