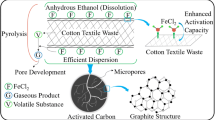
Abstract
Char-based adsorbents (char-FeCl3, char-FeCl2, and char-FeCit) derived from cotton textile waste (CTW) were synthesized by one-step low-temperature pyrolysis approach with different iron salts. The properties of the samples were conducted by BET, SEM, EDS, XRD, XPS, TEM, and FTIR. The results suggested that the surface areas of char-FeCl3 and char-FeCl2 were higher than those of char-FeCit. The presence of Fe2O3 as well as pyrolysis gas (HCl (g) and H2O (g)) could catalyze the formation of porosity. Meanwhile, FeCl3 showed the strongest catalysis effect to decompose cellulose to produce char. The pyrolysis process analysis was investigated by means of thermogravimetry-DSC. FeCl3 and FeCl2 could accelerate the breakage of cellulose structure whereas FeC6H5O7 was not beneficial to form char at low temperature as the incomplete decomposition of citrate. The adsorption property of Cr(VI) for the chars was evaluated. Adsorption processes were fitted well with the Freundlich model, and char-FeCl3 presented the best adsorptive capacity (70.39 mg/g). Thus, this low-temperature pyrolysis method was economical and technologically simplified as well as efficient adsorption capacity of Cr(VI) removal.

Graphical abstract











Similar content being viewed by others
References
Acharyaa J, Sahub JN, Mohantyc CR, Meikap BC (2009) Removal of lead(II) from wastewater by activated carbon developed from tamarind wood by zinc chloride activation. Chem Eng J 149:249–262. https://doi.org/10.1016/j.cej.2008.10.029
Akama Y, Sali A (2002) Extraction mechanism of Cr(VI) on the aqueous two-phase system of tetrabutylammonium bromide and (NH4)2SO4 mixture. Talanta 57:681–686
Angin D (2014) Production and characterization of activated carbon from sour cherry stones by zinc chloride. Fuel 115:804–811. https://doi.org/10.1016/j.fuel.2013.04.060
Bai J, Wang Q, Jiao G (2012) Study on the pore structure of oil shale during low-temperature pyrolysis. Energy Procedia 17:1689–1696. https://doi.org/10.1016/j.egypro.2012.02.299
Balachandran M (2014) Bio-processing of indian coals by microorganisms: an investigation. J Environ Res Dev 9:209–215
Bao S et al (2019) In-situ porous nano-Fe3O4/C composites derived from citrate precursor as anode materials for lithium-ion batteries. Mater Chem Phys 225:379–383. https://doi.org/10.1016/j.matchemphys.2018.12.072
Bedia J, Belver C, Ponce S, Rodriguez J, Rodriguez JJ (2018) Adsorption of antipyrine by activated carbons from FeCl3-activation of Tara gum. Chem Eng J 333:58–65. https://doi.org/10.1016/j.cej.2017.09.161
Cazetta AL, Pezoti O, Bedin KC, Silva TL, Paesano Junior A, Asefa T, Almeida VC (2016) Magnetic activated carbon derived from biomass waste by concurrent synthesis: efficient adsorbent for toxic dyes. ACS Sustain Chem Eng 4:1058–1068. https://doi.org/10.1021/acssuschemeng.5b01141
Chiu K-L, Ng DHL (2012) Synthesis and characterization of cotton-made activated carbon fiber and its adsorption of methylene blue in water treatment. Biomass Bioenergy 46:102–110. https://doi.org/10.1016/j.biombioe.2012.09.023
Cuhadaroglu D, Uygun OA (2008) Production and characterization of activated carbon from a biuminous coal by chemical activation. Afr J Biotechnol 7:3703–3710. https://doi.org/10.1016/j.protis.2008.04.002
Daud WMAW, Ali WSW (2004) Comparison on pore development of activated carbon produced from palm shell and coconut shell. Bioresour Technol 93:63–69. https://doi.org/10.1016/j.biortech.2003.09.015
Demiral H, Demiral İ, Tümsek F, Karabacakoğlu B (2008) Adsorption of chromium(VI) from aqueous solution by activated carbon derived from olive bagasse and applicability of different adsorption models. Chem Eng J 144:188–196. https://doi.org/10.1016/j.cej.2008.01.020
Dong X, Lu C, Zhou P, Zhang S, Wang L, Li D (2015) Polyacrylonitrile/lignin sulfonate blend fiber for low-cost carbon fiber. RSC Adv 5:42259–42265. https://doi.org/10.1039/c5ra01241d
Duranoglu D, Trochimczukb AW, Bekera U (2012) Kinetics and thermodynamics of hexavalent chromium adsorption onto activated carbon derived from acrylonitrile-divinylbenzene copolymer. Chem Eng J 187:193–202. https://doi.org/10.1016/j.cej.2012.01.120
Dutta S, Bhattacharyya A, Ganguly A, Gupta S, Basu S (2011) Application of response surface methodology for preparation of low-cost adsorbent from citrus fruit peel and for removal of methylene blue. Desalination 275:26–36. https://doi.org/10.1016/j.desal.2011.02.057
Enniya I, Rghioui L, Jourani A (2018) Adsorption of hexavalent chromium in aqueous solution on activated carbon prepared from apple peels. Sustain Chem Pharm 7:9–16. https://doi.org/10.1016/j.scp.2017.11.003
Fu KF et al (2014) Physicochemical and adsorptive properties of activated carbons from Arundo donax Linn utilizing different iron salts as activating agents. J Taiwan Inst Chem Eng 45:3007–3015. https://doi.org/10.1016/j.jtice.2014.08.026
Gong X, Guo Z, Wang Z, Gong X, Guo Z, Wang Z (2009) Variation of char structure during anthracite pyrolysis catalyzed by Fe2O3 and its influence on char combustion reactivity. Energy Fuel 23:4547–4552
González JF, Román S, Encinar JM, Martínez G (2009) Pyrolysis of various biomass residues and char utilization for the production of activated carbons. J Anal Appl Pyrolysis 85:134–141. https://doi.org/10.1016/j.jaap.2008.11.035
Goswami M, Borah L, Mahanta D, Phukan P (2014) Equilibrium modeling, kinetic and thermodynamic studies on the adsorption of Cr(VI) using activated carbon derived from matured tea leaves. J Porous Mater 21:1025–1034. https://doi.org/10.1007/s10934-014-9852-1
Hamid SBA, Teh SJ, Lim YS (2015) Catalytic hydrothermal upgrading of α-cellulose using iron salts as a Lewis acid. BioResources 10:5974–5986. https://doi.org/10.15376/biores.10.3.5974-5986
Hanoğlu A, Cay A, Yanık J (2019) Production of biochars from textile fibres through torrefaction and their characterisation. Energy 166:664–673. https://doi.org/10.1016/j.energy.2018.10.123
He J et al (2016) Preparation of highly porous carbon from sustainable α-cellulose for superior removal performance of tetracycline and sulfamethazine from water. RSC Adv 6:28023–28033. https://doi.org/10.1039/c6ra00277c
Kanungo SB, Mishr SK (1996) Thermal dehydration and decomposition of FeCl3·xH2O. J Therm Anal 46:1487–1500
Kumar R, Bishnoi NR, Garima, Bishnoi K (2008) Biosorption of chromium(VI) from aqueous solution and electroplating wastewater using fungal biomass. Chem Eng J 135:202–208. https://doi.org/10.1016/j.cej.2007.03.004
Lee SM, Choi WJ, Hwang K, Kim J-H, Lee J-Y (2014) Effect of catalyst concentration and reaction time on one-step synthesized hypercrosslinked polyxylene. Macromol Res 22:481–486. https://doi.org/10.1007/s13233-014-2065-2
Li W-H, Yue Q-Y, Gao B-Y, Wang X-J, Qi Y-F, Zhao Y-Q, Li Y-J (2011) Preparation of sludge-based activated carbon made from paper mill sewage sludge. Desalination 278:179–185. https://doi.org/10.1016/j.desal.2011.05.020
Li J, Zhang X, Zhang M, Xiu H, He H (2015) Ultrasonic enhance acid hydrolysis selectivity of cellulose with HCl-FeCl3 as catalyst. Carbohydr Polym 117:917–922. https://doi.org/10.1016/j.carbpol.2014.10.028
Li B et al (2019) Facile modification of activated carbon with highly dispersed nano-sized α-Fe2O3 for enhanced removal of hexavalent chromium from aqueous solutions. Chemosphere 224:220–227. https://doi.org/10.1016/j.chemosphere.2019.02.121
Liang H, Song B, Peng P, Jiao G, Yan X, She D (2019) Preparation of three-dimensional honeycomb carbon materials and their adsorption of Cr(VI). Chem Eng J 367:9–16. https://doi.org/10.1016/j.cej.2019.02.121
Lin SH, Kiang CD (2003) Chromic acid recovery from waste acid solution by an ion exchange. Chem Eng J 92:193–199. https://doi.org/10.1016/S1385-8947(02)00140-7
Liu L, Sun J, Cai C, Wang S, Pei H, Zhang J (2009) Corn stover pretreatment by inorganic salts and its effects on hemicellulose and cellulose degradation. Bioresour Technol 100:5865–5871. https://doi.org/10.1016/j.biortech.2009.06.048
Liu W, Zhang J, Zhang C, Ren L (2012) Preparation and evaluation of activated carbon-based iron-containing adsorbents for enhanced Cr(VI) removal: mechanism study. Chem Eng J 189–190:295–302. https://doi.org/10.1016/j.cej.2012.02.082
Liu WJ, Tian K, He YR, Jiang H, Yu HQ (2014) High-yield harvest of nanofibers/mesoporous carbon composite by pyrolysis of waste biomass and its application for high durability electrochemical energy storage. Environ Sci Technol 48:13951–13959. https://doi.org/10.1021/es504184c
Louvain N et al (2013) One-shot versus stepwise gas–solid synthesis of iron trifluoride: investigation of pure molecular F2 fluorination of chloride precursors. CrystEngComm 15:3664–3671. https://doi.org/10.1039/c3ce27033e
Luo Y, Street J, Steele P, Entsminger E, Guda V (2016) Activated carbon derived from pyrolyzed pinewood char using elevated temperature, KOH, H3PO4, and H2O2. BioResources 11:10433–10447. https://doi.org/10.15376/biores.11.4.10433-10447
Mills P, Sullivan JL (1983) A study of the core level electrons in iron and its three oxides by means of X-ray photoelectron spectroscopy. J Phys D Appl Phys 16:723–732
Mostashari SM, Mostashari SZ (2008) Combustion pathway of cotton fabrics treated by ammonium sulfate as a flame-retardant studied by TG. J Therm Anal Calorim 91:437–441
Muxel AA, Gimenez SMN, de Souza Almeida FA, da Silva Alfaya RV, da Silva Alfaya AA (2011) Cotton Fiber/ZrO2, a new material for adsorption of Cr(VI) ions in water. Clean: Soil, Air, Water 39:289–295. https://doi.org/10.1002/clen.201000165
Nahil MA, Williams PT (2010) Activated carbons from acrylic textile waste. J Anal Appl Pyrolysis 89:51–59. https://doi.org/10.1016/j.jaap.2010.05.005
Ohmukai Y, Hasegawa I, Fujisawa H, Okuma O, Mae K (2008) Production of an iron-loaded carbonaceous material through pyrolyzing biomass impregnated with FeCl2. Fuel 87:2041–2049. https://doi.org/10.1016/j.fuel.2007.12.013
Oliveira LC, Pereira E, Guimaraes IR, Vallone A, Pereira M, Mesquita JP, Sapag K (2009) Preparation of activated carbons from coffee husks utilizing FeCl3 and ZnCl2 as activating agents. J Hazard Mater 165:87–94. https://doi.org/10.1016/j.jhazmat.2008.09.064
Rangabhashiyam S, Selvaraju N (2015a) Adsorptive remediation of hexavalent chromiumfromsynthetic wastewater by a natural and ZnCl2 activated Sterculia guttata shell. J Mol Liq 207:39–49. https://doi.org/10.1016/j.molliq.2015.03.018
Rangabhashiyam S, Selvaraju N (2015b) Efficacy of unmodified and chemically modified Swietenia mahagoni shells for the removal of hexavalent chromium from simulated wastewater. J Mol Liq 209:487–497. https://doi.org/10.1016/j.molliq.2015.06.033
Rudnev VS, Morozova VP, Lukiyanchuk IV, Tkachenko IA, Adigamova MV, Nedozorov PM (2018) The effect of iron precursors in an electrolyte on the formation, composition, and magnetic properties of oxide coatings on titanium. Prot Met Phys Chem 53:1005–1014. https://doi.org/10.1134/s2070205117060193
Rufford TE, Hulicova-Jurcakova D, Zhu Z, Lu GQ (2011) A comparative study of chemical treatment by FeCl3, MgCl2, and ZnCl2 on microstructure, surface chemistry, and double-layercapacitance of carbons from waste biomass. J Mater Res 25:1451–1459. https://doi.org/10.1557/JMR.2010.0186s
Salnikow K, Zhitkovich A (2008) Genetic and epigenetic mechanisms in metal carcinogenesis and cocarcinogenesis: nickel, arsenic, and chromium. Chem Res Toxicol 21:28–44. https://doi.org/10.1021/tx700198a
Shen Y, Fu Y (2018) KOH-activated rice husk char via CO2 pyrolysis for phenol adsorption. Mater Today Energy 9:397–406. https://doi.org/10.1016/j.mtener.2018.07.005
Shouman MA, Fathy NA, Khedr SA, Attia AA (2013) Comparative biosorption studies of hexavalent chromium ion onto raw and modified palm branches. Adv Phys Che 2013:1–9. https://doi.org/10.1155/2013/159712
Silva TL, Cazetta AL, PSC S, Zhang T, Asefa T, Almeida VC (2018) Mesoporous activated carbon fibers synthesized from denim fabric waste: efficient adsorbents for removal of textile dye from aqueous solutions. J Clean Prod 171:482–490. https://doi.org/10.1016/j.jclepro.2017.10.034
Sun Y, Yue Q, Gao B, Li Q, Huang L, Yao F, Xu X (2012) Preparation of activated carbon derived from cotton linter fibers by fused NaOH activation and its application for oxytetracycline (OTC) adsorption. J Colloid Interface Sci 368:521–527. https://doi.org/10.1016/j.jcis.2011.10.067
Tang S, Chen Y, Xie R, Jiang W, Jiang Y (2016) Preparation of activated carbon from corn cob and its adsorption behavior on Cr(VI) removal. Water Sci Technol 73:2654–2661. https://doi.org/10.2166/wst.2016.120
Wang S, Liu Q, Liao Y, Luo Z, Cen K (2007) A study on the mechanism research on cellulose pyrolysis under catalysis of metallic salts. Korean J Chem Eng 24:336–340
Wang M, Ren K, Wang L (2008) Iron-catalyzed ligand-free carbon-selenium (or tellurium) coupling of arylboronic acids with diselenides and ditellurides. Adv Synth Catal 351:1586–1594. https://doi.org/10.1002/adsc.200900095
Wang L, Yao Y, Sun L, Mao Y, Lu W, Huang S, Chen W (2014) Rapid removal of dyes under visible irradiation over activated carbon fibers supported Fe(III)–citrate at neutral pH. Sep Purif Technol 122:449–455. https://doi.org/10.1016/j.seppur.2013.11.029
Wu S, Chen G, Kim NY, Ni K, Zeng W, Zhao Y, Tao Z, Ji H, Lee Z, Zhu Y (2016) Creating pores on graphene platelets by low-temperature KOH activation for enhanced electrochemical performance. Small 12:2376–2384. https://doi.org/10.1002/smll.201503855
Xu Z, Tian D, Sun Z, Zhang D, Zhou Y, Chen W, Deng H (2019) Highly porous activated carbon synthesized by pyrolysis of polyester fabric wastes with different iron salts: pore development and adsorption behavior. Colloids Surf A Physicochem Eng Asp 565:180–187. https://doi.org/10.1016/j.colsurfa.2019.01.007
Yu Q, Zhuang X, Yuan Z, Qi W, Wang Q, Tan X (2011) The effect of metal salts on the decomposition of sweet sorghum bagasse in flow-through liquid hot water. Bioresour Technol 102:3445–3450. https://doi.org/10.1016/j.biortech.2010.10.084
Yuan J, Giordano C, Antonietti M (2010) Ionic liquid monomers and polymers as precursors of highly conductive, mesoporous, graphitic carbon nanostructures. Chem Mater 22:5003–5012. https://doi.org/10.1021/cm1012729
Zhang X, Li YX, Li GY, Hu CW (2015a) Preparation of Fe/activated carbon directly from rice husk pyrolytic carbon and its application in catalytic hydroxylation of phenol. RSC Adv 5:4984–4992. https://doi.org/10.1039/c4ra13248c
Zhang YJ, Ou JL, Duan ZK, Xing ZJ, Wang Y (2015b) Adsorption of Cr(VI) on bamboo bark-based activated carbon in the absence and presence of humic acid. Colloid Surf A 481:108–116. https://doi.org/10.1016/j.colsurfa.2015.04.050
Zhang S et al (2016) The role and mechanism of K2CO3 and Fe3O4 in the preparation of magnetic peanut shell based activated carbon. Powder Technol 295:152–160. https://doi.org/10.1016/j.powtec.2016.03.034
Zhou QQ, Chen XY, Wang B (2012) An activation-free protocol for preparing porous carbon from calcium citrate and the capacitive performance. Microporous Mesoporous Mater 158:155–161. https://doi.org/10.1016/j.micromeso.2012.03.031
Zhu X, Liu Y, Luo G, Qian F, Zhang S, Chen J (2014) Facile fabrication of magnetic carbon composites from hydrochar via simultaneous activation and magnetization for triclosan adsorption. Environ Sci Technol 48:5840–5848. https://doi.org/10.1021/es500531c
Zhu J, Xu D, Wang C, Qian W, Guo J, Yan F (2017) Ferric citrate-derived N-doped hierarchical porous carbons for oxygen reduction reaction and electrochemical supercapacitors. Carbon 115:1–10. https://doi.org/10.1016/j.carbon.2016.12.084
Funding
This work was financially supported by the National Natural Science Foundation of China (21707090), Chinese Postdoctoral Science Foundation (2017M611590), and Shanghai Natural Science Foundation (14ZR1428900).
Author information
Authors and Affiliations
Corresponding authors
Additional information
Responsible editor: Tito Roberto Cadaval Jr
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Xu, Z., Gu, S., Sun, Z. et al. Synthesis of char-based adsorbents from cotton textile waste assisted by iron salts at low pyrolysis temperature for Cr(VI) removal. Environ Sci Pollut Res 27, 11012–11025 (2020). https://doi.org/10.1007/s11356-019-07588-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-019-07588-4