Abstract
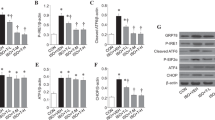
Exposure to sevoflurane and other inhalational anesthetics can induce cognitive impairment in elderly patients. Studies have indicated that methylene blue (MB) has beneficial effects on multiple neurodegenerative diseases and the mechanism involves mitochondrial function preservation. However, whether MB can attenuate the cognitive decline induced by sevoflurane in aged mice requires further investigation. Forty-five 18-month-old C57/BL mice were used to establish a model of sevoflurane-induced cognitive impairment in which the mice were exposed to 3% sevoflurane for 2 h. Mice in the MB group were intraperitoneally injected with MB at a dose of 5 mg/kg before sevoflurane inhalation. The Morris water maze test was used to evaluate the learning and memory performances. We also examined changes in mitochondrial morphology as well as the expression and interaction of related proteins in the aged hippocampus. Parkin, BCL2/adenovirus E1B 19 kDa protein-interacting protein 3 (BNIP3), mitochondrial dynamin-related protein 1 (Drp1), small ubiquitin-like modifier (SUMO2/3), SUMO-specific proteases 3 (SENP3), and ubiquitin‐like conjugating enzyme 9 expression in the mouse hippocampus was detected by western blotting, and SUMO2/3-Drp1 was examined by coimmunoprecipitation. Exposure to sevoflurane increased SENP3 expression and Drp1 deSUMOylation in the aged hippocampus and resulted in cognitive deficiency. MB attenuated sevoflurane-induced memory loss and mitochondrial fragmentation and decreased Drp1 deSUMOylation in the aged hippocampus. This neuroprotective effect provides a mechanistic explanation for how the SUMOylation status of Drp1 acts as a key switch in the cognitive dysfunction induced by sevoflurane.




Similar content being viewed by others
Abbreviations
- MB:
-
Methylene blue
- BNIP3:
-
BCL2/adenovirus E1B 19 kDa protein-interacting protein 3
- Drp1:
-
Mitochondrial dynamin-related protein 1
- MWM:
-
Morris water maze test
- SUMO2/3:
-
Small ubiquitin-like modifier
- SENP3:
-
SUMO-specific proteases 3
- UBC9:
-
Ubiquitin‐like conjugating enzyme 9
- Con:
-
Control group
- Sev:
-
Sevoflurane group
- Sev + MB:
-
Sevoflurane + methylene blue group
References
Geng YJ, Wu QH, Zhang RQ (2017) Effect of propofol, sevoflurane, and isoflurane on postoperative cognitive dysfunction following laparoscopic cholecystectomy in elderly patients: a randomized controlled trial. J Clin Anesth 38:165–171
Qiao Y, Feng H, Zhao T et al (2015) Postoperative cognitive dysfunction after inhalational anesthesia in elderly patients undergoing major surgery: the influence of anesthetic technique, cerebral injury and systemic inflammation. BMC Anesthesiol 15:154
Xu F, Armstrong R, Urrego D et al (2016) The mitochondrial division inhibitor Mdivi-1 rescues mammalian neurons from anesthetic-induced cytotoxicity. Mol Brain 9:35
Shan Y, Sun S, Yang F et al (2018) Dexmedetomidine protects the developing rat brain against the neurotoxicity wrought by sevoflurane: role of autophagy and Drp1-Bax signaling. Drug Des Devel Ther 12:3617–3624
Fonseca TB, Sánchez-Guerrero Á, Milosevic I et al (2019) Mitochondrial fission requires DRP1 but not dynamins. Nature 570(7761):E34–E42
Zuo W, Zhang S, Xia CY et al (2014) Mitochondria autophagy is induced after hypoxic/ischemic stress in a Drp1 dependent manner: the role of inhibition of Drp1 in ischemic brain damage. Neuropharmacology 86:103–115
Zuo W, Yang PF, Chen J (2016) Drp-1, a potential therapeutic target for brain ischaemic stroke. Br J Pharmacol 173:1665–1677
Yeh ET, Gong L, Kamitani T (2000) Ubiquitin-like proteins: new wines in new bottles. Gene 248:1–14
Gareau JR, Lima CD (2010) The SUMO pathway: emerging mechanisms that shape specificity, conjugation and recognition. Nat Rev Mol Cell Biol 11:861–871
Yeh ET (2009) SUMOylation and De-SUMOylation: wrestling with life's processes. J Biol Chem 284:8223–8227
Guo C, Wilkinson KA, Evans AJ (2017) SENP3-mediated deSUMOylation of Drp1 facilitates interaction with Mff to promote cell death. Sci Rep 7:43811
Gao L, Zhao Y, He J et al (2018) The desumoylating enzyme sentrin-specific protease 3 contributes to myocardial ischemia reperfusion injury. J Genet Genomics 45:125–135
An J, Zhou Y, Zhang M et al (2019) Exenatide alleviates mitochondrial dysfunction and cognitive impairment in the 5xFAD mouse model of Alzheimer's disease. Behav Brain Res 370:111932
Fernandez A, Meechan DW, Karpinski BA et al (2019) Mitochondrial dysfunction leads to cortical under-connectivity and cognitive impairment. Neuron 102:1127–42.e3
Gonzalez-Lima F, Barksdale BR, Rojas JC (2014) Mitochondrial respiration as a target for neuroprotection and cognitive enhancement. Biochem Pharmacol 88(4):584–593
Wainwright M, Crossley KB (2002) Methylene blue–a therapeutic dye for all seasons? J Chemother 14:431–443
Lin X, Wu BB, Xing QY et al (2015) Methylene blue, a potential therapeutic target for mitochondria dysfunction. Int Immunopharmacol 28:1106–1107
Tucker D, Lu Y, Zhang Q (2018) From mitochondrial function to neuroprotection-an emerging role for methylene blue. Mol Neurobiol. 55(6):5137–5153
Peng S, Zhang Y, Li GJ, Zhang DX et al (2012) The effect of sevoflurane on the expression of M1 acetylcholine receptor in the hippocampus and cognitive function of aged rats. Mol Cell Biochem. 361(1–2):229–233
Yan WJ, Wang DB, Ren DQ et al (2019) AMPKα1 overexpression improves postoperative cognitive dysfunction in aged rats through AMPK-Sirt1 and autophagy signaling. J Cell Biochem. 120:11633–11641
Fang F, Lin W, Ling X et al (2016) The hippocampal cyclin D1 expression is involved in postoperative cognitive dysfunction after sevoflurane exposure in aged mice. Life Sci 160:34–40
Xu F, Armstrong R, Urrego D et al (2016) The mitochondiral division inhibitor Mdivi-1 rescues mammalian neurons from anesthetic-induced cytotoxicity. Mol Brain 9:35
Vutskits L, Xie Z (2016) Lasting impact of general anaesthesia on the brain: mechanisms and relevance. Nat Rev Neurosci 17:705–717
Lu H, Liufu N, Dong Y et al (2017) Sevoflurane acts on ubiquitination-proteasome pathway to reduce postsynaptic density 95 protein levels in young mice. Anesthesiology 127:961–975
Rojas JC, Bruchey AK, Gonzalez-Lima F (2012) Neurometabolic mechanisms for memory enhancement and neuroprotection of methylene blue. Prog Neurobiol 96:32–45
Gureev AP, Syromyatnikov MY, Gorbacheva TM et al (2016) Methylene blue improves sensorimotor phenotype and decreases anxiety in parallel with activating brain mitochondria biogenesis in mid-age mice. Neurosci Res 113:19–27
Vorhees CV, Williams MT (2006) Morris water maze: procedures for assessing spatial and related forms of learning and memory. Nat Protoc 1:848–858
McBride HM, Neuspiel M, Wasiak S (2006) Mitochondria: more than just a powerhouse. Curr Biol 16:R551–560
Rikka S, Quinsay MN, Thomas RL et al (2011) Bnip3 impairs mitochondrial bioenergetics and stimulates mitochondrial turnover. Cell Death Differ 18:721–731
Wasiak S, Zunino R, McBride HM (2007) Bax/Bak promote sumoylation of DRP1 and its stable association with mitochondria during apoptotic cell death. J Cell Biol 177:439–450
Figueroa-Romero C, Iniguez-Lluhi JA, Stadler J et al (2009) SUMOylation of the mitochondrial fission protein Drp1 occurs at multiple nonconsensus sites within the B domain and is linked to its activity cycle. FASEB J 23:3917–3927
Guo C, Hildick KL, Luo J et al (2013) SENP3-mediated deSUMOylation of dynamin-related protein 1 promotes cell death following ischaemia. EMBO J 32:1514–1528
Hay RT (2007) SUMO-specific proteases: a twist in the tail. Trends Cell Biol 17:370–376
Gong L, Yeh ET (2006) Characterization of a family of nucleolar SUMO-specific proteases with preference for SUMO-2 or SUMO-3. J Biol Chem 281:15869–15877
Fanselow MS, Dong HW (2010) Are the dorsal and ventral hippocampus functionally distinct structures? Neuron 65:7–19
Acknowledgement
This research was supported by the National Natural Science Foundation of China (Grant Nos. 81801082 and 81771160) and the Hubei Provincial Natural Science Foundation (Grant No. 2019CFB102).
Author information
Authors and Affiliations
Contributions
FZ and PF helped carry out the animal experiments and western blotting. JC helped record experimental data. MC, QZ and TC helped analyze the data. CC (Chang Chen) and ZZZ (Zongze Zhang) helped design the experiments and write the manuscript.
Corresponding authors
Ethics declarations
Conflict of interest
No conflicts of interest exist in the submission of this manuscript.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Zheng, F., Fang, P., Chang, J. et al. Methylene Blue Protects Against Sevoflurane-Induced Cognitive Dysfunction by Suppressing Drp1 deSUMOylation in Aged Mice. Neurochem Res 45, 956–963 (2020). https://doi.org/10.1007/s11064-020-02976-6
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11064-020-02976-6