Abstract
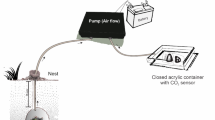
Leaf-cutter ant nests are biogeochemical hot spots where ants live and import vegetation to grow fungus. Metabolic activity and (in wet tropical forests) soil gas flux to the nest may result in high nest CO2 concentrations if not adequately ventilated. Wind-driven ventilation mitigates high CO2 concentrations in grasslands, but little is known about exchange for forest species faced with prolonged windless conditions. We studied Atta cephalotes nests located under dense canopy (leaf area index > 5) in a wet tropical rainforest in Costa Rica, where wind events are infrequent. We instrumented nests with thermocouples and flow-through CO2 sensing chambers. The results showed that CO2 concentrations exiting leaf-cutter ant nests follow a diel pattern with higher values at night. We developed an efflux model based on pressure differences that evaluated the observed CO2 diel pattern in terms of ventilation by (1) free convection (warm, less dense air rises out the nest more prominently at night) and (2) episodic wind-forced convection events providing occasional supplemental ventilation during daytime. Average greenhouse gas emissions were estimated through nest vents at about 78 kg CO2eq nest−1 year−1. At the ecosystem level, leaf-cutter ant nest vents accounted for 0.2% to 1% of total rainforest soil emissions. In wet, clayey tropical soils, leaf-cutter ant nests act as free convection-driven conduits for exporting CO2 and other greenhouse gases produced within the nest (fungus and ant respiration, refuse decay), and by roots and soil microbes surrounding the nest. This allows A. cephalotes nests to be ventilated without reliable wind conditions.




Similar content being viewed by others
Data availability
Data and code are available at https://doi.org/10.6084/m9.figshare.7789601. Detailed information about how to build the customized monitoring devices is available from the authors on request.
References
Aronson EL, Dierick D, Botthoff JK et al (2019) ENSO-influenced drought drives methane flux dynamics in a tropical wet forest soil. J Geophys Res Biogeosci 124:2267–2276. https://doi.org/10.1029/2018JG004832
Blanton CM, Ewel JJ (1985) Leaf-cutting ant herbivory in successional and agricultural tropical ecosystems. Ecology 66:861–869. https://doi.org/10.2307/1940548
Bollazzi M, Roces F (2007) To build or not to build: circulating dry air organizes collective building for climate control in the leaf-cutting ant Acromyrmex ambiguus. Anim Behav 74:1349–1355. https://doi.org/10.1016/j.anbehav.2007.02.021
Bollazzi M, Roces F (2010) Leaf-cutting ant workers (Acromyrmex heyeri) trade off nest thermoregulation for humidity control. J Ethol 28:399–403. https://doi.org/10.1007/s10164-010-0207-3
Bollazzi M, Forti LC, Roces F (2012) Ventilation of the giant nests of Atta leaf-cutting ants: does underground circulating air enter the fungus chambers? Insectes Soc 59:487–498
Costa AN, Vasconcelos HL, Vieira-Neto EHM, Bruna EM (2008) Do herbivores exert top-down effects in Neotropical savannas? Estimates of biomass consumption by leaf-cutter ants. J Veg Sci 19:849–854. https://doi.org/10.3170/2008-8-18461
Della Lucia T, Gandra LC, Guedes RN (2014) Managing leaf-cutting ants: peculiarities, trends and challenges. Pest Manag Sci 70:14–23
Fernandez-Bou AS, Dierick D, Swanson AC et al (2018) The role of the ecosystem engineer, the leaf-cutter ant Atta cephalotes, on soil CO2 dynamics in a wet tropical rainforest. J Geophys Res Biogeosci. https://doi.org/10.1029/2018JG004723
Forti LC, Moreira AA, da Silva Camargo R et al (2018) Nest architecture development of grass-cutting ants. Rev Bras Entomol 62:46–50. https://doi.org/10.1016/j.rbe.2017.10.002
Hashimoto S, Tanaka N, Suzuki M et al (2004) Soil respiration and soil CO2 concentration in a tropical forest, Thailand. J For Res 9:75–79. https://doi.org/10.1007/s10310-003-0046-y
Hölldobler B, Wilson EO (2009) The superorganism: the beauty, elegance, and strangeness of insect societies. W.W. Norton & Company, USA
Hudson TM, Turner BL, Herz H, Robinson JS (2009) Temporal patterns of nutrient availability around nests of leaf-cutting ants (Atta colombica) in secondary moist tropical forest. Soil Biol Biochem 41:1088–1093. https://doi.org/10.1016/j.soilbio.2009.02.014
Jacoby M (1939) A renovação do oxigênio no ninho da Atta sexdens L. Ministerio de Agricultura, Rio de Janeiro
Jonkman JCM (1976) Biology and ecology of the leaf cutting ant Atta vollenweideri Forel, 1893. Zeitschrift für Angewandte Entomologie 81:140–148. https://doi.org/10.1111/j.1439-0418.1976.tb04221.x
King H, Ocko S, Mahadevan L (2015) Termite mounds harness diurnal temperature oscillations for ventilation. PNAS 112:11589–11593. https://doi.org/10.1073/pnas.1423242112
Kirschke S, Bousquet P, Ciais P et al (2013) Three decades of global methane sources and sinks. Nat Geosci 6:813–823. https://doi.org/10.1038/ngeo1955
Kleber M, Schwendenmann L, Veldkamp E et al (2007) Halloysite versus gibbsite: silicon cycling as a pedogenetic process in two lowland neotropical rain forest soils of La Selva, Costa Rica. Geoderma 138:1–11. https://doi.org/10.1016/j.geoderma.2006.10.004
Kleineidam C, Roces F (2000) Carbon dioxide concentrations and nest ventilation in nests of the leaf-cutting ant Atta vollenweideri. Insect Soc 47:241–248. https://doi.org/10.1007/PL00001710
Kleineidam C, Ernst R, Roces F (2001) Wind-induced ventilation of the giant nests of the leaf-cutting ant Atta vollenweideri. Naturwissenschaften 88:301–305. https://doi.org/10.1007/s001140100235
Meyer ST, Leal IR, Wirth R (2009) Persisting hyper-abundance of leaf-cutting ants (Atta spp.) at the edge of an old Atlantic forest fragment. Biotropica 41:711–716
Moreira A, Forti LC, Andrade AP et al (2004) Nest architecture of Atta laevigata (F. Smith, 1858) (Hymenoptera: Formicidae). Stud Neotrop Fauna Environ 39:109–116. https://doi.org/10.1080/01650520412331333756
Nauer PA, Hutley LB, Arndt SK (2018) Termite mounds mitigate half of termite methane emissions. PNAS 115:13306–13311. https://doi.org/10.1073/pnas.1809790115
Ocko SA, King H, Andreen D et al (2017) Solar-powered ventilation of African termite mounds. J Exp Biol 220:3260–3269. https://doi.org/10.1242/jeb.160895
Oertel C, Matschullat J, Zurba K et al (2016) Greenhouse gas emissions from soils—a review. Chem Erde 76:327–352. https://doi.org/10.1016/j.chemer.2016.04.002
Ohashi M, Finér L, Domisch T et al (2005) CO2 efflux from a red wood ant mound in a boreal forest. Agric For Meteorol 130:131–136. https://doi.org/10.1016/j.agrformet.2005.03.002
Perfecto I, Vandermeer J (1993) Distribution and turnover rate of a population of Atta cephalotes in a tropical rain forest in Costa Rica. Biotropica 25:316–321. https://doi.org/10.2307/2388789
Peterson EW, Hennessey JP (1978) On the use of power laws for estimates of wind power potential. J Appl Meteor 17:390–394. https://doi.org/10.1175/1520-0450(1978)017%3c0390:OTUOPL%3e2.0.CO;2
Pinto-Tomás AA, Anderson MA, Suen G et al (2009) Symbiotic nitrogen fixation in the fungus gardens of leaf-cutter ants. Science 326:1120–1123. https://doi.org/10.1126/science.1173036
Powell RJ, Stradling DJ (1986) Factors influencing the growth of Attamyces bromatificus, a symbiont of attine ants. Trans Br Mycol Soc 87:205–213. https://doi.org/10.1016/S0007-1536(86)80022-5
Quinlan RJ, Cherrett JM (1978) Aspects of the symbiosis of the leaf-cutting ant Acromyrmex octospinosus (Reich) and its food fungus. Ecol Entomol 3:221–230. https://doi.org/10.1111/j.1365-2311.1978.tb00922.x
R Core Team (2017) R: a language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria
Risch A, Schuetz M, Jurgensen MF et al (2005) CO2 emissions from red wood ant (Formica rufa group) mounds: seasonal and diurnal patterns related to air temperature. Ann Zool Fenn 42:283–290
Römer D, Bollazzi M, Roces F (2017) Carbon dioxide sensing in an obligate insect-fungus symbiosis: CO2 preferences of leaf-cutting ants to rear their mutualistic fungus. PLoS ONE 12:e0174597. https://doi.org/10.1371/journal.pone.0174597
Sanderson MG (1996) Biomass of termites and their emissions of methane and carbon dioxide: a global database. Glob Biogeochem Cycles 10:543–557. https://doi.org/10.1029/96GB01893
Sanford Jr RL, Paaby P, Luvall JC, Phillips E (1994) Climate, geomorphology, and aquatic systems. In: McDade LA, Bawa KS, Hespenheide H, Hartshorn GS (eds) La Selva: ecology and natural history of a neotropical rain forest, pp 19–33
Schultz TR, Brady SG (2008) Major evolutionary transitions in ant agriculture. PNAS 105:5435–5440. https://doi.org/10.1073/pnas.0711024105
Schwendenmann L, Veldkamp E (2006) Long-term CO2 production from deeply weathered soils of a tropical rain forest: evidence for a potential positive feedback to climate warming. Glob Change Biol 12:1878–1893
Siqueira F, Ribeiro-Neto J, Tabarelli M, Andersen A, Wirth R, Leal I (2017) Leaf-cutting ant populations profit from human disturbances in tropical dry forest in Brazil. J Trop Ecol 33(5):337–344. https://doi.org/10.1017/S0266467417000311
Soper FM, Sullivan BW, Nasto MK et al (2018) Remotely sensed canopy nitrogen correlates with nitrous oxide emissions in a lowland tropical rainforest. Ecology 99:2080–2089. https://doi.org/10.1002/ecy.2434
Soper FM, Sullivan BW, Osborne BB et al (2019) Leaf-cutter ants engineer large nitrous oxide hot spots in tropical forests. Proc R Soc B Biol Sci 286:20182504. https://doi.org/10.1098/rspb.2018.2504
Sousa-Souto L, de Santos DCJ, Ambrogi BG et al (2012) Increased CO2 emission and organic matter decomposition by leaf-cutting ant nests in a coastal environment. Soil Biol Biochem 44:21–25. https://doi.org/10.1016/j.soilbio.2011.09.008
Stahel G, Geijskes DC (1940) Observations about temperature and moisture in Atta-nests. Rev Entomol 11:775–776
Swanson AC, Schwendenmann L, Allen MF et al (2019) Welcome to the Atta world: a framework for understanding the effects of leaf cutter ants on ecosystem functions. Funct Ecol 33:1386–1399. https://doi.org/10.1111/1365-2435.13319
Urbas P, Araújo MV, Leal IR, Wirth R (2007) Cutting more from cut forests: edge effects on foraging and herbivory of leaf-cutting ants in Brazil. Biotropica 39:489–495. https://doi.org/10.1111/j.1744-7429.2007.00285.x
Verchot LV, Moutinho PR, Davidson EA (2003) Leaf-cutting ant (Atta Sexdens) and nutrient cycling: deep soil inorganic nitrogen stocks, mineralization, and nitrification in Eastern Amazonia. Soil Biol Biochem 35:1219–1222. https://doi.org/10.1016/S0038-0717(03)00183-4
Viana LR, Santos JC, Arruda LJ et al (2004) Foraging patterns of the leaf-cutter ant Atta laevigata (Smith) (Myrmicinae: Attini) in an area of cerrado vegetation. Neotrop Entomol 33:391–393. https://doi.org/10.1590/S1519-566X2004000300019
Wang Q, Song W, Zhang J, Lo S (2018) Bi-directional movement characteristics of Camponotus japonicus ants during nest relocation. J Exp Biol. https://doi.org/10.1242/jeb.181669
Weber NA (1966) Fungus-growing ants. Science 153:587–604
Wilson EO (1980) Caste and division of labor in leaf-cutter ants (Hymenoptera: Formicidae: Atta). Behav Ecol Sociobiol 7:143–156. https://doi.org/10.1007/BF00299520
Wirth R, Beyschlag W, Ryel RJ, Hölldobler B (1997) Annual foraging of the leaf-cutting ant Atta colombica in a semideciduous rain forest in Panama. J Trop Ecol 13:741–757. https://doi.org/10.1017/S0266467400010907
Wirth R, Herz H, Ryel RJ et al (2003) Herbivory of leaf-cutting ants: a case study on Atta colombica in the tropical rainforest of Panama. Springer Science & Business Media, New York
Wirth R, Meyer ST, Almeida WR et al (2007) Increasing densities of leaf-cutting ants (Atta spp.) with proximity to the edge in a Brazilian Atlantic forest. J Trop Ecol 23:501–505. https://doi.org/10.1017/S0266467407004221
Acknowledgements
We thank Adrián Pinto-Tomás, Allan Artavia-León, and the rest of the Pinto-Tomás laboratory at the Universidad de Costa Rica for allowing us to participate in their nest excavations. We also thank Samantha Young, Spencer McDermont, and Timothy Barahona for their help building the electronic devices; our colleagues of the “Atta team” Amanda C. Swanson, Ana Grace Alvarado, Catalina Murillo, Deo Lachman, Emma Aronson, Jane Zelikova, Jon Botthoff, Luitgard Schwendenmann, Michael F. Allen, Odemaris Carrasquillo‐Quintana, Philip Rundel, Shaquetta Johnson, Steven Oberbauer, and Yorelyz Rodríguez‐Reyes for all our joint efforts; Stephen C. Hart, Carlos de la Rosa, and the staff of La Selva Biological Station for their logistical support.
Funding
This work was conducted with permits granted by the “Comisión Institucional de Biodiversidad” (Institutional Biodiversity Committee, University of Costa Rica; resolution VI-8315-2014) and authorized by La Selva Biological Station, and it was supported by the National Science Foundation under Collaborative Awards DEB-1442537, 1442568, 1442622, 1442714, 1624623, and 1624658.
Author information
Authors and Affiliations
Contributions
ASFB led the work as part of his doctoral dissertation, and TCH and DD participated with essential contributions to the experiment conceptualization, custom devices design and fabrication, model development, and revising the manuscript. DD and ASFB conducted the field measurements.
Corresponding author
Additional information
Communicated by Stefan Scheu.
We present a mass balance model to describe CO2 fluxes from massive nests of leaf-cutter ants and designed flow-through chambers to continuously monitor them. Our study revealed a clear diel CO2 emission pattern driven by free convection and sporadic wind-forced convection. At the ecosystem scale, nests are responsible for up to 1% of the total forest soil greenhouse gas emissions, which is impressive for a single ant species that thrives under changing climate and fragmented landscapes.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Fernandez-Bou, A.S., Dierick, D. & Harmon, T.C. Diel pattern driven by free convection controls leaf-cutter ant nest ventilation and greenhouse gas emissions in a Neotropical rain forest. Oecologia 192, 591–601 (2020). https://doi.org/10.1007/s00442-020-04602-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00442-020-04602-2