Abstract
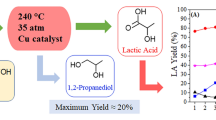
Glycerol conversion to lactic acid (LA) was investigated in aqueous alkali over eight unsupported copper compounds (CuBr2, CuBr, CuCl2, CuCl, CuF2, Cu(NO3)2,CuO, and Cu2O) for studying the effects of anion and valence. Powder X-ray diffraction and scanning electron microscopy measurements indicated that these copper compounds were reduced to metallic copper with different morphologies. Divalent copper compounds exhibited much better performances than the corresponding univalent species, ascribed to their greater reduction heat and higher local reaction temperature. Divalent copper species activity, ionic radius, and the reported reduction potential decreased in the same order: bromide > chloride > floride ≫ nitrate. With increasing reaction temperature, catalyst amount, NaOH concentration and reaction time, glycerol conversion, and LA selectivity increased (due to by-product conversions to LA). Kinetic studies indicated that glycerol disappearance rate was first-order with respect to its concentration. CuBr2 had greater activation energy and therefore exhibited better performance than CuO when reaction temperature was greater than 155 °C. At 185 °C, CuBr2 reached 95.7% lactic acid yield and 98.65% glycerol conversion.






Similar content being viewed by others
References
Razali,N. and Abdullah, A.Z. (2017) Production of lactic acid from glycerol via chemical conversion using solid catalyst: A review. Applied Catalysis A: General, 543, 234–246.
Johnson, D. T., & Taconi, K. A. (2007). The Glycerin glut: Options for the value- added conversion of crude glycerol resulting from biodiesel production. Environmental Progress, 26, 338–348.
Fernado, S., Adhikari, S., Chandrapa, l C. Murali, N. (2006) Biorefineries: Current status, challenges, and future direction. Energy Fuels, 20,1727–1737.
Zhou, C. H., Beltramini, J. N., Fan, Y. X., & Lu, G. Q. (2008). Chemoselective catalytic conversion of glycerol as a biorenewable source to valuable commodity chemicals. Chemical Society Reviews, 37(3), 527–549.
Pagliiaro, M., Ciriminna, R., Kimura, H., Rossi, M., & Pina, C. D. (2007). From glycerol to value- added products. Angewandte Chemie, International Edition, 46, 4434–4440.
Li, K. T., & Wang, C. K. (2012). Esterification of lactic acid over TiO2–Al2O3 catalysts. Applied Catalysis A: General, 433-434, 275–279.
Li, K. T., Wang, C. K., Wang, I., & Wang, C. M. (2011). Esterification of lactic acid over TiO2-ZrO2 catalysts. Applied Catalysis A: General, 392, 180–183.
Li, K. T., Tsai, L. D., Wu, C. H., & Wang, I. K. (2013). Lactic acid esterification on titania-silica binary oxides. Industrial and Engineering Chemistry Research, 52, 4734–4739.
Ghaffar, T., Irshad, M., Anwar, Z., Aqil, T., Zulifqar, Z., Tariq, A., Kamran, M., Ehsan, N., & Mehmood, S. (2014). Recent trends in lactic acid biotechnology: A brief review on production to purification. Journal Radiant Research Application Science, 7, 222–229.
Palacio, R., Torres, S., Royer, S., Mamede, A. S., Lopez, D., & Hernandez, D. (2018). CuO/CeO2 catalysts for glycerol selective conversion to lactic acid. Dalton Transactions, 47, 4572–4582.
Li, K. T., Li, J. Y., & Li, H. H. (2017). Conversion of glycerol to lactic acid over Cu-Zn-Al and Cu-Cr catalysts in alkaline solution. Journal Taiwan Institute Chemical Engineers, 79, 74–79.
Roy, D., & Subramaniam, B. (2011). Cu-based catalysts show low temperature for glycerol conversion to lactic acid. ACS Catalysis, 1, 548–551.
Yang, G. Y., Ke, Y. H., Ren, H. F., Liu, C. L., Yang, R. Z., & Dong, W. S. (2016). The conversion of glycerol to lactic acid catalyzed by ZrO2-supported CuO catalysts. Chemical Engineering Journal, 283, 759–767.
Yin, H., Zhang, C., Yin, H., Gao, D., Shen, L., & Wang, A. (2016). Hydrothermal conversion of glycerol to lactic acid catalyzed by Cu/hydroxyapatite, Cu/MgO, and Cu/ZrO2 and reaction kinetics. Chemical Engineering Journal, 288, 332–343.
Shen, L., Yin, H., Yin, H., Liu,S., Wang, A. (2017) Conversion of glycerol to lactic acid catalyzed by different-sized Cu2O nanoparticles in NaOH aqueous solution. Journal of Nanoscience and Nanotechnology, 17,780–787.
Allen, S. E., Walvoord, R. R., Padilla-Salinas, R., & Kozlowski, M. C. (2013). Aerobic copper-catalyzed organic reactions. Chemical Reviews, 113(8), 6234–6458.
Li, K. T., & Tsai, I. T. (1996). 2,6-Dimethylphenol polymerization with cuprous chloride and n-butylamine catalyst system: Effect of Nal addition. Journal of Polymer Science A: Polymer Chemistry, 34, 3213–3219.
Li, K. T., & Tsai, I. T. (1999). Effect of iodine additives on the polymerization of 2,6-dimethylphenol with copper bromide/ n-butylamine catalyst. Journal Chinese Chemical Engineers, 30, 1–9.
Li, K. T., & Shieh, D. C. (1994). Polymerization of 2, 6-dimethylphenol with mixed-ligand copper complexes. Industrial and Engineering Chemistry Research, 33, 1107–1112.
Teichert,J., Doert, T. and Ruck, M. (2018) Mechanisms of the polyol reduction of copper(ii) salts depending on the anion type and diol chain length. Dalton Transactions, 47, 14085–14093.
Kim, J. Y., Rodriguez, J. A., Hanson, J. C., Frenkel, A.I, & Lee, P. L. (2003). Reduction of CuO and Cu2O with H: H embedding and kinetic effects in the formation of suboxides. Journal of American Chemical Society, 125, 10684–10692.
Hinrichsen, K., Kochloefl, K. and Muhler, M. (2008). Water Gas Shift and COS Removal. In Handbook of Heterogeneous Catalysis (Ertl, G., Knozinger, H., Schuth, F., Weitkamp, J., eds.) Wiley-VCH , Weinheim, p.2911.
Atomic and Ionic Radius. Available from: https://chem.libretexts.org/ Courses/Bellarmine_University/BU%3A_Chem_103_(Christianson)/Phase_3%3A_Atoms_and_Molecules_-_the_Underlying_Reality/8%3A_Periodic_Trends_in_Elements_and_Compounds/8.2%3A_Atomic_and_Ionic_Radius. Accessed September 20, 2019.
Isse, A. A., Lin, C. Y., Coote, M. L., & Gennaro, A. (2011). Estimation of standard reduction potentials of halogen atoms and alkyl halides. The Journal of Physical Chemistry, 115(4), 678–684.
Standard Electrode Potential (data page). Available from: http://en.wikipedia.org /wiki/ Standard_electrode_potential_(data_page). Accessed September 20, 2019.
Ukpong, E. J., & Prasad, J. (2007). Effect of conterion, solvent and ligand substituents on the reduction potentials of copper (II) complexes of varioius imidazoles in some nonaqueous solvents. Journal of Pure and Applied Sciences, 13, 533–536.
Fogler, H. S. (2006). Elements of chemical reaction engineering (4th ed.). Upper Saddle River, NJ: Prentice Hall Chap. 5.
Acknowledgments
The authors are grateful to Chia-Hsin Hsu who obtained the H-NMR data.
Funding
The authors received financial support from ROC Ministry of Science and Technology (MOST 104-2221-E-029-023).
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Li, KT., Li, HH. Glycerol Conversion to Lactic Acid with Unsupported Copper Salts and Bulk Cupric Oxide in Aqueous Alkali Media. Appl Biochem Biotechnol 191, 125–134 (2020). https://doi.org/10.1007/s12010-020-03237-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12010-020-03237-6