Abstract
Under proteotoxic stress, some cells survive whereas others die. The mechanisms governing this heterogeneity in cell fate remain unknown. Here we report that condensation and phase transition of heat-shock factor 1 (HSF1), a transcriptional regulator of chaperones1,2, is integral to cell-fate decisions underlying survival or death. During stress, HSF1 drives chaperone expression but also accumulates separately in nuclear stress bodies called foci3,4,5,6. Foci formation has been regarded as a marker of cells actively upregulating chaperones3,6,7,8,9,10. Using multiplexed tissue imaging, we observed HSF1 foci in human tumours. Paradoxically, their presence inversely correlated with chaperone expression. By live-cell microscopy and single-cell analysis, we found that foci dissolution rather than formation promoted HSF1 activity and cell survival. During prolonged stress, the biophysical properties of HSF1 foci changed; small, fluid condensates enlarged into indissoluble gel-like arrangements with immobilized HSF1. Chaperone gene induction was reduced in such cells, which were prone to apoptosis. Quantitative analysis suggests that survival under stress results from competition between concurrent but opposing mechanisms. Foci may serve as sensors that tune cytoprotective responses, balancing rapid transient responses and irreversible outcomes.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Data availability
Source data for Figs. 1–4 and Extended Data Figs. 1–10 are provided with the paper. The numerical data are available through the Synapse SAGE Bionetworks portal with accession number syn20505972. Chemical screen results have been deposited in the NCBI PubChem Bioassay database under assay ID (AID) 1347162. All other data supporting the findings of this study are available from the corresponding author on reasonable request.
Code availability
Matlab codes used to perform the t-CyCIF and live-cell image analysis are available on GitHub at https://github.com/santagatalab. Live-cell tracking was performed using the p53cinema Matlab GUI (https://github.com/balvahal/p53CinemaManual). The repositories are public, and the codes are freely downloadable from the GitHub website. Further information and requests for resources and reagents should be directed to and will be fulfilled by the corresponding author.
References
Lindquist, S. The heat-shock response. Annu. Rev. Biochem. 55, 1151–1191 (1986).
Vihervaara, A. & Sistonen, L. HSF1 at a glance. J. Cell Sci. 127, 261–266 (2014).
Sarge, K. D., Murphy, S. P. & Morimoto, R. I. Activation of heat shock gene transcription by heat shock factor 1 involves oligomerization, acquisition of DNA-binding activity, and nuclear localization and can occur in the absence of stress. Mol. Cell Biol. 13, 1392–1407 (1993).
Cotto, J., Fox, S. & Morimoto, R. HSF1 granules: a novel stress-induced nuclear compartment of human cells. J. Cell Sci. 110, 2925–2934 (1997).
Jolly, C., Morimoto, R., Robert-Nicoud, M. & Vourc’h, C. HSF1 transcription factor concentrates in nuclear foci during heat shock: relationship with transcription sites. J. Cell Sci. 110, 2935–2941 (1997).
Jolly, C., Usson, Y. & Morimoto, R. I. Rapid and reversible relocalization of heat shock factor 1 within seconds to nuclear stress granules. Proc. Natl Acad. Sci. USA 96, 6769–6774 (1999).
Nonaka, T., Akimoto, T., Mitsuhashi, N., Tamaki, Y. & Nakano, T. Changes in the number of HSF1 positive granules in the nucleus reflects heat shock semiquantitatively. Cancer Lett. 202, 89–100 (2003).
Au, Q., Kanchanastit, P., Barber, J. R., Ng, S. C. & Zhang, B. High-content image-based screening for small-molecule chaperone amplifiers in heat shock. J. Biomol. Screen. 13, 953–959 (2008).
Biamonti, G. & Vourc’h, C. Nuclear stress bodies. Cold Spring Harb. Perspect. Biol. 2, a000695 (2010).
Chowdhary, S., Kainth, A. S., Pincus, D. & Gross, D. S. Heat shock factor 1 drives intergenic association of its target gene loci upon heat shock. Cell Rep. 26, 18–28.e5 (2019).
Li, J., Labbadia, J. & Morimoto, R. I. Rethinking HSF1 in stress, development, and organismal health. Trends Cell Biol. 27, 895–905 (2017).
Anckar, J. & Sistonen, L. Regulation of HSF1 function in the heat stress response: implications in aging and disease. Annu. Rev. Biochem. 80, 1089–1115 (2011).
Metz, A., Soret, J., Vourc’h, C., Tazi, J. & Jolly, C. A key role for stress-induced satellite III transcripts in the relocalization of splicing factors into nuclear stress granules. J. Cell Sci. 117, 4551–4558 (2004).
Rizzi, N. et al. Transcriptional activation of a constitutive heterochromatic domain of the human genome in response to heat shock. Mol. Biol. Cell 15, 543–551 (2004).
Jolly, C. et al. In vivo binding of active heat shock transcription factor 1 to human chromosome 9 heterochromatin during stress. J. Cell Biol. 156, 775–781 (2002).
Jolly, C. et al. Stress-induced transcription of satellite III repeats. J. Cell Biol. 164, 25–33 (2004).
Eymery, A., Souchier, C., Vourc’h, C. & Jolly, C. Heat shock factor 1 binds to and transcribes satellite II and III sequences at several pericentromeric regions in heat-shocked cells. Exp. Cell Res. 316, 1845–1855 (2010).
Holmberg, C. I., Illman, S. A., Kallio, M., Mikhailov, A. & Sistonen, L. Formation of nuclear HSF1 granules varies depending on stress stimuli. Cell Stress Chaperones 5, 219–228 (2000).
Lin, J.-R. et al. Highly multiplexed immunofluorescence imaging of human tissues and tumors using t-CyCIF and conventional optical microscopes. eLife 7, e31657 (2018).
Du, Z. et al. Qualifying antibodies for image-based immune profiling and multiplexed tissue imaging. Nat. Protoc. 14, 2900–2930 (2019).
Calderwood, S. K. & Gong, J. Heat shock proteins promote cancer: it’s a protection racket. Trends Biochem. Sci. 41, 311–323 (2016).
Banaszynski, L. A., Chen, L.-C., Maynard-Smith, L. A., Ooi, A. G. L. & Wandless, T. J. A rapid, reversible, and tunable method to regulate protein function in living cells using synthetic small molecules. Cell 126, 995–1004 (2006).
Zuo, J., Baler, R., Dahl, G. & Voellmy, R. Activation of the DNA-binding ability of human heat shock transcription factor 1 may involve the transition from an intramolecular to an intermolecular triple-stranded coiled-coil structure. Mol. Cell. Biol. 14, 7557–7568 (1994).
Deng, J. et al. BH3 profiling identifies three distinct classes of apoptotic blocks to predict response to ABT-737 and conventional chemotherapeutic agents. Cancer Cell 12, 171–185 (2007).
Mateju, D. et al. An aberrant phase transition of stress granules triggered by misfolded protein and prevented by chaperone function. EMBO J. 36, 1669–1687 (2017).
Kroschwald, S., Maharana, S. & Simon, A. Hexanediol: a chemical probe to investigate the material properties of membrane-less compartments. Matters 3, e201702000010 (2017).
Wang, J. et al. A molecular grammar governing the driving forces for phase separation of prion-like RNA binding proteins. Cell 174, 688–699.e16 (2018).
Neudegger, T., Verghese, J., Hayer-Hartl, M., Hartl, F. U. & Bracher, A. Structure of human heat-shock transcription factor 1 in complex with DNA. Nat. Struct. Mol. Biol. 23, 140–146 (2016).
Guettouche, T., Boellmann, F., Lane, W. S. & Voellmy, R. Analysis of phosphorylation of human heat shock factor 1 in cells experiencing a stress. BMC Biochem. 6, 4 (2005).
Budzyński, M. A., Puustinen, M. C., Joutsen, J. & Sistonen, L. Uncoupling stress-inducible phosphorylation of heat shock factor 1 from its activation. Mol. Cell. Biol. 35, 2530–2540 (2015).
Santagata, S. et al. High levels of nuclear heat-shock factor 1 (HSF1) are associated with poor prognosis in breast cancer. Proc. Natl Acad. Sci. USA 108, 18378–18383 (2011).
Mendillo, M. L. et al. HSF1 drives a transcriptional program distinct from heat shock to support highly malignant human cancers. Cell 150, 549–562 (2012).
Hyman, A. A., Weber, C. A. & Jülicher, F. Liquid–liquid phase separation in biology. Annu. Rev. Cell Dev. Biol. 30, 39–58 (2014).
Chong, S. et al. Imaging dynamic and selective low-complexity domain interactions that control gene transcription. Science 351, eaar2555 (2018).
Pessina, F. et al. Functional transcription promoters at DNA double-strand breaks mediate RNA-driven phase separation of damage-response factors. Nat. Cell Biol. 21, 1286–1299 (2019).
Kroschwald, S. et al. Different material states of Pub1 condensates define distinct modes of stress adaptation and recovery. Cell Rep. 23, 3327–3339 (2018).
Riback, J. A. et al. Stress-triggered phase separation is an adaptive, Evolutionarily tuned response. Cell 168, 1028–1040 (2017).
Trivedi, P. et al. The inner centromere is a biomolecular condensate scaffolded by the chromosomal passenger complex. Nat. Cell Biol. 21, 1127–1137 (2019).
Suderman, R., Bachman, J. A., Smith, A., Sorger, P. K. & Deeds, E. J. Fundamental trade-offs between information flow in single cells and cellular populations. Proc. Natl Acad. Sci. USA 114, 5755–5760 (2017).
Cherkasov, V. et al. Coordination of translational control and protein homeostasis during severe heat stress. Curr. Biol. 23, 2452–2462 (2013).
Hiraoka, Y., Sedat, J. W. & Agard, D. A. Determination of three-dimensional imaging properties of a light microscope system. Partial confocal behavior in epifluorescence microscopy. Biophy. J. 57, 325–333 (1990).
Aguet, F., Antonescu, C. N., Mettlen, M., Schmid, S. L. & Danuser, G. Advances in analysis of low signal-to-noise images link dynamin and AP2 to the functions of an endocytic checkpoint. Dev. Cell 26, 279–291 (2013).
Acknowledgements
This work was supported by NIH grants R01-CA194005 (S.S.), U54-CA225088 (P.K.S. and S.S.), R00-CA188679 (K.A.S.) and T32HL007627 (G.G.), the Ludwig Center at Harvard (P.K.S. and S.S.) and the Harvard T.H. Chan School of Public Health Dean’s Fund for Scientific Advancement (K.A.S.). This work was supported in part by the Koch Institute Support Grant P30-CA14051 and the Dana-Farber/Harvard Cancer Center Support Grant no. P30-CA06516 from the National Cancer Institute. We thank J. Lin and J. Muhlich for help with CyCIF imaging and analysis, D. Landgraft, M. Shoulders and G. Lahav for providing reagents and S. Alberti for helpful discussions. We thank the Koch Institute Swanson Biotechnology Center for technical support, specifically J. H. Cheah and C. K. Soule in the High Throughput Sciences Facility and the Confocal Microscopy Core at Brigham and Women’s Hospital.
Author information
Authors and Affiliations
Contributions
Conceptualization: G.G., S.L.L., K.A.S., P.K.S., L.W. and S.S. Methodology: G.G., C.Y., G.N.J. and K.A.S. Software: G.G. and C.Y. Validation: G.G., R.R. and C.G.L. Formal analysis: G.G. Investigation: G.G., R.R., G.N.J. and C.G.L. Resources: S.L.L., K.A.S., P.K.S. and S.S. Data curation: G.G. Writing, original draft; G.G. and S.S. Writing, reviewing and editing: G.G., R.R., C.Y., G.N.J., C.G.L., K.A.S., P.K.S., L.W. and S.S. Visualization: G.G., C.Y., G.N.J., R.R. and C.G.L. Supervision: S.L.L., K.A.S., P.K.S., L.W. and S.S. Project administration: S.L.L. and S.S. Funding acquisition: S.L.L., P.K.S. and S.S.
Corresponding author
Ethics declarations
Competing interests
P.K.S. is a member of the Scientific Advisory Board of RareCyte and co-founder of Glencoe Software, which contributes to and supports the open-source OME/OMERO image informatics software used in this paper. S.S. is a consultant for RareCyte. The other authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data
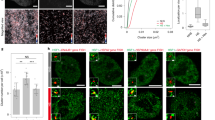
Extended Data Fig. 1 HSF1 foci quantification in colon adenocarcinoma tissues.
a,b, Composite images of immunofluorescence data of HSF1 (clone 10H8, green) and DAPI nuclear stain (blue) from a tissue sections of colon adenocarcinoma with adjacent non-neoplastic colon tissue. T, tumor; N, normal. a, Insets are two regions at the borders between tumor and normal tissue. b, Insets 1-3 tumor tissue, inset 4 normal colon tissue. c, High power composite image of t-CyCIF data showing CDX2 (red), αSMA (purple) CD45 (yellow). d, Quantification of t-CyCIF data from three colon adenocarcinoma whole tissue sections indicating the lineage of cells positive for HSF1 foci (“Foci+”, HSF1-FI > 0.05). e, Bar graph of Foci+ cells in colon adenocarcinoma cases (n = 93 patients; from tissue microarray (TMA); four cores per patient, average +/- SEM) from t-CyCIF data. f, Scatter plot of the frequency of Foci+ cells in tumor versus non-tumor compartments (CD45+ immune cells, yellow; αSMA+ stromal cells, purple). Each dot represents CD45 or αSMA data from a single core from TMA. g, Bar graph of Foci+ cells from t-CyCIF data from TMA (tumor, red; immune, yellow; stroma, purple, n = 87 patients, average + SEM). h, 2D single-cell, spatially-averaged heatmap of t-CyCIF data of HSP70 and HSP90 expression normalized over median expression (scalebar 1mm). i, Kernel density (KD) estimated frequency distribution plot of HSP70 chaperone protein expression in normal colon tissue (red) and adenocarcinoma tissue (blue) from t-CyCIF. j, Top, 2D density plot of HSF1 concentration versus chaperone proteins from t-CyCIF of colon adenocarcinoma in Fig. 1b, c (n ~ 250,000 cells). Bottom, corresponding scatter plot (subsampling of 1,000 cells, one dot per cell; dashed line, linear fit). k, Scatter plot of mean HSF1-FI versus mean chaperone levels of the TMA (one dot per core; color represents the number of cells in core). Source data are provided in Source Data Extended Data Fig. 1.
Extended Data Fig. 2 HSF1 foci quantification in myeloma/plasmacytoma tissues.
a,b, 2D cell density plots of two antibodies against Hsp70 (a) and Hsp90 (b) protein from 9 patient samples of plasmacytoma/myeloma from Fig. 1e–k. c–f, Statistics for each of the 9 plasmacytoma/myeloma patient samples. c, Single-cell scatter plot of signal from Hsp70 EPR16892 antibody versus Hsp90 C45G5 antibody. Red lines represent the threshold identified in Fig. 1 for which cells were considered positive or negative for the corresponding chaperone protein expression. d, percentage of cells with HSF1 foci (HSF1-FI > 0.05) divided by cells positive or negative for both HSP70 and HSP90 chaperones (“Chap++” and “Chap–” respectively). e, Number of cells in each Chap++ and Chap–category. f, Mean HSF1 levels per chaperone category. Source data are provided in Source Data Extended Data Fig. 2.
Extended Data Fig. 3 Time-lapse microscopy of tagged HSF1 in tissue culture cell lines.
a, Western blot on U2OS cell lines expressing fluorescent protein tagged HSF1. Lanes: 1) U2OS, 2) U2OS HSF1-mVenus, 3) U2OS HSF1-mEGFP, 4) U2OS HSF1-mTQ2, 5) U2OS HSF1Δp-mTQ2, 6)-7) U2OS HSF1-mVenus FKBP-cHSF1-mTQ2,6) no Shield-1, 7) 500nM Shield-1 for 24 hours. Top part of membrane, anti-HSF1 monoclonal antibody. Bottom part of membrane, anti-GAPDH antibody (see Unprocessed Blots Extended Data Fig. 3 for unprocessed blot image). b, Quantification of bands in lanes 2-4 from western blot image in a (see Methods for Quantification details). c, Left, kernel density estimate (KD) pdf of HSF1 antibody signal level in cells from 9 plasmacytoma/myeloma patient samples from Fig. 1e–k (total of n ~ 2*106 cells). Right, KD pdf of HSF1 antibody level in U2OS (blue) and U2OS HSF1-mVenus cells (orange), n ~ 17,000 and 20,000 cells. d, Fold induction of HSPA1B, HSPA6, and HSP90AA1. RT–qPCR from bulk cell populations in U2OS, U2OS HSF1-mEGFP 3 hours post MG132 2.5uM (bars are biological replicates). e,f, Time-lapse microscopy of U2OS HSF1-mVenus cells treated with 42 °C temperature (scalebar = 10 µm) and quantification (n ~ 16,000-24,000 cells, median + 75th percentile, see Source Data Extended Data Fig. 3 for exact cell numbers and single cell data). f, Single-cell HSF1-FI quantification. g, Dynamics of the percentage of cells with foci after proteotoxic stressor addition (U2OS HSF1-mVenus cells, live cell time-lapse microscopy, 30 minutes intervals, n ~ 260 cells, see Source Data Extended Data Fig. 3), using three different thresholds to HSF1-FI to define the foci positivity, 0.025, 0.05 and 0.1. h, Fold induction of HSP70 genes (HSPA1B, HSPA6) and HSP90AA1 using RT–qPCR from bulk cell populations after gradient doses of MG132 and STA9090 (MG132: 5, 2.5, 1.25, 0.625 µM; STA9090: 50, 25, 12.5, 6.25 nM). Statistics and Source Data are provided in Source Data Extended Data Fig. 3.
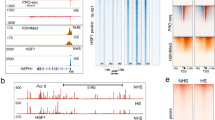
Extended Data Fig. 4 Relationship between HSF1 foci and HSF1 transcriptional activity.
a, Time-lapse heatmaps for HSF1-FI and HSP70 Transcriptional Reporter (TR) induction dTR/dt time derivative (one cell per row, n=152 cells). Black line, entry time of HSF1-FI < 0.2 b, Scatter plot of HSF1-FI versus dTR/dt from all instances from panel a (n=3,400 cell measurements, red line = linear fit). c,d, Scatter plot of TR versus (c.) time-integrated and (d.) maximum HSF1-FI 6 hours post 50nM STA9090 (Pearson correlation, bootstrapping p-values). e, Single-cell trace examples of time relationship between HSF1-FI and TR. Each dot represents a single-cell measurement, the color represents the time post STA9090 50nM addition (starting at 2 hours). Blue lines, linear fit y = α*x + β. f, Bootstrapping on parameter “α” in fits from panel e. Blue line, KD pdf of the α parameters from cells with R2>0.5. The matching between foci time dynamics and transcriptional reporter (TR) induction were randomly permuted 1,000 times. Red line, mean distribution of the α parameters for the permuted traces over all 1,000 permutations (light red area, 25th to 75th percentile). g, HSPA1A RNA FISH time course on U2OS HSF1-mEGFP cells post 100 nM STA9090 (n > 2200 cells per sample, see Source Data Extended Data Fig. 4 for statistics details). h, RNA FISH for HSPA1A on HCT15 HSF1-mVenus cells and U2OS HSF1-mEGFP cells after proteasome inhibition (Bortezomib 100 nM, 4 hours post). i-j, Immunofluorescence on U2OS HSF1-GFP cells (HSF1 green, HSP70 red, clone C92F3A-5). 1 µM MG132 overnight treatment. scalebar = 10 µm and j, quantification. Yellow, HSF1-FI<0.01, n = 1430 cells. Red, 0.01 < HSF1-FI < 0.2, n = 1156 cells. Blue HSF1-FI > 0.2, n = 492 cells. Inset, KD estimated distributions (two-sided KS p-values: high vs medium focied cells, p~10-41, medium vs low focied cells, p~10-18). Source data are provided in Source Data Extended Data Fig. 4.
Extended Data Fig. 5 Relationship between HSF1 foci and cell death by time-lapse microscopy.
a, Time-lapse microscopy traces of HSF1-GFP cell concentration from single cells followed for 12 hours at 30 minute intervals post 2.5 µM MG132 (mean +/- SEM). Cells are separated between those that survived (blue, n = 96 cells) and those that died (red, n = 54 cells) as in Fig. 3a. b,c, Bar graph of HSF1-FI (b) and HSF1 concentration (c) at 5 and 10 hours after 2.5 µM MG132 comparing cells that survive (blue) and cells that die (red) during the time lapse microscopy experiment in panel a, and Fig. 3a (two-sided KS p-value with 0.05 significance threshold, mean +/- SEM). d, Immunofluorescence on U2OS cells with mVenus protein endogenously inserted in C-terminus of HSF1 genomic locus (HSF1-mVenusKI). Two independent clones (called “B12” and “C5”) were analysed. Scalebar = 10 µm. e, Quantification of time-lapse microscopy on U2OS HSF1-mVenusKI clone B12 and C5 after 5 µM MG132. Red = cells that died during the 14 hours experiment (n = 153 cells and n = 156 cells), green = cells that survived (n = 101 cells and n = 199 cells, mean +/- SEM). Source Data are provided in Source Data Extended Data Fig. 5.
Extended Data Fig. 6 Relationship between HSF1 foci and cell death.
a, Quantification of single-cell time-lapse microscopy on U2OS cHSF1-CFP after induction with 250 nM Shield-1. Upper panels, HSF1 Focus Index (HSF1-FI). Lower panels, total cHSF1 induction. Cells are divided between levels of total cHSF1 induction, on the left are cells with low induction level and on the right are cells with high induction levels. Red = cells that died during the 29 hours experiment n = 12 cells and n = 14 cells, green = cells that survived n = 182 cells and n = 27 cells (two-sided KS p-value with 0.05 significance threshold, 20-minute intervals, mean +/- SEM). b, BIM peptide titration on cells treated with 1.2 µM MG132. Cells increasingly lost more cytochrome c. Full cell distribution (yellow line) is divided into cells that released cytochrome c (“dying cells”, red dashed distribution) and cells that had not released cytochrome c (“healthy cells”, dashed blue line distribution). Histograms show the distribution of HSF1-FI in healthy (left column) and dying cells (right column). Across BIM concentrations, the foci levels in dying cells is higher than in healthy cells. Source Data are provided in Source Data Extended Data Fig. 6.
Extended Data Fig. 7 Small molecule imaging-based screen for HSF1 foci modifiers.
a, Summary scatter of Selleck Cambridge Cancer Library high content imaging screening results for changes in HSF1 foci. Each dot is a compound out of total of 392 compounds. Each compound was tested in duplicated wells each imaged in 36 non-overlapping fields. Total cell count per compound well varied between 2000 and 8000 cells (see Supplementary Information Table 3, 4 and 5 for compound details and full results). Compounds of interest (red dots) are either proteotoxic compounds or topoisomerase inhibitors. b, Zoomed in scatter of grey shaded area in a. c. Sample images from high content imaging screen (full single field displayed from one of two replicated wells, scalebar = 10 µm).
Extended Data Fig. 8 High resolution 3-dimensional reconstruction of HSF1 foci in fixed cells.
a–f, Single cells statistics from U2OS HSF1-mEGFP cells fixed with paraformaldehyde at 6 time points from 0 to 8 hours after the addition of 2.5 µM MG132. Z-stacked images were deconvolved and both cells and HSF1 foci segmented. a, Deconvolved images of U2OS HSF1-mEGFP cells from z-stack imaging. Top row, x-y 2D view. Second row, 3-dimensional view of single cells with surface rendering in Imaris and their overlay (2 µm scalebar). A total of n = 377 cells analyzed. b, Histogram of cube root of foci volume for all foci in experiment from panel a (n = 3850 foci total). Color lines represent the 3-component Gaussian mixture model fitting of the histogram (green line global fit, orange, yellow and purple single component fits weighted by component contribution). The black lines represent the threshold identified by the GMM and used to separate foci between small (< 136 voxels) medium (between 136 and 592 voxels) and large foci (> 592 voxels). c–f, Single cells statistics. c–e, Blue, one dot per cell, n = 44, 67, 65, 44, 77 and 77 cells. Black, mean +/- SD. c, Total GFP signal, d, nucleus area, and e, mean focus volume for all foci independent of size (n = 10, 2092, 2589, 1684, 2410, 2334 foci per time point, mean +/- SEM). f, Mean fluorescent signal of HSF1 foci stratified by large (red), medium (green) and small (blue) foci volume (mean +/- 25th percentile). Source data are provided in Source Data Extended Data Fig. 8.
Extended Data Fig. 9 Tagged HSF1 FRAP traces and statistics.
a–d, Fluorescent recovery after photobleaching (FRAP) single-cell traces and exponential fits. Region of interest photobleached after 5 seconds from start of imaging. a,b, Whole focus FRAP traces after 2.5 µM MG132 treatment a, 2 hours after treatment and b, 8 hours after treatment. c,d, Half-focus FRAP traces after 2.5 µM MG132 treatment. c, 1.5 hours after treatment. d, 8 hours after treatment. e, Whole focus FRAP Thalf recovery time estimates after 2.5 µM MG132 addition (mean +/- SEM, n = 28, 17, 27, 19, 19, 10 foci per time point). f,g, Whole focus FRAP on HCT15 HSF1-YFP parameter estimates 1.5 and 5 hours after 5 µM MG132 treatment. f, Immobile fraction and g, Thalf recovery time (n = 14 foci, two-sided KS p-value). h, Half-focus FRAP Thalf recovery time estimates 1.5 and 8 hours after the addition of 2.5 µM MG132 (mean +/- SEM, n = 30 and 32 foci per time point, two-sided KS p-value). Source Data are provided in Source Data Extended Data Fig. 9.
Extended Data Fig. 10 Further characterization of HSF1 foci solubility.
a,b. Sample images of time-lapse microscopy of U2OS HSF1-GFP cells after 10% 1,6-hexandiol addition (images acquired at 20 second intervals; cells were pre-stressed with 10 µM MG132). A total of 188 cells was imaged and analyzed. a, Example of cells in which most of the HSF1 foci rapidly dissolved after the addition of 1,6-hexandiol. The timescale of dissolution (20-30 seconds) is an order of magnitude faster than the timescale of cell membrane permeabilization (200-300 seconds). 1,6-hexanediol was applied one hour after addition of MG132 when most foci were liquid condensates (scalebar = 10 µm). b, Examples of cells showing that HSF1 foci are still visible after cell membrane permeabilization had occurred due to the addition of 1,6-hexandiol, which was applied 8 h after addition of MG132 when many foci were enlarged granular aggregates. See Fig. 4 for quantification and statistics and Source Data Extended Data Fig. 10 for details on cell numbers (scalebar = 10 µm). c, Fold induction of HSP90AA1 and inducible HSP70 genes (HSPA1B, HSPA6) using RT–qPCR from bulk cell populations after gradient doses of MG132 (2.5, 1.25, 0.62, 0.31 µM, median of 4 technical replicates). Blue bars = U2OS HSF1 wild type. Green bars = U2OS HSF1Δp mutant (both tagged with CFP). Source Data are provided in Source Data Extended Data Fig. 10.
Supplementary information
Supplementary Video 1
Example of cell death and foci dissolution events. U2OS HSF1-mVenus cells after 1 µM MG132 treatment – 15 min intervals, 16 hours length.
Supplementary Video 2
Examples of HSF1 foci fluid-like dynamics. U2OS FKBP-cHSF1-TQ2 post 500 nM Shield-1, 5 min intervals.
Supplementary Video 3
3D reconstruction of stacked confocal images of U2OS HSF1-mEGFP. Cells were treated with 2.5 µM MG132 and imaged between 15 min and 6 hours, every 5 min with 34 Z-stacks at 0.35 µm. Using Imaris (BITPLANE) Images were stacked, aligned and zoomed-in on one focus to show aggregation dynamics.
Supplementary Tables 1–6
Supplementary Table 1: List of antibodies. Supplementary Table 2: List of plasmids. Supplementary Table 3: Small molecule screening experimental details. Supplementary Table 4: List of compounds in the high-content imaging screening library. Supplementary Table 5: High-content imaging screen results. Supplementary Table 6: Subset of high-content imaging screen results for compounds of biological interest.
Source data
Source Data Fig. 1
Statistical source data
Source Data Fig. 2
Statistical source data
Source Data Fig. 3
Statistical source data
Source Data Fig. 4
Statistical source data
Extended Data Fig. 1
Statistical source data
Extended Data Fig. 2
Statistical source data
Extended Data Fig. 3
Statistical source data
Extended Data Fig. 3
Unprocessed blots
Extended Data Fig. 4
Statistical source data
Extended Data Fig. 5
Statistical source data
Extended Data Fig. 6
Statistical source data
Extended Data Fig. 7
Statistical source data
Extended Data Fig. 8
Statistical source data
Extended Data Fig. 9
Statistical source data
Extended Data Fig. 10
Statistical source data
Rights and permissions
About this article
Cite this article
Gaglia, G., Rashid, R., Yapp, C. et al. HSF1 phase transition mediates stress adaptation and cell fate decisions. Nat Cell Biol 22, 151–158 (2020). https://doi.org/10.1038/s41556-019-0458-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41556-019-0458-3
This article is cited by
-
A chaperone-like function of FUS ensures TAZ condensate dynamics and transcriptional activation
Nature Cell Biology (2024)
-
Temporal coordination of the transcription factor response to H2O2 stress
Nature Communications (2024)
-
Physicochemical investigations of clouding development and physicochemical properties of Triton X-100 and levofloxacin hemihydrate mixture: influence of sodium salts composition
Colloid and Polymer Science (2023)
-
H3K27ac mediated SS18/BAFs relocation regulates JUN induced pluripotent-somatic transition
Cell & Bioscience (2022)
-
Reversible phase separation of HSF1 is required for an acute transcriptional response during heat shock
Nature Cell Biology (2022)