Abstract
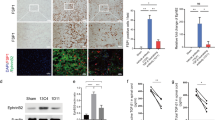
Despite the experimental evidence pointing to a significant role of the Wnt family of proteins in physiological and pathological rodent spinal cord functioning, its potential relevance in the healthy and traumatically injured human spinal cord as well as its therapeutic potential in spinal cord injury (SCI) are still poorly understood. To get further insight into these interesting issues, we first demonstrated by quantitative Real-Time PCR and simple immunohistochemistry that detectable mRNA expression of most Wnt components, as well as protein expression of all known Wnt receptors, can be found in the healthy human spinal cord, supporting its potential involvement in human spinal cord physiology. Moreover, evaluation of Frizzled (Fz) 1 expression by double immunohistochemistry showed that its spatio-temporal and cellular expression pattern in the traumatically injured human spinal cord is equivalent to that observed in a clinically relevant model of rat SCI and suggests its potential involvement in SCI progression/outcome. Accordingly, we found that long-term lentiviral-mediated overexpression of the Fz1 ligand Wnt1 after rat SCI improves motor functional recovery, increases myelin preservation and neuronal survival, and reduces early astroglial reactivity and NG2+ cell accumulation, highlighting the therapeutic potential of Wnt1 in this neuropathological situation.












Similar content being viewed by others
References
Grainger S, Willert K (2018) Mechanisms of Wnt signaling and control. Wiley Interdiscip Rev Syst Biol Med. https://doi.org/10.1002/wsbm.1422
van Amerongen R (2012) Alternative Wnt pathways and receptors. Cold Spring Harb Perspect Biol 4(10):a007914. https://doi.org/10.1101/cshperspect.a007914
Niehrs C (2012) The complex world of WNT receptor signalling. Nat Rev Mol Cell Biol 13(12):767–779. https://doi.org/10.1038/nrm3470
Engelhardt B, Liebner S (2014) Novel insights into the development and maintenance of the blood–brain barrier. Cell Tissue Res 355(3):687–699. https://doi.org/10.1007/s00441-014-1811-2
Brafman D, Willert K (2017) Wnt/beta-catenin signaling during early vertebrate neural development. Dev Neurobiol 77(11):1239–1259. https://doi.org/10.1002/dneu.22517
Guo F, Lang J, Sohn J, Hammond E, Chang M, Pleasure D (2015) Canonical Wnt signaling in the oligodendroglial lineage—puzzles remain. Glia 63(10):1671–1693. https://doi.org/10.1002/glia.22813
Salinas PC (2012) Wnt signaling in the vertebrate central nervous system: from axon guidance to synaptic function. Cold Spring Harb Perspect Biol 4(2):a008003. https://doi.org/10.1101/cshperspect.a008003
Helfer G, Tups A (2016) Hypothalamic Wnt signalling and its role in energy balance regulation. J Neuroendocrinol 28(3):12368. https://doi.org/10.1111/jne.12368
Fortress AM, Frick KM (2016) Hippocampal Wnt signaling: memory regulation and hormone interactions. Neuroscientist 22(3):278–294. https://doi.org/10.1177/1073858415574728
Ohata S, Nakatani J, Herranz-Perez V, Cheng J, Belinson H, Inubushi T, Snider WD, Garcia-Verdugo JM, Wynshaw-Boris A, Alvarez-Buylla A (2014) Loss of Dishevelleds disrupts planar polarity in ependymal motile cilia and results in hydrocephalus. Neuron 83(3):558–571. https://doi.org/10.1016/j.neuron.2014.06.022
Obermeier B, Daneman R, Ransohoff RM (2013) Development, maintenance and disruption of the blood–brain barrier. Nat Med 19(12):1584–1596. https://doi.org/10.1038/nm.3407
Oliva CA, Montecinos-Oliva C, Inestrosa NC (2018) Wnt signaling in the central nervous system: new insights in health and disease. Prog Mol Biol Transl Sci 153:81–130. https://doi.org/10.1016/bs.pmbts.2017.11.018
Xing L, Anbarchian T, Tsai JM, Plant GW, Nusse R (2018) Wnt/beta-catenin signaling regulates ependymal cell development and adult homeostasis. Proc Natl Acad Sci USA 115(26):E5954–E5962. https://doi.org/10.1073/pnas.1803297115
Gonzalez-Fernandez C, Arevalo-Martin A, Paniagua-Torija B, Ferrer I, Rodriguez FJ, Garcia-Ovejero D (2016) Wnts are expressed in the ependymal region of the adult spinal cord. Mol Neurobiol 54(8):6342–6355. https://doi.org/10.1007/s12035-016-0132-8
Gonzalez-Fernandez C, Gonzalez P, Andres-Benito P, Ferrer I, Rodriguez FJ (2019) Wnt signaling alterations in the human spinal cord of amyotrophic lateral sclerosis cases: spotlight on Fz2 and Wnt5a. Mol Neurobiol 56(10):6777–6791. https://doi.org/10.1007/s12035-019-1547-9
L'Episcopo F, Tirolo C, Caniglia S, Testa N, Morale MC, Serapide MF, Pluchino S, Marchetti B (2014) Targeting Wnt signaling at the neuroimmune interface for dopaminergic neuroprotection/repair in Parkinson's disease. J Mol Cell Biol 6(1):13–26. https://doi.org/10.1093/jmcb/mjt053
Xie C, Li Z, Zhang GX, Guan Y (2014) Wnt signaling in remyelination in multiple sclerosis: friend or foe? Mol Neurobiol 49(3):1117–1125. https://doi.org/10.1007/s12035-013-8584-6
Lambert C, Cisternas P, Inestrosa NC (2015) Role of Wnt signaling in central nervous system injury. Mol Neurobiol 53(4):2297–2311. https://doi.org/10.1007/s12035-015-9138-x
Libro R, Bramanti P, Mazzon E (2016) The role of the Wnt canonical signaling in neurodegenerative diseases. Life Sci 158:78–88. https://doi.org/10.1016/j.lfs.2016.06.024
Suwala AK, Hanaford A, Kahlert UD, Maciaczyk J (2016) Clipping the wings of glioblastoma: modulation of WNT as a novel therapeutic strategy. J Neuropathol Exp Neurol 75(5):388–396. https://doi.org/10.1093/jnen/nlw013
Lee Y, Lee JK, Ahn SH, Lee J, Nam DH (2016) WNT signaling in glioblastoma and therapeutic opportunities. Lab Investig 96(2):137–150. https://doi.org/10.1038/labinvest.2015.140
Tapia-Rojas C, Inestrosa NC (2018) Loss of canonical Wnt signaling is involved in the pathogenesis of Alzheimer's disease. Neural Regen Res 13(10):1705–1710. https://doi.org/10.4103/1673-5374.238606
Profyris C, Cheema SS, Zang D, Azari MF, Boyle K, Petratos S (2004) Degenerative and regenerative mechanisms governing spinal cord injury. Neurobiol Dis 15(3):415–436. https://doi.org/10.1016/j.nbd.2003.11.015
Oyinbo CA (2011) Secondary injury mechanisms in traumatic spinal cord injury: a nugget of this multiply cascade. Acta Neurobiol Exp (Wars) 71(2):281–299
Reilly P (2007) The impact of neurotrauma on society: an international perspective. Prog Brain Res 161:3–9. https://doi.org/10.1016/S0079-6123(06)61001-7
Priebe MM, Chiodo AE, Scelza WM, Kirshblum SC, Wuermser LA, Ho CH (2007) Spinal cord injury medicine. 6. Economic and societal issues in spinal cord injury. Arch Phys Med Rehabil 88 (3 Suppl 1):S84–88. https://doi.org/10.1016/j.apmr.2006.12.005
Witiw CD, Fehlings MG (2015) Acute spinal cord injury. J Spinal Disord Tech 28(6):202–210. https://doi.org/10.1097/BSD.0000000000000287
Rowland JW, Hawryluk GW, Kwon B, Fehlings MG (2008) Current status of acute spinal cord injury pathophysiology and emerging therapies: promise on the horizon. Neurosurg Focus 25(5):E2. https://doi.org/10.3171/FOC.2008.25.11.E2
Ahuja CS, Fehlings M (2016) Concise review: bridging the gap: novel neuroregenerative and neuroprotective strategies in spinal cord injury. Stem Cells Transl Med 5(7):914–924. https://doi.org/10.5966/sctm.2015-0381
Fernandez-Martos CM, Gonzalez-Fernandez C, Gonzalez P, Maqueda A, Arenas E, Rodriguez FJ (2011) Differential expression of Wnts after spinal cord contusion injury in adult rats. PLoS ONE 6(11):e27000. https://doi.org/10.1371/journal.pone.0027000
Gonzalez P, Fernandez-Martos CM, Gonzalez-Fernandez C, Arenas E, Rodriguez FJ (2012) Spatio-temporal expression pattern of frizzled receptors after contusive spinal cord injury in adult rats. PLoS ONE 7(12):e50793. https://doi.org/10.1371/journal.pone.0050793
Gonzalez P, Fernandez-Martos CM, Arenas E, Rodriguez FJ (2013) The Ryk receptor is expressed in glial and fibronectin-expressing cells after spinal cord injury. J Neurotrauma 30(10):806–817. https://doi.org/10.1089/neu.2012.2613
Gonzalez-Fernandez C, Fernandez-Martos CM, Shields SD, Arenas E, Javier Rodriguez F (2014) Wnts are expressed in the spinal cord of adult mice and are differentially induced after injury. J Neurotrauma 31(6):565–581. https://doi.org/10.1089/neu.2013.3067
Wu D, Pan W (2010) GSK3: a multifaceted kinase in Wnt signaling. Trends Biochem Sci 35(3):161–168. https://doi.org/10.1016/j.tibs.2009.10.002
Miyashita T, Koda M, Kitajo K, Yamazaki M, Takahashi K, Kikuchi A, Yamashita T (2009) Wnt-ryk signaling mediates axon growth inhibition and limits functional recovery after spinal cord injury. J Neurotrauma 26(7):955–964. https://doi.org/10.1089/neu.2008.0776
Liu Y, Wang X, Lu CC, Kerman R, Steward O, Xu XM, Zou Y (2008) Repulsive Wnt signaling inhibits axon regeneration after CNS injury. J Neurosci 28(33):8376–8382. https://doi.org/10.1523/JNEUROSCI.1939-08.2008
Hollis ER 2nd, Zou Y (2012) Reinduced Wnt signaling limits regenerative potential of sensory axons in the spinal cord following conditioning lesion. Proc Natl Acad Sci USA 109(36):14663–14668. https://doi.org/10.1073/pnas.1206218109
Suh HI, Min J, Choi KH, Kim SW, Kim KS, Jeon SR (2011) Axonal regeneration effects of Wnt3a-secreting fibroblast transplantation in spinal cord-injured rats. Acta Neurochir (Wien) 153(5):1003–1010. https://doi.org/10.1007/s00701-011-0945-1
Rodriguez JP, Coulter M, Miotke J, Meyer RL, Takemaru K, Levine JM (2014) Abrogation of beta-catenin signaling in oligodendrocyte precursor cells reduces glial scarring and promotes axon regeneration after CNS injury. J Neurosci 34(31):10285–10297. https://doi.org/10.1523/JNEUROSCI.4915-13.2014
Hollis ER 2nd, Ishiko N, Pessian M, Tolentino K, Lee-Kubli CA, Calcutt NA, Zou Y (2015) Remodelling of spared proprioceptive circuit involving a small number of neurons supports functional recovery. Nat Commun 6:6079. https://doi.org/10.1038/ncomms7079
Wehner D, Tsarouchas TM, Michael A, Haase C, Weidinger G, Reimer MM, Becker T, Becker CG (2017) Wnt signaling controls pro-regenerative collagen XII in functional spinal cord regeneration in zebrafish. Nat Commun 8(1):126. https://doi.org/10.1038/s41467-017-00143-0
Strand NS, Hoi KK, Phan TMT, Ray CA, Berndt JD, Moon RT (2016) Wnt/beta-catenin signaling promotes regeneration after adult zebrafish spinal cord injury. Biochem Biophys Res Commun 477(4):952–956. https://doi.org/10.1016/j.bbrc.2016.07.006
Park JH, Min J, Baek SR, Kim SW, Kwon IK, Jeon SR (2013) Enhanced neuroregenerative effects by scaffold for the treatment of a rat spinal cord injury with Wnt3a-secreting fibroblasts. Acta Neurochir (Wien) 155(5):809–816. https://doi.org/10.1007/s00701-013-1663-7
Zhang G, Lei F, Zhou Q, Feng D, Bai Y (2016) Combined application of Rho-ROCKII and GSK-3beta inhibitors exerts an improved protective effect on axonal regeneration in rats with spinal cord injury. Mol Med Rep 14(6):5180–5188. https://doi.org/10.3892/mmr.2016.5918
Renault-Mihara F, Katoh H, Ikegami T, Iwanami A, Mukaino M, Yasuda A, Nori S, Mabuchi Y, Tada H, Shibata S, Saito K, Matsushita M, Kaibuchi K, Okada S, Toyama Y, Nakamura M, Okano H (2011) Beneficial compaction of spinal cord lesion by migrating astrocytes through glycogen synthase kinase-3 inhibition. EMBO Mol Med 3(11):682–696. https://doi.org/10.1002/emmm.201100179
Cuzzocrea S, Genovese T, Mazzon E, Crisafulli C, Di Paola R, Muia C, Collin M, Esposito E, Bramanti P, Thiemermann C (2006) Glycogen synthase kinase-3 beta inhibition reduces secondary damage in experimental spinal cord trauma. J Pharmacol Exp Ther 318(1):79–89. https://doi.org/10.1124/jpet.106.102863
Hollis ER 2nd, Ishiko N, Yu T, Lu CC, Haimovich A, Tolentino K, Richman A, Tury A, Wang SH, Pessian M, Jo E, Kolodkin A, Zou Y (2016) Ryk controls remapping of motor cortex during functional recovery after spinal cord injury. Nat Neurosci 19(5):697–705. https://doi.org/10.1038/nn.4282
Yin ZS, Zu B, Chang J, Zhang H (2008) Repair effect of Wnt3a protein on the contused adult rat spinal cord. Neurol Res 30(5):480–486. https://doi.org/10.1179/174313208X284133
L'Episcopo F, Serapide MF, Tirolo C, Testa N, Caniglia S, Morale MC, Pluchino S, Marchetti B (2011) A Wnt1 regulated frizzled-1/beta-catenin signaling pathway as a candidate regulatory circuit controlling mesencephalic dopaminergic neuron-astrocyte crosstalk: therapeutical relevance for neuron survival and neuroprotection. Mol Neurodegener 6:49. https://doi.org/10.1186/1750-1326-6-49
Gonzalez P, Rodriguez FJ (2016) Analysis of the expression of the Wnt family of proteins and its modulatory role on cytokine expression in non activated and activated astroglial cells. Neurosci Res 114:16–29. https://doi.org/10.1016/j.neures.2016.08.003
L'Episcopo F, Tirolo C, Testa N, Caniglia S, Morale MC, Cossetti C, D'Adamo P, Zardini E, Andreoni L, Ihekwaba AE, Serra PA, Franciotta D, Martino G, Pluchino S, Marchetti B (2011) Reactive astrocytes and Wnt/beta-catenin signaling link nigrostriatal injury to repair in 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine model of Parkinson's disease. Neurobiol Dis 41(2):508–527. https://doi.org/10.1016/j.nbd.2010.10.023
Yu L, Guan Y, Wu X, Chen Y, Liu Z, Du H, Wang X (2013) Wnt Signaling is altered by spinal cord neuronal dysfunction in amyotrophic lateral sclerosis transgenic mice. Neurochem Res 38(9):1904–1913. https://doi.org/10.1007/s11064-013-1096-y
Gonzalez-Fernandez C, Mancuso R, Del Valle J, Navarro X, Rodriguez FJ (2016) Wnt signaling alteration in the spinal cord of amyotrophic lateral sclerosis transgenic mice: special focus on frizzled-5 cellular expression pattern. PLoS ONE 11(5):e0155867. https://doi.org/10.1371/journal.pone.0155867
Wang S, Guan Y, Chen Y, Li X, Zhang C, Yu L, Zhou F, Wang X (2013) Role of Wnt1 and Fzd1 in the spinal cord pathogenesis of amyotrophic lateral sclerosis-transgenic mice. Biotechnol Lett 35(8):1199–1207. https://doi.org/10.1007/s10529-013-1199-1
Matei N, Camara J, McBride D, Camara R, Xu N, Tang J, Zhang JH (2018) Intranasal Wnt3a attenuates neuronal apoptosis through Frz1/PIWIL1a/FOXM1 pathway in MCAO rats. J Neurosci 38(30):6787–6801. https://doi.org/10.1523/JNEUROSCI.2352-17.2018
Wang Y, Bao DJ, Xu B, Cheng CD, Dong YF, Wei XP, Niu CS (2019) Neuroprotection mediated by the Wnt/Frizzled signaling pathway in early brain injury induced by subarachnoid hemorrhage. Neural Regen Res 14(6):1013–1024. https://doi.org/10.4103/1673-5374.250620
Chacon MA, Varela-Nallar L, Inestrosa NC (2008) Frizzled-1 is involved in the neuroprotective effect of Wnt3a against Abeta oligomers. J Cell Physiol 217(1):215–227. https://doi.org/10.1002/jcp.21497
Dijksterhuis JP, Petersen J, Schulte G (2014) WNT/Frizzled signalling: receptor-ligand selectivity with focus on FZD-G protein signalling and its physiological relevance: IUPHAR Review 3. Br J Pharmacol 171(5):1195–1209. https://doi.org/10.1111/bph.12364
Gazit A, Yaniv A, Bafico A, Pramila T, Igarashi M, Kitajewski J, Aaronson SA (1999) Human frizzled 1 interacts with transforming Wnts to transduce a TCF dependent transcriptional response. Oncogene 18(44):5959–5966. https://doi.org/10.1038/sj.onc.1202985
Kajiwara K, Kamamoto M, Ogata S, Tanihara M (2008) A synthetic peptide corresponding to residues 301–320 of human Wnt-1 promotes PC12 cell adhesion and hippocampal neural stem cell differentiation. Peptides 29(9):1479–1485. https://doi.org/10.1016/j.peptides.2008.05.013
Chong ZZ, Shang YC, Hou J, Maiese K (2010) Wnt1 neuroprotection translates into improved neurological function during oxidant stress and cerebral ischemia through AKT1 and mitochondrial apoptotic pathways. Oxid Med Cell Longev 3(2):153–165. https://doi.org/10.4161/oxim.3.2.11758
Zhang J, Gotz S, Vogt Weisenhorn DM, Simeone A, Wurst W, Prakash N (2015) A WNT1-regulated developmental gene cascade prevents dopaminergic neurodegeneration in adult En1(+/−) mice. Neurobiol Dis 82:32–45. https://doi.org/10.1016/j.nbd.2015.05.015
Giry-Laterriere M, Verhoeyen E, Salmon P (2011) Lentiviral vectors. Methods Mol Biol 737:183–209. https://doi.org/10.1007/978-1-61779-095-9_8
Barde I, Salmon P, Trono D (2010) Production and titration of lentiviral vectors. Curr Protoc Neurosci Chapter 4:Unit 4 21. https://doi.org/10.1002/0471142301.ns0421s53
Fernandez-Martos CM, Gonzalez P, Rodriguez FJ (2012) Acute leptin treatment enhances functional recovery after spinal cord injury. PLoS ONE 7(4):e35594. https://doi.org/10.1371/journal.pone.0035594
Garcia-Ovejero D, Arevalo-Martin A, Paniagua-Torija B, Florensa-Vila J, Ferrer I, Grassner L, Molina-Holgado E (2015) The ependymal region of the adult human spinal cord differs from other species and shows ependymoma-like features. Brain 138(Pt 6):1583–1597. https://doi.org/10.1093/brain/awv089
Meyronet D, Dorey A, Massoma P, Rey C, Alix E, Silva K, Perrin C, Quadrio I, Perret-Liaudet A, Streichenberger N, Thomasset N, Honnorat J, Arzberger T, Kretzschmar H (2015) The workflow from post-mortem human brain sampling to cell microdissection: a Brain Net Europe study. J Neural Transm (Vienna) 122(7):975–991. https://doi.org/10.1007/s00702-015-1378-4
Paniagua-Torija B, Arevalo-Martin A, Molina-Holgado E, Molina-Holgado F, Garcia-Ovejero D (2015) Spinal cord injury induces a long-lasting upregulation of interleukin-1beta in astrocytes around the central canal. Neuroscience 284:283–289. https://doi.org/10.1016/j.neuroscience.2014.10.013
Hong LTA, Kim YM, Park HH, Hwang DH, Cui Y, Lee EM, Yahn S, Lee JK, Song SC, Kim BG (2017) An injectable hydrogel enhances tissue repair after spinal cord injury by promoting extracellular matrix remodeling. Nat Commun 8(1):533. https://doi.org/10.1038/s41467-017-00583-8
Basso DM, Beattie MS, Bresnahan JC (1996) Graded histological and locomotor outcomes after spinal cord contusion using the NYU weight-drop device versus transection. Exp Neurol 139(2):244–256. https://doi.org/10.1006/exnr.1996.0098
Hamers FP, Koopmans GC, Joosten EA (2006) CatWalk-assisted gait analysis in the assessment of spinal cord injury. J Neurotrauma 23(3–4):537–548. https://doi.org/10.1089/neu.2006.23.537
Hamers FP, Lankhorst AJ, van Laar TJ, Veldhuis WB, Gispen WH (2001) Automated quantitative gait analysis during overground locomotion in the rat: its application to spinal cord contusion and transection injuries. J Neurotrauma 18(2):187–201. https://doi.org/10.1089/08977150150502613
Batka RJ, Brown TJ, McMillan KP, Meadows RM, Jones KJ, Haulcomb MM (2014) The need for speed in rodent locomotion analyses. Anat Rec (Hoboken) 297(10):1839–1864. https://doi.org/10.1002/ar.22955
Leszczynska AN, Majczynski H, Wilczynski GM, Slawinska U, Cabaj AM (2015) Thoracic hemisection in rats results in initial recovery followed by a late decrement in locomotor movements, with changes in coordination correlated with serotonergic innervation of the ventral horn. PLoS ONE 10(11):e0143602. https://doi.org/10.1371/journal.pone.0143602
MacDonald BT, He X (2012) Frizzled and LRP5/6 receptors for Wnt/beta-catenin signaling. Cold Spring Harb Perspect Biol 4(12):a007880. https://doi.org/10.1101/cshperspect.a007880
Mardones MD, Andaur GA, Varas-Godoy M, Henriquez JF, Salech F, Behrens MI, Couve A, Inestrosa NC, Varela-Nallar L (2016) Frizzled-1 receptor regulates adult hippocampal neurogenesis. Mol Brain 9:29. https://doi.org/10.1186/s13041-016-0209-3
Zhao Y, Zhang Q, Xi J, Xiao B, Li Y, Ma C (2015) Neuroprotective effect of fasudil on inflammation through PI3K/Akt and Wnt/beta-catenin dependent pathways in a mice model of Parkinson's disease. Int J Clin Exp Pathol 8(3):2354–2364
Chong ZZ, Li F, Maiese K (2007) Cellular demise and inflammatory microglial activation during beta-amyloid toxicity are governed by Wnt1 and canonical signaling pathways. Cell Signal 19(6):1150–1162. https://doi.org/10.1016/j.cellsig.2006.12.009
Bournat JC, Brown AM, Soler AP (2000) Wnt-1 dependent activation of the survival factor NF-kappaB in PC12 cells. J Neurosci Res 61(1):21–32. https://doi.org/10.1002/1097-4547(20000701)61:1%3c21:AID-JNR3%3e3.0.CO;2-7
Wei L, Sun C, Lei M, Li G, Yi L, Luo F, Li Y, Ding L, Liu Z, Li S, Xu P (2013) Activation of Wnt/beta-catenin pathway by exogenous Wnt1 protects SH-SY5Y cells against 6-hydroxydopamine toxicity. J Mol Neurosci 49(1):105–115. https://doi.org/10.1007/s12031-012-9900-8
Simons M, Nave KA (2015) Oligodendrocytes: myelination and axonal support. Cold Spring Harb Perspect Biol 8(1):a020479. https://doi.org/10.1101/cshperspect.a020479
Levine J (2016) The reactions and role of NG2 glia in spinal cord injury. Brain Res 1638(Pt B):199–208. https://doi.org/10.1016/j.brainres.2015.07.026
Hackett AR, Lee JK (2016) Understanding the NG2 glial scar after spinal cord injury. Front Neurol 7:199. https://doi.org/10.3389/fneur.2016.00199
Sofroniew MV, Vinters HV (2010) Astrocytes: biology and pathology. Acta Neuropathol 119(1):7–35. https://doi.org/10.1007/s00401-009-0619-8
Karimi-Abdolrezaee S, Billakanti R (2012) Reactive astrogliosis after spinal cord injury-beneficial and detrimental effects. Mol Neurobiol 46(2):251–264. https://doi.org/10.1007/s12035-012-8287-4
David S, Kroner A (2011) Repertoire of microglial and macrophage responses after spinal cord injury. Nat Rev Neurosci 12(7):388–399. https://doi.org/10.1038/nrn3053
Zhou X, He X, Ren Y (2014) Function of microglia and macrophages in secondary damage after spinal cord injury. Neural Regen Res 9(20):1787–1795. https://doi.org/10.4103/1673-5374.143423
Onishi K, Hollis E, Zou Y (2014) Axon guidance and injury-lessons from Wnts and Wnt signaling. Curr Opin Neurobiol 27:232–240. https://doi.org/10.1016/j.conb.2014.05.005
Hollis ER 2nd (2016) Axon guidance molecules and neural circuit remodeling after spinal cord injury. Neurotherapeutics 13(2):360–369. https://doi.org/10.1007/s13311-015-0416-0
Garcia AL, Udeh A, Kalahasty K, Hackam AS (2018) A growing field: the regulation of axonal regeneration by Wnt signaling. Neural Regen Res 13(1):43–52. https://doi.org/10.4103/1673-5374.224359
Filous AR, Schwab JM (2018) Determinants of axon growth, plasticity, and regeneration in the context of spinal cord injury. Am J Pathol 188(1):53–62. https://doi.org/10.1016/j.ajpath.2017.09.005
O'Shea TM, Burda JE, Sofroniew MV (2017) Cell biology of spinal cord injury and repair. J Clin Investig 127(9):3259–3270. https://doi.org/10.1172/JCI90608
Ghosh M, Pearse DD (2014) The role of the serotonergic system in locomotor recovery after spinal cord injury. Front Neural Circuits 8:151. https://doi.org/10.3389/fncir.2014.00151
Lein ES, Hawrylycz MJ, Ao N, Ayres M, Bensinger A, Bernard A, Boe AF, Boguski MS, Brockway KS, Byrnes EJ, Chen L, Chen TM, Chin MC, Chong J, Crook BE, Czaplinska A, Dang CN, Datta S, Dee NR, Desaki AL, Desta T, Diep E, Dolbeare TA, Donelan MJ, Dong HW, Dougherty JG, Duncan BJ, Ebbert AJ, Eichele G, Estin LK, Faber C, Facer BA, Fields R, Fischer SR, Fliss TP, Frensley C, Gates SN, Glattfelder KJ, Halverson KR, Hart MR, Hohmann JG, Howell MP, Jeung DP, Johnson RA, Karr PT, Kawal R, Kidney JM, Knapik RH, Kuan CL, Lake JH, Laramee AR, Larsen KD, Lau C, Lemon TA, Liang AJ, Liu Y, Luong LT, Michaels J, Morgan JJ, Morgan RJ, Mortrud MT, Mosqueda NF, Ng LL, Ng R, Orta GJ, Overly CC, Pak TH, Parry SE, Pathak SD, Pearson OC, Puchalski RB, Riley ZL, Rockett HR, Rowland SA, Royall JJ, Ruiz MJ, Sarno NR, Schaffnit K, Shapovalova NV, Sivisay T, Slaughterbeck CR, Smith SC, Smith KA, Smith BI, Sodt AJ, Stewart NN, Stumpf KR, Sunkin SM, Sutram M, Tam A, Teemer CD, Thaller C, Thompson CL, Varnam LR, Visel A, Whitlock RM, Wohnoutka PE, Wolkey CK, Wong VY, Wood M, Yaylaoglu MB, Young RC, Youngstrom BL, Yuan XF, Zhang B, Zwingman TA, Jones AR (2007) Genome-wide atlas of gene expression in the adult mouse brain. Nature 445(7124):168–176. https://doi.org/10.1038/nature05453
Zhang YK, Huang ZJ, Liu S, Liu YP, Song AA, Song XJ (2013) WNT signaling underlies the pathogenesis of neuropathic pain in rodents. J Clin Investig 123(5):2268–2286. https://doi.org/10.1172/JCI65364
Shi Y, Shu J, Gelman BB, Lisinicchia JG, Tang SJ (2013) Wnt signaling in the pathogenesis of human HIV-associated pain syndromes. J Neuroimmune Pharmacol 8(4):956–964. https://doi.org/10.1007/s11481-013-9474-4
Ftouh S, Akbar MT, Hirsch SR, de Belleroche JS (2005) Down-regulation of Dickkopf 3, a regulator of the Wnt signalling pathway, in elderly schizophrenic subjects. J Neurochem 94(2):520–530. https://doi.org/10.1111/j.1471-4159.2005.03239.x
Riise J, Plath N, Pakkenberg B, Parachikova A (2015) Aberrant Wnt signaling pathway in medial temporal lobe structures of Alzheimer's disease. J Neural Transm (Vienna) 122(9):1303–1318. https://doi.org/10.1007/s00702-015-1375-7
Zhang Z, Schittenhelm J, Guo K, Buhring HJ, Trautmann K, Meyermann R, Schluesener HJ (2006) Upregulation of frizzled 9 in astrocytomas. Neuropathol Appl Neurobiol 32(6):615–624. https://doi.org/10.1111/j.1365-2990.2006.00770.x
Miyaoka T, Seno H, Ishino H (1999) Increased expression of Wnt-1 in schizophrenic brains. Schizophr Res 38(1):1–6
Bruggink KA, Kuiperij HB, Gloerich J, Otte-Holler I, Rozemuller AJ, Claassen JA, Kusters B, Verbeek MM (2015) Dickkopf-related protein 3 is a potential Abeta-associated protein in Alzheimer's disease. J Neurochem 134(6):1152–1162. https://doi.org/10.1111/jnc.13216
Kim Y, Hong M, Do IG, Ha SY, Lee D, Suh YL (2015) Wnt5a, Ryk and Ror2 expression in glioblastoma subgroups. Pathol Res Pract 211(12):963–972. https://doi.org/10.1016/j.prp.2015.10.001
Wu W, Tian Y, Wan H, Song Y, Li J, Zhang L (2013) The expressions of Wnt/beta-catenin pathway-related components in brainstem gliomas. Can J Neurol Sci 40(3):355–360
Yang Z, Wang Y, Fang J, Chen F, Liu J, Wu J (2010) Expression and aberrant promoter methylation of Wnt inhibitory factor-1 in human astrocytomas. J Exp Clin Cancer Res 29:26. https://doi.org/10.1186/1756-9966-29-26
Seo DK, Kim JH, Min J, Yoon HH, Shin ES, Kim SW, Jeon SR (2017) Enhanced axonal regeneration by transplanted Wnt3a-secreting human mesenchymal stem cells in a rat model of spinal cord injury. Acta Neurochir (Wien) 159(5):947–957. https://doi.org/10.1007/s00701-017-3097-0
Zhang J, Li S, Wu Y (2017) Recovery of spinal cord injury following electroacupuncture in rats through enhancement of Wnt/beta-catenin signaling. Mol Med Rep 16(2):2185–2190. https://doi.org/10.3892/mmr.2017.6801
Shinozuka T, Takada R, Yoshida S, Yonemura S, Takada S (2019) Wnt produced by stretched roof-plate cells is required for the promotion of cell proliferation around the central canal of the spinal cord. Development 146(2):dev159343. https://doi.org/10.1242/dev.159343
Alizadeh A, Karimi-Abdolrezaee S (2016) Microenvironmental regulation of oligodendrocyte replacement and remyelination in spinal cord injury. J Physiol 594(13):3539–3552. https://doi.org/10.1113/JP270895
Papastefanaki F, Matsas R (2015) From demyelination to remyelination: the road toward therapies for spinal cord injury. Glia 63(7):1101–1125. https://doi.org/10.1002/glia.22809
Gaesser JM, Fyffe-Maricich SL (2016) Intracellular signaling pathway regulation of myelination and remyelination in the CNS. Exp Neurol 283(Pt B):501–511. https://doi.org/10.1016/j.expneurol.2016.03.008
Tawk M, Makoukji J, Belle M, Fonte C, Trousson A, Hawkins T, Li H, Ghandour S, Schumacher M, Massaad C (2011) Wnt/beta-catenin signaling is an essential and direct driver of myelin gene expression and myelinogenesis. J Neurosci 31(10):3729–3742. https://doi.org/10.1523/JNEUROSCI.4270-10.2011
Jahn O, Tenzer S, Werner HB (2009) Myelin proteomics: molecular anatomy of an insulating sheath. Mol Neurobiol 40(1):55–72. https://doi.org/10.1007/s12035-009-8071-2
Greer JM, Lees MB (2002) Myelin proteolipid protein—the first 50 years. Int J Biochem Cell Biol 34(3):211–215
Kikuchi A, Yamamoto H, Sato A, Matsumoto S (2011) New insights into the mechanism of Wnt signaling pathway activation. Int Rev Cell Mol Biol 291:21–71. https://doi.org/10.1016/B978-0-12-386035-4.00002-1
Spinsanti P, De Vita T, Caruso A, Melchiorri D, Misasi R, Caricasole A, Nicoletti F (2008) Differential activation of the calcium/protein kinase C and the canonical beta-catenin pathway by Wnt1 and Wnt7a produces opposite effects on cell proliferation in PC12 cells. J Neurochem 104(6):1588–1598. https://doi.org/10.1111/j.1471-4159.2007.05111.x
Schiavone D, Dewilde S, Vallania F, Turkson J, Di Cunto F, Poli V (2009) The RhoU/Wrch1 Rho GTPase gene is a common transcriptional target of both the gp130/STAT3 and Wnt-1 pathways. Biochem J 421(2):283–292. https://doi.org/10.1042/BJ20090061
Shang YC, Chong ZZ, Wang S, Maiese K (2011) Erythropoietin and Wnt1 govern pathways of mTOR, Apaf-1, and XIAP in inflammatory microglia. Curr Neurovasc Res 8(4):270–285
Cregg JM, DePaul MA, Filous AR, Lang BT, Tran A, Silver J (2014) Functional regeneration beyond the glial scar. Exp Neurol 253:197–207. https://doi.org/10.1016/j.expneurol.2013.12.024
Sofroniew MV (2009) Molecular dissection of reactive astrogliosis and glial scar formation. Trends Neurosci 32(12):638–647. https://doi.org/10.1016/j.tins.2009.08.002
Kilander MB, Halleskog C, Schulte G (2011) Recombinant WNTs differentially activate beta-catenin-dependent and -independent signalling in mouse microglia-like cells. Acta Physiol (Oxf) 203(3):363–372. https://doi.org/10.1111/j.1748-1716.2011.02324.x
Halleskog C, Mulder J, Dahlstrom J, Mackie K, Hortobagyi T, Tanila H, Kumar Puli L, Farber K, Harkany T, Schulte G (2011) WNT signaling in activated microglia is proinflammatory. Glia 59(1):119–131. https://doi.org/10.1002/glia.21081
Halleskog C, Dijksterhuis JP, Kilander MB, Becerril-Ortega J, Villaescusa JC, Lindgren E, Arenas E, Schulte G (2012) Heterotrimeric G protein-dependent WNT-5A signaling to ERK1/2 mediates distinct aspects of microglia proinflammatory transformation. J Neuroinflammation 9:111. https://doi.org/10.1186/1742-2094-9-111
Halleskog C, Schulte G (2013) WNT-3A and WNT-5A counteract lipopolysaccharide-induced pro-inflammatory changes in mouse primary microglia. J Neurochem 125(6):803–808. https://doi.org/10.1111/jnc.12250
Chen X, Hao J, Fu T, Liu J, Yu M, He S, Qian R, Zhang F (2016) Temporal and spatial expression of LGR5 after acute spinal cord injury in adult rats. Neurochem Res 41(10):2645–2654. https://doi.org/10.1007/s11064-016-1977-y
Endo M, Ubulkasim G, Kobayashi C, Onishi R, Aiba A, Minami Y (2017) Critical role of Ror2 receptor tyrosine kinase in regulating cell cycle progression of reactive astrocytes following brain injury. Glia 65(1):182–197. https://doi.org/10.1002/glia.23086
Lutgen V, Narasipura SD, Sharma A, Min S, Al-Harthi L (2016) beta-Catenin signaling positively regulates glutamate uptake and metabolism in astrocytes. J Neuroinflammation 13(1):242. https://doi.org/10.1186/s12974-016-0691-7
Ding S, Xu Z, Yang J, Liu L, Huang X, Wang X, Zhuge Q (2017) The involvement of the decrease of astrocytic Wnt5a in the cognitive decline in minimal hepatic encephalopathy. Mol Neurobiol 54(10):7949–7963. https://doi.org/10.1007/s12035-016-0216-5
Yang J, Zhang X, Wu Y, Zhao B, Liu X, Pan Y, Liu Y, Ding Y, Qiu M, Wang YZ, Zhao G (2016) Wnt/beta-catenin signaling mediates the seizure-facilitating effect of postischemic reactive astrocytes after pentylenetetrazole-kindling. Glia 64(6):1083–1091. https://doi.org/10.1002/glia.22984
Wanner IB, Anderson MA, Song B, Levine J, Fernandez A, Gray-Thompson Z, Ao Y, Sofroniew MV (2013) Glial scar borders are formed by newly proliferated, elongated astrocytes that interact to corral inflammatory and fibrotic cells via STAT3-dependent mechanisms after spinal cord injury. J Neurosci 33(31):12870–12886. https://doi.org/10.1523/JNEUROSCI.2121-13.2013
Oklinski MK, Skowronski MT, Skowronska A, Rutzler M, Norgaard K, Nieland JD, Kwon TH, Nielsen S (2016) Aquaporins in the spinal cord. Int J Mol Sci 17(12). https://doi.org/10.3390/ijms17122050
Olsen ML, Campbell SC, McFerrin MB, Floyd CL, Sontheimer H (2010) Spinal cord injury causes a wide-spread, persistent loss of Kir4.1 and glutamate transporter 1: benefit of 17 beta-oestradiol treatment. Brain 133 (Pt 4):1013–1025. https://doi.org/10.1093/brain/awq049
Lepore AC, O'Donnell J, Bonner JF, Paul C, Miller ME, Rauck B, Kushner RA, Rothstein JD, Fischer I, Maragakis NJ (2011) Spatial and temporal changes in promoter activity of the astrocyte glutamate transporter GLT1 following traumatic spinal cord injury. J Neurosci Res 89(7):1001–1017. https://doi.org/10.1002/jnr.22624
Lepore AC, O'Donnell J, Kim AS, Yang EJ, Tuteja A, Haidet-Phillips A, O'Banion CP, Maragakis NJ (2011) Reduction in expression of the astrocyte glutamate transporter, GLT1, worsens functional and histological outcomes following traumatic spinal cord injury. Glia 59(12):1996–2005. https://doi.org/10.1002/glia.21241
Boyce VS, Mendell LM (2014) Neurotrophic factors in spinal cord injury. Handb Exp Pharmacol 220:443–460. https://doi.org/10.1007/978-3-642-45106-5_16
Darian-Smith C (2009) Synaptic plasticity, neurogenesis, and functional recovery after spinal cord injury. Neuroscientist 15(2):149–165. https://doi.org/10.1177/1073858408331372
Liu J, Yang X, Jiang L, Wang C, Yang M (2012) Neural plasticity after spinal cord injury. Neural Regen Res 7(5):386–391. https://doi.org/10.3969/j.issn.1673-5374.2012.05.010
Onishi K, Hollis E, Zou Y (2014) Axon guidance and injury-lessons from Wnts and Wnt signaling. Curr Opin Neurobiol 27C:232–240. https://doi.org/10.1016/j.conb.2014.05.005
Liu Y, Shi J, Lu CC, Wang ZB, Lyuksyutova AI, Song XJ, Zou Y (2005) Ryk-mediated Wnt repulsion regulates posterior-directed growth of corticospinal tract. Nat Neurosci 8(9):1151–1159. https://doi.org/10.1038/nn1520
Fenstermaker AG, Prasad AA, Bechara A, Adolfs Y, Tissir F, Goffinet A, Zou Y, Pasterkamp RJ (2010) Wnt/planar cell polarity signaling controls the anterior-posterior organization of monoaminergic axons in the brainstem. J Neurosci 30(47):16053–16064. https://doi.org/10.1523/JNEUROSCI.4508-10.2010
Perrin FE, Noristani HN (2019) Serotonergic mechanisms in spinal cord injury. Exp Neurol 318:174–191. https://doi.org/10.1016/j.expneurol.2019.05.007
Li L, Hutchins BI, Kalil K (2010) Wnt5a induces simultaneous cortical axon outgrowth and repulsive turning through distinct signaling mechanisms. Sci Signal 3 (147):pt2
Serradj N, Agger SF, Hollis ER 2nd (2017) Corticospinal circuit plasticity in motor rehabilitation from spinal cord injury. Neurosci Lett 652:94–104. https://doi.org/10.1016/j.neulet.2016.12.003
Voloshanenko O, Gmach P, Winter J, Kranz D, Boutros M (2017) Mapping of Wnt-Frizzled interactions by multiplex CRISPR targeting of receptor gene families. FASEB J 31(11):4832–4844. https://doi.org/10.1096/fj.201700144R
Dhamdhere GR, Fang MY, Jiang J, Lee K, Cheng D, Olveda RC, Liu B, Mulligan KA, Carlson JC, Ransom RC, Weis WI, Helms JA (2014) Drugging a stem cell compartment using Wnt3a protein as a therapeutic. PLoS ONE 9(1):e83650. https://doi.org/10.1371/journal.pone.0083650
Acknowledgements
We would like to thank Virginia Pérez, Sandra Vázquez, and the technical staff from the Service of Microscopy and Image Analysis and the Service of Flow Cytometry at the National Hospital for Paraplegics for their outstanding technical help, especially to Javier Mazarío for his essential participation in the design of the automatized cell count method. We also like to thank Sam David for his help to obtain the lesioned human spinal cord samples. This work has been supported by the Fondo de Investigación Sanitaria (FIS) of Instituto de Salud Carlos III (Grant number PI12/2895; FEDER co-funded) and the Ministerio de Ciencia, Innovación y Universidades (Grant number RTI2018-097775-B-I00; FEDER co-funded).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no conflicts of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
18_2019_3427_MOESM1_ESM.tif
Online Resource 1 Determination of the primary antibody specificity used to visualize Fz1. This figure shows representative images obtained from non-lesioned (NL) rat spinal cord sections (a1-6), lesioned rat spinal cord sections (7 days post-injury) (b1-6) and NL human spinal cord sections (c1-6) used to determine the specificity of the anti-Fz1 primary antibody. (a1-2), (b1-2) and (c1-2) correspond to images obtained from spinal cord sections processed for the immunohistochemical visualization of Fz1 and used as positive controls (C+); (a3-4), (b3-4) and (c3-4) correspond to images obtained from spinal cord sections processed for the immunohistochemical visualization of Fz1 without the primary antibody and used as negative controls (C-); (a5-6), (b5-6) and (c5-6) correspond to images obtained from spinal cord sections processed for the immunohistochemical visualization of Fz1 with the primary antibody pre-adsorbed with its corresponding blocking peptide (1:20 antibody/blocking peptide weight excess) (1/20). (a), (b) and (c) correspond to schematic drawings of prototypical NL rat, lesioned rat and and NL human spinal cord sections respectively, indicating the approximate areas shown in the corresponding images. Scale bars = 100 µm. (TIF 64125 kb)
18_2019_3427_MOESM2_ESM.tif
Online Resource 2 Determination of the proper functioning of the lentiviral vector generated to overexpress Wnt1. This figure shows the data and representative images obtained from the different studies performed in cultured B1a cells to evaluate the proper functioning of the lentiviral vector generated to overexpress Wnt1. Briefly, to determine the multiplicity of infection (MOI) that should be used to conduct this set of experiments we tested the effects of various MOI (MOI 5, 10, 20 and 40) on different aspects such as cell number (a), percentage of transduced cells (b) and GFP intensity (c) at 3 and 7 days post-transduction (dpt) (n = 3 per group) by flow cytometry. Based in the results obtained we selected MOI 10 to perform the subsequent experiments, since it induced a similar transduction efficiency to that observed with higher MOIs (MOI 20 and 40) (b), led to a high transgene expression (c) and did not affect cell viability and/or proliferation (a). The existence of significant differences between groups in a-c was assessed by Two-way ANOVA followed by Bonferroni post-hoc test (#, ## and ### = p < 0.05, p < 0.01 and p < 0.001 vs. 3 dpt; *, ** and *** = p < 0.05, p < 0.01 and p < 0.001 vs. non-transduced cells (NT); &, && and &&& = p < 0.05, p < 0.01 and p < 0.001 vs. the immediately lower MOI). We next determined by Real Time PCR and western blot that, in comparison with NT cells and those transduced with a lentiviral vector that only induce the expression of the green fluorescence protein (GFP) (LV-GFP), transduction with a lentiviral vector that induce the expression of both GFP and Wnt1 (LV-Wnt1) induced a robust increase in Wnt1 expression at both the mRNA (d, n = 3 per group) and protein levels (e, n = 3 per group) at 7 dpt, when we have previously observed the maximum transduction efficiency (b) and transgene expression (c). The existence of statistically significant differences between groups in e was assessed by One-way ANOVA followed by Bonferroni post-hoc test (*** represent p < 0.001 vs. NT; $$$ represent p < 0.001 vs. LV-GFP). It should be noted that Wnt1 is a soluble protein that acts in both autocrine and paracrine fashions and, thus, its correct secretion to the extracellular environment is a critical step to exert its multiple biological activities. In this regard, we found that the protein levels of Wnt1 were also strongly upregulated in the culture medium of cells transduced with LV-Wnt1 in comparison with those of NT and cells transduced with LV-GFP (f, n = 3 per group), demonstrating that Wnt1 was conveniently secreted to the extracellular space. We next aimed to evaluate whether overexpressed Wnt1 was able to activate the canonical Wnt/β-catenin signaling pathway which is the main Wnt signaling pathway that is activated by Wnt1. For this purpose, we determined that incubation of cell cultures with recombinant Wnt1 (100 and 200 ng/ml) (120-17, Peprotech) during 1 h led to the activation of the canonical Wnt/β-catenin signaling pathway (g) (n = 3 per group and time of incubation). The existence of significant differences between groups in g was assessed by Two-way ANOVA followed by Bonferroni post-hoc test (* = p < 0.05 vs. control). Finally, we found that, in comparison with cell cultures incubated with conditioned medium from cells transduced with LV-GFP (1:5) (CM LV-GFP), cell cultures incubated during 1 h with conditioned medium from cells cultures transduced with LV-Wnt1 (1:5) (CM LV-Wnt1) induced a similar activation of this signaling pathway than that observed in cell cultures incubated during 1 h with conditioned medium from cell cultures transduced with LV-GFP (1:5) and supplemented with 200 ng/ml of recombinant Wnt1 (CM LV-GFP+ Wnt1) (h, n = 3 per group), demonstrating that overexpressed Wnt1 is biologically active. The existence of significant differences between groups in h was assessed by One-way ANOVA followed by Bonferroni post-hoc test (* = p < 0.05 vs. CM LV-GFP). In all cases, data are presented as the mean + SEM. (TIF 4260 kb)
18_2019_3427_MOESM3_ESM.tif
Online Resource 3 Determination of the injection method of lentiviral vectors in vivo. This figure shows the results obtained from the experiments performed to establish the in vivo intraparenchymal injection method of the lentiviral vectors in the lesioned rat spinal cord. (a) and (b) shows schematic drawings from the two methods tested. In the first one (3ip), one injection per rostro-caudal level (stereotaxic coordinates: 0 mm lateral and 1 mm depth) was performed in three rostro-caudal levels corresponding to the lesion epicenter, 1.5 mm rostral and 1.5 mm caudal (a). In the second one (6ip), two injections per rostro-caudal level (stereotaxic coordinates: 0.6/-0.6 mm lateral and 1 mm depth) were performed in the same rostro-caudal levels detailed above (b). Evaluation of transduction at 7 days post-injury showed that both injection methods induced a similar transduction in longitudinal sections corresponding to the median line, where transduced green fluorescence protein (GFP)+ cells were observed surrounding the lesioned area, mainly rostral and caudal to the lesion epicenter, and to a lesser extent in the injury site (c-e) (non-injected (NT) group, n = 2; 3ip group, n = 3; 6ip group, n = 3) (arrows in d and e point the areas where GFP+ cells were observed). However, those animals that received 6 injections of lentiviral particles displayed a wider lateral distribution of transduced GFP+ cells than those that received 3 injections (f). As a consequence, we selected the second injection method for the subsequent experiments performed to evaluate the effects exerted by Wnt1 overexpression in the traumatically injured spinal cord. Scale bars in c-e represent 1 mm. (TIF 7420 kb)
18_2019_3427_MOESM4_ESM.tif
Online Resource 4 Evaluation of transduction at 7 days after SCI. This figure shows representative images from the evaluation of transduction at 7 days post-injury in those animals used to evaluate the effects of Wnt1 overexpression after SCI, which were injected with a lentiviral vector that only induce the expression of the green fluorescence protein (GFP) (LV-GFP control group) (n = 5) or with a lentiviral vector that induce the expression of both GFP and Wnt1 (LV-Wnt1 group) (n = 5). (a), shows a schematic drawing of a prototypical lesioned longitudinal spinal cord section and indicates the approximate rostro-caudal levels where the representative images were obtained. b, b1, c, c1, d, d1, e, e1, f and f1 shows representative images from the LV-GFP control group, while g, g1, h, h1, i, i1, j, j1, k and k1 show representative images from the LV-Wnt1 group. Squares in a-k indicate the areas shown in the corresponding higher magnification images. Arrows in a1-k1 point to the cells shown in the corresponding insets. Scale bars in a-k represent 500 µm, while those in a1-k1 and in their corresponding insets represent 100 and 20 µm, respectively. (TIF 17121 kb)
18_2019_3427_MOESM5_ESM.tif
Online Resource 5 Evaluation of transduction at 14 days after SCI. This figure shows representative images from the evaluation of transduction at 14 days post-injury in those animals used to evaluate the effects of Wnt1 overexpression after SCI, which were injected with a lentiviral vector that only induce the expression of the green fluorescence protein (GFP) (LV-GFP control group) (n = 5) or with a lentiviral vector that induce the expression of both GFP and Wnt1 (LV-Wnt1 group) (n = 5). (a), shows a schematic drawing of a prototypical lesioned longitudinal spinal cord section and indicates the approximate rostro-caudal levels where the representative images were obtained. b, b1, c, c1, d, d1, e, e1, f and f1 shows representative images from the LV-GFP control group, while g, g1, h, h1, i, i1, j, j1, k and k1 show representative images from the LV-Wnt1 group. Squares in a-k indicate the areas shown in the corresponding higher magnification images. Arrows in a1-k1 point to the cells shown in the corresponding insets. Scale bars in a-k represent 500 µm, while those in a1-k1 and in their corresponding insets represent 100 and 20 µm, respectively. (TIF 12872 kb)
18_2019_3427_MOESM6_ESM.tif
Online Resource 6 Evaluation of transduction at 126 days after SCI. This figure shows representative images from the evaluation of transduction at 126 days post-injury in those animals used to evaluate the effects of Wnt1 overexpression after SCI, which were injected with a lentiviral vector that only induce the expression of the green fluorescence protein (GFP) (LV-GFP control group) (n = 10) or with a lentiviral vector that induce the expression of both GFP and Wnt1 (LV-Wnt1 group) (n = 7). (a), shows a schematic drawing of a prototypical lesioned longitudinal spinal cord section and indicates the approximate rostro-caudal levels where the representative images were obtained. b, b1, c, c1, d, d1, e, e1, f and f1 shows representative images from the LV-GFP control group, while g, g1, h, h1, i, i1, j, j1, k and k1 show representative images from the LV-Wnt1 group. Squares in a-k indicate the areas shown in the corresponding higher magnification images. Arrows in a1-k1 point to the cells shown in the corresponding insets. Scale bars in a-k represent 500 µm, while those in a1-k1 and in their corresponding insets represent 100 and 20 µm, respectively. (TIF 17167 kb)
18_2019_3427_MOESM7_ESM.tif
Online Resource 7 Separate analysis of different individual aspects of locomotion analyzed in the 21-point BBB open-field test. This figure shows data obtained from the separate analysis of different individual aspects of locomotion such as coordination (a), paw positions (b), tail position (c), stepping (d), and toe clearance (e), which were evaluated using the 21-point BBB open-field test. The analysis was performed in lesioned animals injected with a lentiviral vector that only induce the expression of the green fluorescence protein (GFP) (LV-GFP control group) or with a lentiviral vector that induce the expression of both GFP and Wnt1 (LV-Wnt1 group) at 1, 3, 7, 14, 21, 35, 49, 63, 77, 91, 105 and 119 days post-injury (dpi). Data obtained from all animals used to evaluate the effects exerted by Wnt1 overexpression, which were sacrificed at 7 (LV-GFP group, n = 5; LV-Wnt1 group, n = 5), 14 (LV-GFP group, n = 5; LV-Wnt1 group, n = 5) and 126 dpi (LV-GFP group, n = 10; LV-Wnt1 group, n = 7), were included in the analysis. The existence of statistically significant differences between groups was assessed by Two-way ANOVA followed by Bonferroni post-hoc test. Data are presented as the mean + SEM. *, ** and *** represent p < 0.05, p < 0.01 and p < 0.001 vs. LV-GFP. (TIF 4261 kb)
Rights and permissions
About this article
Cite this article
González, P., González-Fernández, C., Campos-Martín, Y. et al. Frizzled 1 and Wnt1 as new potential therapeutic targets in the traumatically injured spinal cord. Cell. Mol. Life Sci. 77, 4631–4662 (2020). https://doi.org/10.1007/s00018-019-03427-4
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00018-019-03427-4