Abstract
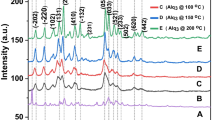
Alumina and phosphorus-alumina with different morphologies were synthesized by the sol–gel method. The morphology, structure, size, phase composition, acidic properties, and thermal behavior were examined by FESEM, TEM, XRD, FT-IR, BET, EDS, NH3-TPD, and TGA-DTA, respectively. It can be found that the morphology of alumina can change from worm-like to spherical by using acetonitrile/2-octanol solvent mixture and by modifying the synthesis steps. Also, the semi-crystalline structure of γ-alumina was changed to amorphous with improved surface area (from 150 to 229 m2 g−1). The addition of phosphorus pentoxide to amorphous alumina with the ratios of 1:9, 2:8 and 1:1 reduced the surface area of the catalyst after calcination from 229 to 129, 16 and 39 m2 g−1, respectively. With the increase of phosphorus/aluminum ratio and an increase in calcination temperature, the structure of phosphorus-alumina transformed from amorphous to crystalline. The reactivity and selectivity of 2-octanol over these composites were monitored using GC and GC-mass analyses.













Similar content being viewed by others
References
Samain L, Jaworski A, Edén M, Ladd DM, Seo DK, Garcia-Garcia FJ, Häussermann U (2014) J Solid State Chem 217:1–8
Naeimi H, Salimi F, Rabiei K (2006) J Mol Catal A 260:100–104
Kabalka GW, Pagni RM (1997) Tetrahedron 53:7999–8065
Yu J, Bai H, Wang J, Li Z, Jiao C, Liu Q, Zhang M, Liu L (2013) N J Chem 37:366–372
Srivastava V, Weng CH, Singh VK, Sharma YC (2011) J Chem Eng Data 56:1414–1422
Sanchez-Valente J, Bokhimi X, Toledo JA (2004) Appl Catal A 264:175–181
Maggi R, Ballini R, Sartori G, Sartorio R (2004) Tetrahedron Lett 45:2297–2299
Siahpoosh SM, Salahi E, Hessari FA, Mobasherpour I (2017) Sigma J Eng Natl Sci 35:441
Chen X, Zheng Y, Huang F, Xiao Y, Cai G, Zhang Y, Zheng Y, Jiang L (2018) ACS Catal 8:11016–11028
Shen M, Song L, Wang J, Wang X (2012) Catal Commun 22:28–33
Digne M, Sautet P, Raybaud P, Euzen P, Toulhoat H (2004) J Catal 226:54–68
Chen C, Feng B, Hu S, Zhang Y, Li S, Gao L (2018) Yu K. Ceram Int 44:216–224
Wang J, Wang Y, Wen J, Shen M, Wang W (2009) Microporous Mesoporous Mater 121:208–218
Gishti K, Iannibello A, Marengo S, Morellili G, Tittarelli P (1984) Appl Catal 12:381–393
Decanio EC, Edwards JC, Scalzo TR, Storm DA, Bruno JW (1991) J Catal 132:498–511
Morterra C, Magnacca G, Demaestri PP (1995) J Catal 152:384–395
ChandraKishore S, Pandurangan A (2013) Chem Eng J 222:472–477
Lei SHI, Zhang ZH, Qiu ZG, Fang GUO, Zhang W, Zhao LF (2015) J Fuel Chem Technol 43:74–80
Mekhemer GA, Nohman AK, Fouad NE, Khalaf HA (2000) Colloid Surf A 161:439–446
Labalme V, Béguin B, Gaillard F, Primet M (2000) Appl Catal A 192:307–316
Blanco A, Campelo JM, Garcia A, Luna D, Marinas JM, Moreno MS (1989) Appl Catal 53:135–156
Petrakis DE, Pomonis PJ, Sdoukos AT (1991) J Chem Soc Faraday Trans 87:1439–1445
Busca G, Ramis G, Lorenzelli V, Rossi PF, La Ginestra A, Patrono P (1989) Langmuir 5:911–916
Ramis G, Rossi PF, Busca G, Lorenzelli V, La Ginestra A, Patrono P (1989) Langmuir 5:917–923
Quartararo J, Guelton M, Rigole M, Amoureux JP, Fernandez C, Grimblot J (1999) J Mater Chem 9:2637–2646
Gu W, Shen M, Chang X, Wang Y, Wang (2007) J Alloys Compd 441:311–316
Li J, Ma H, Sun Q, Ying W, Fang D (2015) Fuel Process Technol 134:32–38
Drüppel K, Hösch A, Franz G (2007) Am Miner 92:1695–1703
Takahashi R, Sato S, Sodesawa T, Arai K, Yabuki M (2005) J Catal 229:24–29
Shi BC, Davis BH (1995) J Catal 157:359–367
Roy S, Mpourmpakis G, Hong DY, Vlachos DG, Bhan A, Gorte RJ (2012) ACS Catal 2:1846–1853
Suhas DP, Aminabhavi TM, Raghu AV (2014) Polym Eng Sci 54:1774–1782
Kwak JH, Mei D, Peden CH, Rousseau R, Szanyi J (2011) J Catal Lett 141:649–655
Dabbagh HA, Zamani M (2011) Appl Catal A 404:141–148
Rouquerol J, Avnir D, Fairbridge CW, Everett DH, Haynes JM, Pernicone N, Ramsay JD, Sing KS, Unger KK (1994) Pure Appl Chem 66:1739–1758
Zdravkov BD, Čermák JJ, Šefara M, Janků J (2007) Cent Eur J Chem 5:385–395
Echaroj S, Santikunaporn M, Chavadej S (2015) React Kinet Mech Cat 114:75–91
Yang W, Hu J (2013) J Nanopart Res 15:1786
Sarkar D, Mohapatra D, Ray S, Bhattacharyya S, Adak S, Mitra N (2007) J Mater Sci 42:1847–1855
Yung SW, Chiang HY, Lai YS, Wu FB, Fu C, Lee YM (2015) Ceram Int 41:877–888
Parida KM, Pradhan AC, Das J, Sahu N (2009) Mater Chem Phys 113:244–248
Colomban PH (1988) J Mater Sci Lett 7:1324–1326
Ahsan MR, Uddin MA, Mortuza MG (2005) J Pure Appl Phys 43:89
Selasteen FD, Raj SAC, Moses AA, Prince FE, Getsy RE, Elakkiya R (2016) J Cryst Process Technol 6:11
Mtalsi K, Jei T, Montes M, Tayane S (2001) J Chem Technol Biotechnol 76:128–138
Gao Q, Chen J, Li S, Xu R (1996) Microporous Mater 7:219–223
Li X, Zhang W, Liu G, Jiang L, Zhu X, Pan C, Jiang D, Tang A (2003) React Kinet Catal Lett 79:365–371
Wang X, Liang F, Huang C, Li Y, Chen B (2016) Catal Sci Technol 6:6551–6560
Rosseto R, dos Santos ÁC, Galembeck F (2006) J Braz Chem Soc 17:1465–1472
Kostestkyy P, Yu J, Gorte RJ, Mpourmpakis G (2014) Catal Sci Technol 4:3861–3869
Acknowledgements
This project was financially supported by the Isfahan University of Technology Council, and College of Pardis, Chemistry Section, Isfahan University of Technology, Isfahan which the authors gratefully acknowledge.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Nazer, S., Dabbagh, H.A., Najafi Chermahini, A. et al. Surface modification of alumina with P2O5 and its application in 2-octanol dehydration. Reac Kinet Mech Cat 129, 265–282 (2020). https://doi.org/10.1007/s11144-019-01717-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11144-019-01717-3