Abstract
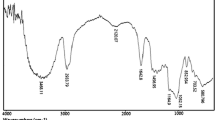
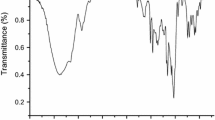
In the present study, β-cyclodextrin-based nanosponges were prepared by reaction of β-cyclodextrin with dicarbonyl imidazole as a crosslinker agent. The synthesized polycarbonate nanosponge was characterized by FT-IR spectroscopy, thermogravimetric analysis, scanning electron microscopy and Brunauer–Emmett–Teller. The catalytic activity of β-cyclodextrin-based nanosponges was successfully tested in the one-pot three-component condensation of kojic acid, aromatic aldehydes and various aromatic amines. The superiority of this new strategy is short reaction times, high yields and utilizing of biodegradable and nontoxic nanocatalyst.










Similar content being viewed by others
References
S. Torne, S. Darandale, P. Vavia, F. Trotta, R. Cavalli, Pharm. Dev. Technol. 18, 619 (2013)
M.J. Nasab, A.R. Kiasat, R. Zarasvandi, React. Kinet. Mech. Catal. 124, 767 (2018)
S. Sadjadi, M.M. Heravi, M. Daraie, Res. Chem. Intermed. 43, 843 (2017)
S. Khajeh Dangolani, S. Sharifat, F. Panahi, A. Khalafi-Nezhad, Inorg. Chim. Acta 494, 256 (2019)
D. Zhao, L. Zhao, C.-S. Zhu, W.-Q. Huang, J.-L. Hu, J. Incl. Phenom. Macrocycl Chem. 63, 195 (2009)
G. Tejashri, B. Amrita, J. Darshana, Acta Pharm. 63, 335 (2013)
F. Trotta, R. Cavalli, Compos. Interfaces 16, 39 (2009)
J. Li, Q. Jiang, P. Deng, Q. Chen, M. Yu, J. Shang, W. Li, J. Pharm. Pharmacol. 69, 663 (2017)
S. Selvamuthukumar, S. Anandam, K. Krishnamoorthy, M. Rajappan, J. Pharm. Pharm. Sci. 15, 103 (2012)
M. Shringirishi, S.K. Prajapati, A. Mahor, S. Alok, P. Yadav, A. Verma, Asian Pac. J. Trop. Dis. 4, S519 (2014)
S. Sadjadi, M.M. Heravi, M. Raja, Carbohydr. Polym. 185, 48 (2018)
S. Sadjadi, M.M. Heravi, M. Malmir, Appl. Organomet. Chem. 32, e4286 (2018)
D. Zhang, P. Lv, C. Zhou, Y. Zhao, X. Liao, B. Yang, Mater. Sci. Eng. C. 1, 872 (2018)
S. Sadjadi, M. Malmir, M.M. Heravi, M. Raja, Int. J. Biol. Macromol. 128, 638 (2019)
W. Liang, M. Zhao, X. Wei, Z. Yan, W. Wu, F. Caldera, F. Trotta, Y. Inoue, D. Su, Z. Zhong, RSC Adv. 7, 17184 (2017)
X. Wei, W. Liang, W. Wu, C. Yang, F. Trotta, F. Caldera, A. Mele, T. Nishimoto, Y. Inoue, Org. Biomol. Chem. 13, 2905 (2015)
W. Liang, C. Yang, D. Zhou, H. Haneoka, M. Nishijima, G. Fukuhara, T. Mori, F. Castiglione, A. Mele, F. Caldera, Chem. Commun. 49, 3510 (2013)
R. Kardooni, A.R. Kiasat, J. Taiwan Inst. Chem. Eng. 87, 241 (2018)
R. Kardooni, A.R. Kiasat, H. Motamedi, J. Taiwan Inst. Chem. Eng. 81, 373 (2017)
G. Brahmachari, S. Das, RSC Adv. 4, 7380 (2014)
H.S. Rho, S.M. Ahn, D.S. Yoo, M.K. Kim, D.H. Cho, J.Y. Cho, Bioorg. Med. Chem. Lett. 20, 6569 (2010)
M. Zirak, B. Eftekhari-Sis, Turk. J. Chem. 39, 439 (2015)
A. Maleki, R. Firouzi-Haji, Z. Hajizadeh, Int. J. Biol. Macromol. 116, 320 (2018)
R. Baharfar, H. Alinezhad, R. Azimi, Res. Chem. Intermed. 41, 8637 (2015)
P.R. Likhar, G.N. Reddy, M.R. Reddy, Res. Chem. Intermed. 42, 5983 (2016)
A. Rahmati, Z. Khalesi, T. Kenarkoohi, Comb. Chem. High Throughput Screening 17, 132 (2014)
Z. Lasemi, B. Sadeghi, F. Amiri Tavasoli, Iran. J. Catal. 6, 51 (2016)
R. Teimuri-Mofrad, A. Shahrisa, M. Gholamhosseini-Nazari, N. Arsalani, Res. Chem. Intermed. 42, 3425 (2016)
B.S. Reddy, M.R. Reddy, C. Madan, K.P. Kumar, M.S. Rao, Bioorg. Med. Chem. Lett. 20, 7507 (2010)
H.-Y. Zhang, X.-P. Hao, L.-P. Mo, S.-S. Liu, W.-B. Zhang, Z.-H. Zhang, New J. Chem. 41, 7108 (2017)
E.A. Kataev, M. Ramana Reddy, G. Niranjan Reddy, V.H. Reddy, C. Suresh Reddy, B.V. Subba Reddy, New J. Chem. 40, 1693 (2016)
A.R. Kiasat, S. Nazari, J. Davarpanah, J. Serb. Chem. Soc. 79, 401 (2014)
N.E. Sabzi, A. Kiasat, Catal. Lett. 148, 2654 (2018)
S. Sadjadi, M.M. Heravi, M. Raja, Int. J. Biol. Macromol. 122, 228 (2019)
R. Shelat, S. Chandra, A. Khanna, Int. J. Biol. Macromol. 110, 357 (2018)
A.R. Kiasat, S. Nazari, Catal. Sci. Technol 2, 1056 (2012)
H.-J. Niu, L. Zhang, J.-J. Feng, Q.-L. Zhang, H. Huang, A.-J. Wang, J. Colloid Interface Sci. 552, 744 (2019)
Y. Chen, A.-J. Wang, P.-X. Yuan, X. Luo, Y. Xue, J.-J. Feng, Biosens. Bioelectron. 132, 294 (2019)
Y.-C. Shi, J.-J. Feng, X.-X. Lin, L. Zhang, J. Yuan, Q.-L. Zhang, A.-J. Wang, Electrochim. Acta 293, 504 (2019)
H.-J. Niu, A.-J. Wang, L. Zhang, J.-J. Guo, J.-J. Feng, Mater. Chem. Front. 3, 1849 (2019)
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Kardooni, R., Kiasat, A.R. & Eskandari Sabzi, N. Hyper-cross-linked β-cyclodextrin nanosponge: a three-dimensional, porous and biodegradable catalyst in the one-pot synthesis of kojic acid-based heterocyclic compounds. Res Chem Intermed 46, 1857–1868 (2020). https://doi.org/10.1007/s11164-019-04067-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11164-019-04067-w