Abstract
Background and objectives
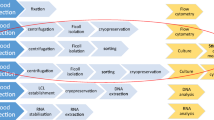
Liquid platelet rich fibrin (PRF; often referred to as injectable PRF) has been utilized as an injectable formulation of PRF that is capable of stimulating tissue regeneration. Our research group recently found that following standard L-PRF protocols (2700 RPM for 12 min), a massive increase in platelets and leukocytes was observed directly within the buffy-coat layer directly above the red blood cell layer. The purpose of this study was to develop a novel harvesting technique to isolate liquid PRF directly from this buffy coat layer and to compare this technique to standard i-PRF.
Materials and methods
Standard high g-force L-PRF and low g-force i-PRF protocols were utilized to separate blood layers. Above the red blood corpuscle layer, sequential 100-μL layers of plasma were harvested (12 layers total; i.e., 1.2 mL, which represents the total i-PRF volume), and 3 layers (3 × 100 μL) were harvested from the red blood cell layer to quantify blood cells. Each layer was then sent for complete blood count (CBC) analysis, and the cell numbers were quantified including red blood cells, leukocytes, neutrophils, lymphocytes, monocytes, and platelets. The liquid PRF that was directly collected from the buffy-coat layer following L-PRF protocols was referred to as concentrated PRF (C-PRF).
Results
The i-PRF protocol typically yielded a 2- to 3-fold increase in platelets and a l.5-fold increase in leukocyte concentration from the 1- to 1.2-mL plasma layer compared to baseline concentrations in whole blood. While almost no cells were found in the first 4-mL layer of L-PRF, a massive accumulation of platelets and leukocytes was found directly within the buffy coat layer demonstrating extremely high concentrations of cells in this 0.3–0.5-mL layer (~ 20-fold increases). We therefore proposed harvesting this 0.3- to 0.5-mL layer directly above the red blood cell corpuscle layer as liquid C-PRF. In general, i-PRF was able to increase platelet numbers by ~ 250%, whereas a 1200–1700% increase in platelet numbers could easily be achieved by harvesting this 0.3–0.5 mL of C-PRF (total platelet concentrations of > 2000–3000 × 109 cells/L).
Conclusion
While conventional i-PRF protocols increase platelet yield by 2-3-fold and leukocyte yield by 50%, we convincingly demonstrated the ability to concentrate platelets and leukocytes over 10-fold by harvesting the 0.3–0.5 mL of C-PRF within the buffy coat following L-PRF protocols.
Clinical relevance
Previous studies have demonstrated only a slight increase in platelet and leukocyte concentrations in i-PRF. The present study described a novel harvesting technique with over a 10-fold increase in platelets and leukocytes that can be further utilized for tissue regeneration.





Similar content being viewed by others
References
Chow TW, McIntire LV, Peterson DM (1983) Importance of plasma fibronectin in determining PFP and PRP clot mechanical properties. Thromb Res 29:243–248
Delaini F, Poggi A, Donati MB (1982) Enhanced affinity for arachidonic acid in platelet-rich plasma from rats with Adriamycin-induced nephrotic syndrome. Thromb Haemost 48:260–262
Marx RE (2004) Platelet-rich plasma: evidence to support its use. Journal of oral and maxillofacial surgery : official journal of the American Association of Oral and Maxillofacial Surgeons 62:489–496
Marx RE, Carlson ER, Eichstaedt RM, Schimmele SR, Strauss JE, Georgeff KR (1998) Platelet-rich plasma: growth factor enhancement for bone grafts. Oral Surg Oral Med Oral Pathol Oral Radiol Endod 85:638–646
Cai YZ, Zhang C, Lin XJ (2015) Efficacy of platelet-rich plasma in arthroscopic repair of full-thickness rotator cuff tears: a meta-analysis. Journal of shoulder and elbow surgery / American Shoulder and Elbow Surgeons [et al] 24:1852–1859. https://doi.org/10.1016/j.jse.2015.07.035
Meheux CJ, McCulloch PC, Lintner DM, Varner KE, Harris JD (2016) Efficacy of intra-articular platelet-rich plasma injections in knee osteoarthritis: a systematic review. Arthroscopy : the journal of arthroscopic & related surgery : official publication of the Arthroscopy Association of North America and the International Arthroscopy Association 32:495–505. https://doi.org/10.1016/j.arthro.2015.08.005
Singh B, Goldberg LJ (2016) Autologous Platelet-Rich Plasma for the Treatment of Pattern Hair Loss. Am J Clin Dermatol. https://doi.org/10.1007/s40257-016-0196-2
Anfossi G, Trovati M, Mularoni E, Massucco P, Calcamuggi G, Emanuelli G (1989) Influence of propranolol on platelet aggregation and thromboxane B2 production from platelet-rich plasma and whole blood. Prostaglandins Leukot Essent Fat Acids 36:1–7
Fijnheer R, Pietersz RN, de Korte D, Gouwerok CW, Dekker WJ, Reesink HW, Roos D (1990) Platelet activation during preparation of platelet concentrates: a comparison of the platelet-rich plasma and the buffy coat methods. Transfusion 30:634–638
Choukroun J, Adda F, Schoeffler C, Vervelle A (2001) Une opportunité en paro-implantologie: le PRF. Implantodontie 42:e62
Toffler M (2014) Guided bone regeneration (GBR) using cortical bone pins in combination with leukocyte-and platelet-rich fibrin (L-PRF). Compend Contin Educ Dent (Jamesburg, NJ : 1995) 35:192-198.
Lekovic V, Milinkovic I, Aleksic Z, Jankovic S, Stankovic P, Kenney E, Camargo P (2012) Platelet-rich fibrin and bovine porous bone mineral vs. platelet-rich fibrin in the treatment of intrabony periodontal defects. J Periodontal Res 47:409–417
Shivashankar VY, Johns DA, Vidyanath S, Sam G (2013) Combination of platelet rich fibrin, hydroxyapatite and PRF membrane in the management of large inflammatory periapical lesion. J Conserv Dent 16:261
Sammartino G, Dohan Ehrenfest DM, Carile F, Tia M, Bucci P (2011) Prevention of hemorrhagic complications after dental extractions into open heart surgery patients under anticoagulant therapy: the use of leukocyte- and platelet-rich fibrin. The Journal of oral implantology 37:681–690. https://doi.org/10.1563/aaid-joi-d-11-00001
Hoaglin DR, Lines GK (2013) Prevention of localized osteitis in mandibular third-molar sites using platelet-rich fibrin. International journal of dentistry 2013:875380. https://doi.org/10.1155/2013/875380
Suttapreyasri S, Leepong N (2013) Influence of platelet-rich fibrin on alveolar ridge preservation. The Journal of craniofacial surgery 24:1088–1094. https://doi.org/10.1097/SCS.0b013e31828b6dc3
Yelamali T, Saikrishna D (2015) Role of platelet rich fibrin and platelet rich plasma in wound healing of extracted third molar sockets: a comparative study. Journal of maxillofacial and oral surgery 14:410–416. https://doi.org/10.1007/s12663-014-0638-4
Anilkumar K, Geetha A, Umasudhakar, Ramakrishnan T, Vijayalakshmi R, Pameela E (2009) Platelet-rich-fibrin: a novel root coverage approach. Journal of Indian Society of Periodontology 13:50–54. https://doi.org/10.4103/0972-124x.51897
Jankovic S, Aleksic Z, Klokkevold P, Lekovic V, Dimitrijevic B, Kenney EB, Camargo P (2012) Use of platelet-rich fibrin membrane following treatment of gingival recession: a randomized clinical trial. The International journal of periodontics & restorative dentistry 32:e41–e50
Eren G, Tervahartiala T, Sorsa T, Atilla G (2015) Cytokine (interleukin-1beta) and MMP levels in gingival crevicular fluid after use of platelet-rich fibrin or connective tissue graft in the treatment of localized gingival recessions. J Periodontal Res. https://doi.org/10.1111/jre.12325
Jain V, Triveni MG, Kumar AB, Mehta DS (2012) Role of platelet-rich-fibrin in enhancing palatal wound healing after free graft. Contemporary clinical dentistry 3:S240–S243. https://doi.org/10.4103/0976-237x.101105
Kulkarni MR, Thomas BS, Varghese JM, Bhat GS (2014) Platelet-rich fibrin as an adjunct to palatal wound healing after harvesting a free gingival graft: a case series. Journal of Indian Society of Periodontology 18:399–402. https://doi.org/10.4103/0972-124x.134591
Femminella B, Iaconi MC, Di Tullio M, Romano L, Sinjari B, D’Arcangelo C, De Ninis P, Paolantonio M (2016) Clinical comparison of platelet-rich fibrin and a gelatin sponge in the management of palatal wounds after epithelialized free gingival graft harvest: a randomized clinical trial. J Periodontol 87:103–113. https://doi.org/10.1902/jop.2015.150198
Ajwani H, Shetty S, Gopalakrishnan D, Kathariya R, Kulloli A, Dolas RS, Pradeep AR (2015) Comparative evaluation of platelet-rich fibrin biomaterial and open flap debridement in the treatment of two and three wall intrabony defects. Journal of international oral health : JIOH 7:32–37
di Lauro AE, Abbate D, Dell’Angelo B, Iannaccone GA, Scotto F, Sammartino G (2015) Soft tissue regeneration using leukocyte-platelet rich fibrin after exeresis of hyperplastic gingival lesions: two case reports. J Med Case Rep 9:252. https://doi.org/10.1186/s13256-015-0714-5
Miron RJ, Fujioka-Kobayashi M, Bishara M, Zhang Y, Hernandez M, Choukroun J (2016) Platelet-Rich fibrin and soft tissue wound healing: a systematic review. Tissue Eng B Rev. https://doi.org/10.1089/ten.TEB.2016.0233
Medina-Porqueres I, Alvarez-Juarez P (2015) The efficacy of platelet-rich plasma injection in the management of hip osteoarthritis: a systematic review protocol. Musculoskeletal care. https://doi.org/10.1002/msc.1115
Salamanna F, Veronesi F, Maglio M and Della Bella E (2015) New and emerging strategies in platelet-rich plasma application in musculoskeletal regenerative procedures: general overview on still open questions and outlook. 2015:846045. doi: https://doi.org/10.1155/2015/846045
Albanese A, Licata ME, Polizzi B, Campisi G (2013) Platelet-rich plasma (PRP) in dental and oral surgery: from the wound healing to bone regeneration. Immunity & ageing : I & A 10:23. https://doi.org/10.1186/1742-4933-10-23
Panda S, Doraiswamy J, Malaiappan S, Varghese SS, Del Fabbro M (2014) Additive effect of autologous platelet concentrates in treatment of intrabony defects: a systematic review and meta-analysis. J Investig Clin Dent. https://doi.org/10.1111/jicd.12117
Miron RJ, Fujioka-Kobayashi M, Hernandez M, Kandalam U, Zhang Y, Ghanaati S, Choukroun J (2017) Injectable platelet rich fibrin (i-PRF): opportunities in regenerative dentistry? Clin Oral Investig 21:2619–2627. https://doi.org/10.1007/s00784-017-2063-9
Abd El Raouf M, Wang X, Miusi S, Chai J, Mohamed AbdEl-Aal AB, Nefissa Helmy MM, Ghanaati S, Choukroun J, Choukroun E, Zhang Y and Miron RJ (2017) Injectable-platelet rich fibrin using the low speed centrifugation concept improves cartilage regeneration when compared to platelet-rich plasma. Platelets:1-9. doi: https://doi.org/10.1080/09537104.2017.1401058
Wang X, Zhang Y, Choukroun J, Ghanaati S and Miron RJ (2017) Behavior of gingival fibroblasts on titanium implant surfaces in combination with either injectable-PRF or PRP. Int J Mol Sci 18. doi: https://doi.org/10.3390/ijms18020331
Wang X, Zhang Y, Choukroun J, Ghanaati S, Miron RJ (2018) Effects of an injectable platelet-rich fibrin on osteoblast behavior and bone tissue formation in comparison to platelet-rich plasma. Platelets 29:48–55. https://doi.org/10.1080/09537104.2017.1293807
Miron RJ, Chai J, Zheng S, Feng M, Sculean A, Zhang Y (2019) A novel method for evaluating and quantifying cell types in platelet rich fibrin and an introduction to horizontal centrifugation. J Biomed Mater Res A. https://doi.org/10.1002/jbm.a.36734
Nunes CR, Roedersheimer M, Simske S, Luttges M (1995) Effect of microgravity, temperature, and concentration on fibrin and collagen assembly. Microgravity science and technology 8:125–130
Ghanaati S, Booms P, Orlowska A, Kubesch A, Lorenz J, Rutkowski J, Landes C, Sader R, Kirkpatrick C, Choukroun J (2014) Advanced platelet-rich fibrin: a new concept for cell-based tissue engineering by means of inflammatory cells. The Journal of oral implantology 40:679–689. https://doi.org/10.1563/aaid-joi-D-14-00138
Bielecki T, Dohan Ehrenfest DM, Everts PA, Wiczkowski A (2012) The role of leukocytes from L-PRP/L-PRF in wound healing and immune defense: new perspectives. Curr Pharm Biotechnol 13:1153–1162
Martin P (1997) Wound healing--aiming for perfect skin regeneration. Science 276:75–81
Barrick B, Campbell EJ, Owen CA (1999) Leukocyte proteinases in wound healing: roles in physiologic and pathologic processes. Wound Repair Regen 7:410–422
Kobayashi E, Fluckiger L, Fujioka-Kobayashi M, Sawada K, Sculean A, Schaller B, Miron RJ (2016) Comparative release of growth factors from PRP, PRF, and advanced-PRF. Clin Oral Investig 20:2353–2360. https://doi.org/10.1007/s00784-016-1719-1
Fujioka-Kobayashi M, Miron RJ, Hernandez M, Kandalam U, Zhang Y and Choukroun J (2016) Optimized platelet rich fibrin with the low speed concept: growth factor release, biocompatibility and cellular response. J Periodontol:1-17. doi: https://doi.org/10.1902/jop.2016.160443
Funding
This work was supported by funds from the National Key R&D Program of China (2018YFC1105300 to Yufeng Zhang).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflicts of interest.
Ethical approval
No ethical approval was required for this study, as the human samples were not identified.
Informed consent
For this study, informed written consent was provided to conduct the outlined experiments prior to the blood draw.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Miron, R.J., Chai, J., Zhang, P. et al. A novel method for harvesting concentrated platelet-rich fibrin (C-PRF) with a 10-fold increase in platelet and leukocyte yields. Clin Oral Invest 24, 2819–2828 (2020). https://doi.org/10.1007/s00784-019-03147-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00784-019-03147-w