Abstract
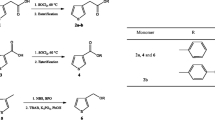
2,2,6,6-Tetramethylpiperidine 1-oxyl (TEMPO) is a versatile compound which is used mainly in organic oxidation reactions and organic battery applications. Incorporating TEMPO into a polymer could provide many applications into its repertoire. In this study, new polymeric TEMPO derivatives are synthesized and characterized to be used as catalysts in the presence of another polymer which constitutes N-halamine functionality. These polymeric mixtures were superior since the mixture could easily be separated from the reaction medium by simple filtration and reused in some other oxidation reactions numerous times. In turn, an all polymeric oxidation method was developed for oxidation of primary alcohols.





Similar content being viewed by others
References
Lu J, Toy PH (2009). Chem Rev 109(2):815–838
Zarchi MAK, Mousavi SZ (2014). J Polym Res 21:330
Eames J, Watkinson M (2001) Eur. J. Org. Chem. 1213
Chen C, Yang Z, Da Z, Qui F, Cao G, Guan Y, Yang D (2015) J Polym. Res. 22, 10, 188
Dickerson TJ, Reed NN, Janda KD (2002). Chem Rev 102(10):3325–3344
Salimi H, Rahimi A, Pourjavadi A (2007). Monatsh Chem 138(4):363–379
Tsutsuba T, Sogawa H, Takata T (2017). Polym Chem 8:1445–1448
Tzschucke CC, Markert C, Bannwarth W, Roller S, Hebel A, Haag R (2002) Angew. Chem. Int. Ed., 41-3964
Sertkol SB, Sinirlioglu D, Esat B (2015) J. Polym. Res. 22-136
Lewandowski M, Gwozdzinski K (2017) Int. J. Mol. Sci. 18(11)
Soule BP, Hyodo F, Matsumoto K, Simone NL, Cook JA, Krishna MC, Mitchell JB (2007). Free Radic Biol Med 42:1632–1650
Krishna MC, Grahame DA, Samuni A, Mitchell JB, Russo A (1992). Proc Natl Acad Sci 89:5537–5541
Adam W, Saha-Möller CR, Ganeshpure PA (2001). Chem Rev 101:3499–3548
Hoover, J. M.; Ryland, B. L.; Stahl, S. S. J Am Chem Soc 2013, 135, 2357–2367
Dijksman A, Marino-González A, Payeras A, Arends I, Sheldon R (2001). J Am Chem Soc 123:6826–6833
Dickerson TJ, Reed NN, Janda KD (2002). Chem Rev 102:3325–3344
Benaglia M, Puglisi A, Cozzi F (2003). Chem Rev 103:3401–3429
Tsubokawa, N.; Kimoto, T.; Endo, T. J Mol Catal 1995, 101, 45–50
Le D, Dilger M, Pertici V, Diabaté S, Gigmes D, Weiss C, Delaittre G (2019). Angew Chem Int Ed Engl 58(14):4725–4731
de Nooy AEJ, Besemer AC, van Bekkum H (1995). Synthesis:1153–1174
Vogler, T.; Studer, A (2008) Synthesis 1979–1993
Bobbitt JM, Flores MCL (1988). Heterocycles 27:509–533
Cella, J.A.; Kelley J.A.; Kenehan, E. F. J Org Chem 1975, 40, 1860–1862
Anelli, P. L.; Biffi, C.; Montanari, F.; Quici, S. J Org Chem 1987, 52, 2559–2562
De Mico, A.; Margarita, R.; Parlanti, L.; Vescovi, A.; Piancatelli, G. J Org Chem 1997, 62, 6974–6977
Einhorn, J.; Einhorn, C.; Ratajczak, F.; Pierre, J.-L. J Org Chem 1996, 61, 7452–7454
Akdag A, Öztürk P (2013). Synlett 24:2451–2453
Sun, G.; B. Wheatle, W.B.; Worley, S. D. Ind. Eng. Chem. Res.1994331168-170
Akdag A, Webb T, Worley SD (2006). Tetrahedron Lett 47:3509–3510
Akdag, A.; Okur, S.; McKee, M. L.; Worley S.D. J Chem Theory Comput, 2006, 2, 879-884
Ren X, Akdag A, Kocer HB, Worley SD, Broughton RM, Huang TS (2009) Carbohydr. Polym. 220-226
Demir B, Broughton RM, Qiao M, Huang T-S, Worley SD (2017). Molecules 22:1582
Potier J, Commarieu B, Soldera A, Claverie JP (2018). ACS Catal 8:6047–6054
Grubbs, R.H.; Trnka, T.M. Ruthenium in Organic Synthesis, Murahashi, S. (Ed.) Wiley-VCH: Weinheim, Germany, 2005: 6, 156-158
Rintoul L, Micallef AS, Bottle SE (2008) Spectrochim. Acta Part A Mol Biomol Spectrosc 70:713–717
Acknowledgments
We would like to acknowledge Middle East Technical University Research Funds (BAP): BAP-01-03-2015-003, BAP-07-02-2014-007-690, BAP-08-11-2012-120.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
ESM 1
(DOCX 13.0 MB)
Rights and permissions
About this article
Cite this article
Öztürk, P., Yücel, E.A. & Akdag, A. New TEMPO containing polymers for all polymeric oxidation procedure. J Polym Res 27, 30 (2020). https://doi.org/10.1007/s10965-019-1996-x
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10965-019-1996-x