Abstract
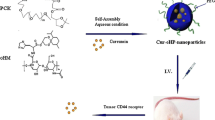
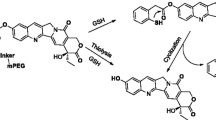
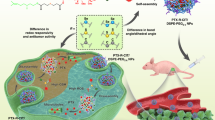
The key to improve the therapeutic efficacy for cancer treatment is to increase the delivery of drugs to tumors. For this purpose, tumor-microenvironment stimuli-responsive materials have great potential. Here, we prepared a new nanomedicine by bonding the conjugate of honokiol (HNK) and 5,6-dimethylxanthenone-4-acetic acid (DMXAA) to a glutathione (GSH)-responsive nanocarrier, poly(α-lipoic acid) polyethylene glycol. The nanomedicine would disintegrate due to the high level of GSH at the tumor sites, achieving the co-delivery of HNK and DMXAA, and realizing the combination therapy through close-range killing by HNK and long-range striking by DMXAA together. In a murine 4T1 breast tumor model, this strategy exhibited high tumor inhibition rate of 93%, and provided a valuable therapeutic choice for cancer therapy.
摘要
提高肿瘤治疗效果的关键在于增加药物肿瘤部位递送. 为此, 肿瘤微环境刺激响应性材料具有很大的潜力. 本文通过将和厚朴酚和DMXAA缀合物键合到GSH响应性聚硫辛酸-聚乙二醇纳米载体上, 制备出一种新型纳米药物. 在肿瘤部位高谷胱甘肽水平时纳米药物解体, 实现和厚朴酚与DMXAA在肿瘤部位的共同递送. 通过和厚朴酚的近程攻击与DMXAA的远程杀伤, 协同治疗乳腺癌. 在小鼠4T1乳腺肿瘤模型中, 该策略显示肿瘤抑制率高达93%, 为肿瘤治疗提供了有价值的候选治疗方案.
Article PDF
Similar content being viewed by others
Change history
22 April 2020
In the version of the article originally published in the volume 63, issue 2, 2020 of <Emphasis Type="Italic">Sci China Mater</Emphasis> (2020, 63 (2): 307–315, <ExternalRef><RefSource>https://doi.org/10.1007/s40843-019-1183-0</RefSource><RefTarget Address="10.1007/s40843-019-1183-0" TargetType="DOI"/></ExternalRef>), the affiliations of two of the authors (Zhaohui Tang and Xuesi Chen) were incompletely labeled. The corrected version of the authors’ affiliations is as below: Zhilin Liu<Superscript>1,2</Superscript>, Zhaohui Tang<Superscript>1,2*</Superscript>, Dawei Zhang<Superscript>1</Superscript>, Jiatan Wu<Superscript>3</Superscript>, Xinghui Si<Superscript>1,2</Superscript>, Na Shen<Superscript>1</Superscript> and Xuesi Chen<Superscript>1,2*</Superscript>
References
Chen H, Gu Z, An H, et al. Precise nanomedicine for intelligent therapy of cancer. Sci China Chem, 2018, 61: 1503–1552
Chen J, Lin L, Yan N, et al. Macrophages loaded CpG and GNR-PEI for combination of tumor photothermal therapy and immunotherapy. Sci China Mater, 2018, 61: 1484–1494
Hasani A, Leighl N. Classification and toxicities of vascular disrupting agents. Clin Lung Cancer, 2011, 12: 18–25
Daei Farshchi Adli A, Jahanban-Esfahlan R, Seidi K, et al. An overview on vadimezan (DMXAA): the vascular disrupting agent. Chem Biol Drug Des, 2018, 91: 996–1006
Bähr O, Gross S, Harter P, et al. ASA404, a vascular disrupting agent, as an experimental treatment approach for brain tumors. Oncol Lett, 2017, 14: 5443–5451
Hohenberger P, Elleni S, Marc F, et al. Vascular disrupting agent ASA 404 evaluated in human isolated ventilated and perfused lung lobes containing NSCLC. Eur J Cancer, 2015, 51: S608
Lv S, Tang Z, Song W, et al. Inhibiting solid tumor growth in vivo by non-tumor-penetrating nanomedicine. Small, 2017, 13: 1600954
Song W, Tang Z, Zhang D, et al. Solid tumor therapy using a cannon and pawn combination strategy. Theranostics, 2016, 6: 1023–1030
Cheng R, Meng F, Deng C, et al. Bioresponsive polymeric nanotherapeutics for targeted cancer chemotherapy. Nano Today, 2015, 10: 656–670
Cheng R, Meng F, Deng C, et al. Dual and multi-stimuli responsive polymeric nanoparticles for programmed site-specific drug delivery. Biomaterials, 2013, 34: 3647–3657
Ganta S, Devalapally H, Shahiwala A, et al. A review of stimuli-responsive nanocarriers for drug and gene delivery. J Control Release, 2008, 126: 187–204
Thornton P, Mart R, Ulijn R. Enzyme-responsive polymer hydrogel particles for controlled release. Adv Mater, 2007, 19: 1252–1256
Chen K, Liao S, Guo S, et al. Enzyme/pH-sensitive dendritic polymer-DOX conjugate for cancer treatment. Sci China Mater, 2018, 61: 1462–1474
Du JZ, Du XJ, Mao CQ, et al. Tailor-made dual pH-sensitive polymer-doxorubicin nanoparticles for efficient anticancer drug delivery. J Am Chem Soc, 2011, 133: 17560–17563
Du JZ, Sun TM, Song WJ, et al. A tumor-acidity-activated charge-conversional nanogel as an intelligent vehicle for promoted tumoral-cell uptake and drug delivery. Angew Chem Int Ed, 2010, 49: 3621–3626
Xu Q, He C, Xiao C, et al. Reactive oxygen species (ROS) responsive polymers for biomedical applications. Macromol Biosci, 2016, 16: 635–646
Yuan F, Li JL, Cheng H, et al. A redox-responsive mesoporous silica based nanoplatform for in vitro tumor-specific fluorescence imaging and enhanced photodynamic therapy. Biomater Sci, 2017, 6: 96–100
Chen D, Zhang G, Li R, et al. Biodegradable, hydrogen peroxide, and glutathione dual responsive nanoparticles for potential programmable paclitaxel release. J Am Chem Soc, 2018, 140: 7373–7376
Elzes MR, Akeroyd N, Engbersen JFJ, et al. Disulfide-functional poly(amido amine)s with tunable degradability for gene delivery. J Control Release, 2016, 244: 357–365
Zhang P, Wu J, Xiao F, et al. Disulfide bond based polymeric drug carriers for cancer chemotherapy and relevant redox environments in mammals. Med Res Rev, 2018, 38: 1485–1510
Perry RR, Mazetta J, Levin M, et al. Glutathione levels and variability in breast tumors and normal tissue. Cancer, 1993, 72: 783–787
Huang ZZ, Chen C, Zeng Z, et al. Mechanism and significance of increased glutathione level in human hepatocellular carcinoma and liver regeneration. Faseb J, 2001, 15: 19–21
Wolf CR, Lewis AD, Carmichael J, et al. The role of glutathione in determining the response of normal and tumour cells to anticancer drugs. Biochm Soc Trans, 1987, 15: 728–730
Schnelldorfer T, Gansauge S, Gansauge F, et al. Glutathione depletion causes cell growth inhibition and enhanced apoptosis in pancreatic cancer cells. Cancer, 2000, 89: 1440–1447
Lee MH, Yang Z, Lim CW, et al. Disulfide-cleavage-triggered chemosensors and their biological applications. Chem Rev, 2013, 113: 5071–5109
Nie JJ, Qiao B, Duan S, et al. Unlockable nanocomplexes with self-accelerating nucleic acid release for effective staged gene therapy of cardiovascular diseases. Adv Mater, 2018, 30: 1801570
Liu Z, Shen N, Tang Z, et al. An eximious and affordable GSH stimulus-responsive poly(α-lipoic acid) nanocarrier bonding combretastatin A4 for tumor therapy. Biomater Sci, 2019, 7: 2803–2811
Kisanuki A, Kimpara Y, Oikado Y, et al. Ring-opening polymerization of lipoic acid and characterization of the polymer. J Polym Sci A Polym Chem, 2010, 48: 5247–5253
Ishida H, Kisanuki A, Endo K. Ring-opening polymerization of aromatic 6-membered cyclic disulfide and characterization of the polymer. Polym J, 2009, 41: 110–117
Acknowledgements
This work was supported by the Ministry of Science and Technology of China (2018ZX09711003-012), the National Natural Science Foundation of China (51873206, 51673189, 51829302, 51503202, 51833010 and 51520105004), and the Program of Scientific Development of Jilin Province (20190103033JH).
Author information
Authors and Affiliations
Contributions
Author contributions Tang Z and Chen X put forward research ideas. Liu Z designed and synthesized the samples, Si X and Wu J performed some chemical synthesis experiments, Shen N and Zhang D performed the animal experiments; Liu Z wrote the paper with support from Tang Z and Chen X. All authors contributed to the general discussion.
Corresponding authors
Additional information
Conflict of interest The authors declare that they have no conflict of interest.
Zhilin Liu is a PhD candidate at the University of Science and Technology of China. His current research focuses on the tumor-microenvironment responsive nanomedicine of drug delivery system.
Zhaohui Tang is a professor in Changchun Institute of Applied Chemistry, Chinese Academy of Sciences. His research interests are focused on polymeric carriers for tumor treatment and combinational therapy. He has published more than 100 papers in SCI journals, such as Progress in Polymer Science, Advanced Materials, Advanced Science, Biomaterials, Small and Chemical Science.
Xuesi Chen is a professor in Changchun Institute of Applied Chemistry, Chinese Academy of Sciences. His research interests are focused on polymers chemistry on biomedical polymers, biodegradable polymers for drug/gene controlled release, bone repair parts and tissue engineering scaffolds. He published more than 600 papers in SCI journals, such as Progress in Polymer Science, Advanced Materials, Journal of the American Chemical Society and Advanced Functional Materials. He was authorized more than 200 Chinese invention patents.
Electronic supplementary material
40843_2019_1183_MOESM1_ESM.pdf
A novel GSH responsive poly(alpha-lipoic acid) nanocarrier bonding with the honokiol-DMXAA conjugate for combination therapy
Rights and permissions
About this article
Cite this article
Liu, Z., Tang, Z., Zhang, D. et al. A novel GSH responsive poly(alpha-lipoic acid) nanocarrier bonding with the honokiol-DMXAA conjugate for combination therapy. Sci. China Mater. 63, 307–315 (2020). https://doi.org/10.1007/s40843-019-1183-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40843-019-1183-0