Abstract
Objective
The aim of this study was to compare the age-dependent diagnostic performance of clinical scores and D-dimer testing to identify patients with suspected pulmonary embolism (PE).
Methods
Consecutive patients with suspected PE referred from the emergency department for computed tomography pulmonary angiography (CTPA) were retrospectively evaluated. Diagnostic scores (classic Wells score (WS), modified WS, simplified WS, revised Geneva score (GS), simplified GS, and YEARS score) were calculated from medical records. Results of D-dimer testing were retrieved from the laboratory database. CTPA was the diagnostic reference standard. Four age groups were analyzed (< 50, 50–64, 65–74, and ≥ 75 years). Statistical analysis used receiver operating characteristics as well as uni- and multivariate analyses with calculation of prediction models. The study was IRB approved.
Results
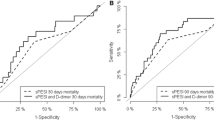
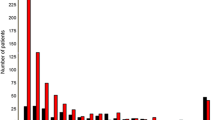
One thousand consecutive patients were included. Areas under the curve (AUC) and accuracies were superior in patients < 50 years. For the classic WS, the AUC decreased by 11% with the optimal cutoff dropping 1.5 points in patients ≥ 75 years; for D-dimer levels, the optimal cutoff was 900 μg/L higher in both ≥ 65 years groups with a max. decrease of the AUC of 9%. In terms of accuracy, the YEARS score performed best across all groups. Classic WS and D-dimer level showed a significant interaction with patient age in prediction models.
Conclusion
D-dimer measurement and clinical scores perform best in patients < 50 years. The YEARS score performs best across all age groups and is therefore recommended.
Key Points
• The probability of pulmonary embolism predicted by fibrin fibrinogen degradation products and clinical scores shows the highest accuracy in patients < 50 years.
• The probability of pulmonary embolism predicted by the YEARS score shows the highest accuracy in each age group.
• Classic Wells score and fibrin fibrinogen degradation products show a significant interaction with patient age in a logistic regression model.





Similar content being viewed by others
Abbreviations
- AUC:
-
Area under the curve
- CTPA:
-
Computed tomography pulmonary angiography
- cWS:
-
Classic Wells score
- DDL:
-
D-dimer level
- DVT:
-
Deep vein thrombosis
- mWS:
-
Modified Wells score
- PE:
-
Pulmonary embolism
- rGS:
-
Revised Geneva score
- ROC:
-
Receiver operating characteristics
- sGS:
-
Simplified Geneva score
- sWS:
-
Simplified Well score
References
Todoran TM, Giri J, Barnes GD et al (2018) Treatment of submassive and massive pulmonary embolism: a clinical practice survey from the second annual meeting of the Pulmonary Embolism Response Team Consortium. J Thromb Thrombolysis 46:39–49
Wells PS, Ginsberg JS, Anderson DR et al (1998) Use of a clinical model for safe management of patients with suspected pulmonary embolism. Ann Intern Med 129:997–1005
Wells PS, Anderson DR, Rodger M et al (2000) Derivation of a simple clinical model to categorize patients probability of pulmonary embolism: increasing the models utility with the SimpliRED D-dimer. Thromb Haemost 83:416–420
Gibson NS, Sohne M, Kruip MJ et al (2008) Further validation and simplification of the Wells clinical decision rule in pulmonary embolism. Thromb Haemost 99:229–234
Wicki J, Perneger TV, Junod AF, Bounameaux H, Perrier A (2001) Assessing clinical probability of pulmonary embolism in the emergency ward: a simple score. Arch Intern Med 161:92–97
Iles S, Hodges AM, Darley JR et al (2003) Clinical experience and pre-test probability scores in the diagnosis of pulmonary embolism. QJM 96:211–215
Klok FA, Mos IC, Nijkeuter M et al (2008) Simplification of the revised Geneva score for assessing clinical probability of pulmonary embolism. Arch Intern Med 168:2131–2136
Bounameaux H, Cirafici P, de Moerloose P et al (1991) Measurement of D-dimer in plasma as diagnostic aid in suspected pulmonary embolism. Lancet 337:196–200
Bounameaux H, de Moerloose P, Perrier A, Reber G (1994) Plasma measurement of D-dimer as diagnostic aid in suspected venous thromboembolism: an overview. Thromb Haemost 71:1–6
Ceriani E, Combescure C, Le Gal G et al (2010) Clinical prediction rules for pulmonary embolism: a systematic review and meta-analysis. J Thromb Haemost 8:957–970
van der Hulle T, Cheung WY, Kooij S et al (2017) Simplified diagnostic management of suspected pulmonary embolism (the YEARS study): a prospective, multicentre, cohort study. Lancet 390:289–297
Righini M, Le Gal G, Perrier A, Bounameaux H (2004) Effect of age on the assessment of clinical probability of pulmonary embolism by prediction rules. J Thromb Haemost 2:1206–1208
Righini M, Le Gal G, Perrier A, Bounameaux H (2005) The challenge of diagnosing pulmonary embolism in elderly patients: influence of age on commonly used diagnostic tests and strategies. J Am Geriatr Soc 53:1039–1045
Kiluk IE, Krajewska A, Kosacka U et al (2017) Different manifestations of pulmonary embolism in younger compared to older patients: clinical presentation, prediction rules and long-term outcomes. Adv Med Sci 62:254–258
Rohacek M, Buatsi J, Szucs-Farkas Z et al (2012) Ordering CT pulmonary angiography to exclude pulmonary embolism: defense versus evidence in the emergency room. Intensive Care Med 38:1345–1351
Sharif S, Eventov M, Kearon C et al (2018) Comparison of the age-adjusted and clinical probability-adjusted D-dimer to exclude pulmonary embolism in the emergency department. Am J Emerg Med. https://doi.org/10.1016/j.ajem.2018.07.053
Schouten HJ, Geersing GJ, Koek HL et al (2013) Diagnostic accuracy of conventional or age adjusted D-dimer cut-off values in older patients with suspected venous thromboembolism: systematic review and meta-analysis. BMJ 346:f2492
Youden WJ (1950) Index for rating diagnostic tests. Cancer 3:32–35
Guo DJ, Zhao C, Zou YD, Huang XH, Hu JM, Guo L (2015) Values of the Wells and revised Geneva scores combined with D-dimer in diagnosing elderly pulmonary embolism patients. Chin Med J (Engl) 128:1052–1057
Chagnon I, Bounameaux H, Aujesky D et al (2002) Comparison of two clinical prediction rules and implicit assessment among patients with suspected pulmonary embolism. Am J Med 113:269–275
Di Marca S, Cilia C, Campagna A et al (2015) Comparison of Wells and revised Geneva rule to assess pretest probability of pulmonary embolism in high-risk hospitalized elderly adults. J Am Geriatr Soc 63:1091–1097
Glober N, Tainter CR, Brennan J et al (2018) Use of the d-dimer for detecting pulmonary embolism in the emergency department. J Emerg Med 54:585–592
Curtis BR, Cox M, Poplawski M, Lyshchik A (2017) Low yield of ventilation and perfusion imaging for the evaluation of pulmonary embolism after indeterminate CT pulmonary angiography. Emerg Radiol 24:525–530
Acknowledgements
We want to thank Hans Tepe and Christine Naedler for their support with the database queries. The study was in part presented at ECR 2018.
Funding
The authors state that this work has not received any funding.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Guarantor
The scientific guarantor of this publication is Sebastian Nagel.
Conflict of interest
The authors of this manuscript declare relationships with the following companies:
Bernd Hamm is Grant Recipient for the Department of Radiology, Charité, and has further received funding from Abbott, AbbVie, Ablative Solutions, Accovion, Achaogen Inc., Actelion Pharmaceuticals, ADIR, Aesculap, AGO, AIF Arbeitsgemeinschaft industrieller Forschungsvereinigungen, AIO Arbeitsgemeinschaft lnternistische Onkologie, Alexion Pharmaceuticals, Amgen, AO Foundation, Arena Pharmaceuticals, art photonics GmbH Berlin, ASR Advanced sleep research, Astellas, AstraZeneca, BARD, Bayer Healthcare, Bayer Schering Pharma, Bayer Vital, BBraun, Berlin-Brandenburger Centrum für Regenerative Therapien (BCRT), Berliner Krebsgesellschaft, Biotronik, Bioven, BMBF Bundesministerium für Bildung und Forschung, Boehring Ingelheimer, Boston Biomedical Inc., BRACCO Group, Brainsgate, Bristol-Myers Squibb, Cascadian Therapeutics, Inc., Celgene, CELLACT Pharma, Celldex Therapeutics, CeIoNova BioSciences, Charité research organisation GmbH, Chiltern, CCovance, CUBIST, Curis, Daiichi, DC Devices, Inc. USA, Delcath Systems, Dermira Inc., Deutsche Krebshilfe, Deutsche Rheuma Liga, DFG, DSM Nutritional Products AG, Dt. Stiftung für Herzforschung, Dynavax, Eisai Ltd.‚ European Knowledge Centre, Mosquito Way, Hatfield, Eli Lilly and Company Ltd., EORTC, Epizyme, Inc., Essex Pharma, EU Programmes, Euroscreen S.A., Fibrex Medical Inc., Focused Ultrasound Surgery Foundation, Fraunhofer Gesellschaft, Galena Biopharma, Galmed Research and Development Ltd., Ganymed, GE, Genentech. Inc., GETNE (Grupo Español de Tumores Neuroendocrinos), Gilead Sciences, Inc., Glaxo Smith Kline, Glycotope GmbH, Berlin, Goethe Uni Frankfurt, Guerbet, Guidant Europe NV, Halozyme, Holaira Inc., ICON (CRO), Immunomedics Inc., Immunocore, Incyte, INC Research, Innate Pharma, InSightec Ltd., Inspiremd, inVentiv Health Clinical UK Ltd, Inventivhealth, IOMEDICO, IONIS, IPSEN Pharma, ISA Therapeutics, lsis Pharmaceuticals Inc., ITM Solution GmbH, Jansen, Kantar Health GmbH (CRO), Karyopharm Therapeutics, Inc., Kendle/MorphoSys AG, Kite Pharma, La Roche, Land Berlin, Lilly GmbH, Lion Biotechnology, Lombard Medical, Loxo Oncology, Inc, LSK BioPartners, USA; Lundbeck GmbH, LUX Biosciences, LYSARC, MacroGenics, MagForce, Medlmmune Inc., Medlmmune Limited, Medpace, Medpace Germany GmbH (CRO), MedPass (CRO), Medronic, Merck, Merrimack Pharmaceuticals Inc, MeVis Medical Solutions AG, Millennium Pharmaceuticals Inc., Mologen, MSD Sharp, NeoVacs SA, Nexus Oncology, Novartis, novocure, Nuvisan, Ockham oncology, Orion Corporation Orion Pharma, Parexel CRO Service, Perceptive, Pfizer GmbH, Pharma Mar, Pharmaceutical Research Associates GmbH (PRA), Pharmacyclics Inc., Philipps, PIQUR Therapeutics Ltd., Pluristem, Portola Pharmaceuticals, PPD (CRO), PRAint, Premier-research, Provectus Biopharmaceuticals, Inc., psi-cro, Pulmonx International Sàrl, Quintiles Gmbh, Respicardia, Roche, Samsung, Sanofi, sanofis-aventis S.A, Schumacher GmbH, Seattle Genetics, Servier (CRO), SGS Life Science Services (CRO), Siemens, Silena Therapeutics, Spectranetics GmbH, Spectrum Pharmaceuticals, St. Jude Medical, Stiftung Wolfgang Schulze, Symphogen, Taiho Pharmaceutical Co., Taqu Therapeutics Ltd., Terumo Medical Corporation, Tesaro, TETEC AG, TEVA, Theorem, Theradex, Threshold Pharmaceuticals Inc., TNS Healthcare GmbH, Toshiba, UCB Pharma, Uni München, VDI/VDE, Winicker Norimed, Wyeth Pharma, Xcovery Holding Company, Zukunftsfond Berlin (TSB).
Stefan Schwartz is receiving grants from Pfizer and Enzon.
Statistics and biometry
One of the authors (Ingo Steffen) has significant statistical expertise.
Informed consent
Written informed consent was waived by the Institutional Review Board.
Ethical approval
Institutional Review Board approval was obtained.
Methodology
• This study is a retrospective study performed at one institution on the diagnostic performance of clinical decision rules and D-dimer measurements in patients who had undergone computed tomography angiography in the diagnostic workup of suspected pulmonary embolism.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Nagel, S.N., Steffen, I.G., Schwartz, S. et al. Age-dependent diagnostic accuracy of clinical scoring systems and D-dimer levels in the diagnosis of pulmonary embolism with computed tomography pulmonary angiography (CTPA). Eur Radiol 29, 4563–4571 (2019). https://doi.org/10.1007/s00330-019-06039-5
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00330-019-06039-5