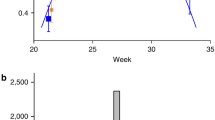
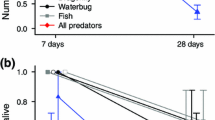
Abstract
Despite being a major selective force, predation can induce puzzling variability in anti-predator responses—from lack of predator aversion to lifelong predator-induced fear. This variability is hypothesised to result from variation in the trade-offs associated with avoiding predators. But critical information on fitness outcomes of these trade-offs associated with anti-predator behaviours is lacking. We tested this trade-off hypothesis in Aedes aegypti, by examining oviposition site selection decisions in response towards larval predation risk and comprehensively measuring the fitness implications of trade-offs of avoiding larval predators, using three fitness measures: larval survival, development time and size. In a field study, we find that adult females show a surprisingly variable response to predators, ranging from attraction to avoidance. This variation is explained by fitness outcomes of oviposition along a predation-risk gradient that we measured in the laboratory. We show that ovipositing females could gain fitness benefits from ovipositing in pools with a low density of predators, rather than in predator-free pools, as predators provide a release from negative density effects of conspecific larvae that might co-occur in a pool. Interacting selection pressures may thus explain diverse prey responses. We suggest other systems in which similarly unexpected prey behaviour is likely to occur.




Similar content being viewed by others
Data accessibility
We would be happy to provide the raw data for the analyses and results reported in this manuscript. On acceptance, we would be happy to archive the data files, R code supporting the results.
References
Achord S, Levin PS, Zabel RW (2003) Density-dependent mortality in Pacific salmon: the ghost of impacts past? Ecol Lett 6(4):335–342. https://doi.org/10.1046/j.1461-0248.2003.00438.x
Agnew P, Hide M, Sidobre C, Michalakis Y (2002) A minimalist approach to the effects of density-dependent competition on insect life-history traits. Ecol Entomol 27(4):396–402. https://doi.org/10.1046/j.1365-2311.2002.00430.x
Albeny-Simões D, Murrell EG, Elliot SL, Andrade MR, Lima E, Juliano SA, Vilela EF (2014) Attracted to the enemy: Aedes aegypti prefers oviposition sites with predator-killed conspecifics. Oecologia 175(2):481–492. https://doi.org/10.1007/s00442-014-2910-1
Alencar J, Mello CF, Barbosa LS, Gil-Santana HR, Maia DA, Marcondes CB, Silva JS (2016) Diversity of yellow fever mosquito vectors in the Atlantic Forest of Rio de Janeiro, Brazil. Rev Soc Bras Med Trop 49(3):351–356. https://doi.org/10.1590/0037-8682-0438-2015
Angelon KA, Petranka JW (2002) Chemicals of predatory mosquitofish (Gambusia affinis) influence selection of oviposition site by Culex mosquitoes. J Chem Ecol 28(4):797–806. https://doi.org/10.1023/A:1015292827514
Armbruster P, Hutchinson RA (2002) Pupal mass and wing length as indicators of fecundity in Aedes albopictus and Aedes geniculatus (Diptera: Culicidae). J Med Entomol 39(4):699–704. https://doi.org/10.1603/0022-2585-39.4.699
Bates D, Mächler M, Bolker B, Walker S (2015) Fitting linear mixed-effects models using lme4. J Stat Softw 67(1):1–48. https://doi.org/10.18637/jss.v067.i01
Belovsky GE, Laws AN, Slade JB (2011) Prey change behaviour with predation threat, but demographic effects vary with prey density: experiments with grasshoppers and birds. Ecol Lett 14(4):335–340. https://doi.org/10.1111/j.1461-0248.2011.01591.x
Blaustein L (1999) Oviposition habitat selection in response to risk of predation: consequences for populations and community structure. In: Wasser SP (ed) Evolutionary processes and theory: modern perspectives. Kluwer Academic Publishers, Dordrecht, The Netherlands, pp 441–456
Briegel H (1990) Metabolic relationship between female body size, reserves, and fecundity of Aedes aegypti. J Insect Physiol 36(3):165–172. https://doi.org/10.1016/0022-1910(90)90118-Y
Brown JS (1999) Vigilance, patch use and habitat selection: foraging under predation risk. Evol Ecol Res 1:49–71
Brown JS, Laundré JW, Gurung M (1999) The ecology of fear: optimal foraging, game theory, and trophic interactions. J Mammal 80(2):385–399. https://doi.org/10.2307/1383287
Chadee DD, Corbet PS (1990) Diel patterns of oviposition indoors of the mosquito, Aedes aegypti (L.) (Diptera: Culicidae) in Trinidad, W.I.: a preliminary study. Ann Trop Med Parasitol 84(1):79–84. https://doi.org/10.1080/00034983.1990.11812435
Chadee D, Ward R (1998) Natural habitats of Aedes aegypti in the Caribbean—a review. J Am Mosq Control Assoc 14(1):5–11
Chase JM, Abrams PA, Grover JP, Diehl S, Chesson P, Holt RD et al (2002) The interaction between predation and competition: a review and synthesis. Ecol Lett 5(2):302–315. https://doi.org/10.1046/j.1461-0248.2002.00315.x
Clements AN (1992) The biology of mosquitoes: development, nutrition and reproduction. Chapman and Hall, London
Clements AN (1999) The biology of mosquitoes: sensory reception and behaviour. CABI, Wallingford
Coleman BT, Hill RA (2014) Living in a landscape of fear: the impact of predation, resource availability and habitat structure on primate range use. Anim Behav 88:165–173. https://doi.org/10.1016/j.anbehav.2013.11.027
Colton YM, Chadee DD, Severson DW (2003) Natural skip oviposition of the mosquito Aedes aegypti indicated by codominant genetic markers. Med Vet Entomol 17:195–204. https://doi.org/10.1046/j.1365-2915.2003.00424.x
Cooper WE (2006) Dynamic risk assessment: prey rapidly adjust flight initiation distance to changes in predator approach speed. Ethology 112(9):858–864. https://doi.org/10.1111/j.1439-0310.2006.01240.x
Creel S, Christianson D (2008) Relationships between direct predation and risk effects. Tree 23(4):194–201. https://doi.org/10.1016/j.tree.2007.12.004
Creel S, Winnie J, Maxwell B, Hamlin K, Creel M (2005) Elk alter habitat selection as an antipredator response to wolves. Ecology 86(12):3387–3397. https://doi.org/10.1890/05-0032
Dill LM, Fraser AHG (1984) Risk of predation and the feeding-behavior of juvenile coho salmon (Oncorhynchus kisutch). Behav Ecol Sociobiol 16(1):65–71
Dupuch A, Magnan P, Bertolo A, Dill LM, Proulx M (2009) Does predation risk influence habitat use by northern redbelly dace Phoxinus eos at different spatial scales? J Fish Biol 74(7):1371–1382. https://doi.org/10.1111/j.1095-8649.2009.02183.x
Eitam A, Blaustein L (2004) Oviposition habitat selection by mosquitoes in response to predator (Notonecta maculata) density. Physiol Entomol 29(2):188–191. https://doi.org/10.1111/j.0307-6962.2004.0372.x
Fincke OM, Yanoviak SP, Hanschu RD (1997) Predation by odonates depresses mosquito abundance in water-filled tree holes in Panama. Oecologia 112:244–253. https://doi.org/10.1007/s004420050307
Gelman A, Weakliem D (2009) Of beauty, sex, and power: statistical challenges in estimating small effects. Am Sci 97:310–316
Gurevitch J, Morrison JA, Hedges LV (2000) The interaction between competition and predation: a meta-analysis of field experiments. Am Nat 155(4):435–453. https://doi.org/10.1086/303337
Harrington LC, Ponlawat A, Edman JD, Scott TW, Vermeylen F (2008) Influence of container size, location, and time of day on oviposition patterns of the dengue vector, Aedes aegypti, in Thailand. Vector Borne Zoonotic Dis 8(3):415–424. https://doi.org/10.1089/vbz.2007.0203
Hebblewhite M, Merrill EH (2009) Trade-offs between predation risk and forage differ between migrant strategies in a migratory ungulate. Ecology 90(12):3445–3454. https://doi.org/10.1890/08-2090.1
Heithaus M, Dill L (2006) Does tiger shark predation risk influence foraging habitat use by bottlenose dolphins at multiple spatial scales? Oikos 114(2):257–264. https://doi.org/10.1111/j.2006.0030-1299.14443.x
Hill RA, Dunbar R (1998) An evaluation of the roles of predation rate and predation risk as selective pressures on primate grouping behaviour. Behaviour 135(4):411–430. https://doi.org/10.1163/156853998793066195
Hixon MA, Jones GP (2005) Competition, predation, and density-dependent mortality in demersal marine fishes. Ecology 86(11):2847–2859. https://doi.org/10.1890/04-1455
Juliana JRS, Kotler BP, Brown JS, Mukherjee S, Bouskila A (2011) The foraging response of gerbils to a gradient of owl numbers. Evol Ecol Res 13(8):869–878
Katz MW, Abramsky Z, Kotler BP, Rosenzweig ML, Altstein O, Roth I, Klimovitsky C (2016) Comparing the non-lethal and lethal effects of predation risk on goldfish anti-predatory behavior. Isr J Ecol Evol 62(3–4):121–127. https://doi.org/10.1080/15659801.2015.1059720
Kneitel JM, Chase JM (2004) Trade-offs in community ecology: linking spatial scales and species coexistence. Ecol Lett 7(1):69–80. https://doi.org/10.1046/j.1461-0248.2003.00551.x
Kotler BP, Brown JS (1988) Environmental heterogeneity and the coexistence of desert rodents. Ann Rev Ecol Syst 19:281–307. https://doi.org/10.1146/annurev.es.19.110188.001433
Kramer WL, Mulla MS (1979) Oviposition attractants and repellents of mosquitoes: oviposition responses of Culex mosquitoes to organic infusions. Env Entomol 8(6):1111–1117. https://doi.org/10.1093/ee/8.6.1111
Laundre JW, Hernandez L, Ripple WJ (2010) The landscape of fear: ecological implications of being afraid. Open Ecol J 3(3):1–7. https://doi.org/10.2174/1874213001003030001
Lima SL (1998) Nonlethal effects in the ecology of predator-prey interactions. BioScience 48(1):25–34. https://doi.org/10.2307/1313225
Livdahl TP, Sugihara G (1984) Non-linear interactions of populations and the importance of estimating per capita rates of change. J Anim Ecol 53(2):573–580
Muriu SM, Coulson T, Mbogo CM, Godfray HCJ (2013) Larval density dependence in Anopheles gambiae s.s., the major African vector of malaria. J Anim Ecol 82(1):166–174. https://doi.org/10.2307/4535
Murphy PJ (2003) Does reproductive site choice in a neotropical frog mirror variable risks facing offspring? Ecol Monogr 73(1):45–67. https://doi.org/10.1890/0012-9615(2003)073%5b0045:drscia%5d2.0.co;2
Naniwadekar R, Mishra C, Datta A (2015) Fruit resource tracking by hornbill species at multiple scales in a tropical forest in India. J Trop Ecol 31:1–14. https://doi.org/10.1017/S0266467415000449
Okasanen L, Fretwell SD, Arruda J, Niemela P (1981) Exploitation ecosystems in gradients of primary productivity. Am Nat 118(2):240–261. https://doi.org/10.1086/283817
Paine RT (2010) Trophic cascades: predators, prey and the changing dynamics of nature. Island Press, Washington DC
Preisser EL, Bolnick DI (2008) The many faces of fear: comparing the pathways and impacts of nonconsumptive predator effects on prey populations. PLoS One 3(6):5–8. https://doi.org/10.1371/journal.pone.0002465
Preisser EL, Bolnick DI, Bernard MF (2005) Scared to death? The effect of intimidation and consumption in predator prey interactions. Ecology 86(2):501–509. https://doi.org/10.1890/04-0719
Preisser EL, Bolnick DI, Grabowski JH (2009) Resource dynamics influence the strength of non-consumptive predator effects on prey. Ecol Lett 12(4):315–323. https://doi.org/10.1111/j.1461-0248.2009.01290.x
R Core Team (2017) R: a language and environment for statistical computing. R Foundation for Statistical Computing, Vienna
Relyea RA (2001) The relationship between predation risk and antipredator responses in larval anurans. Ecology 82(2):541–554. https://doi.org/10.1890/0012-9658(2001)082%5b0541:trbpra%5d2.0.co;2
Rieger JF, Binckley CA, Resetarits WJ Jr (2004) Larval performance and oviposition site preference along a predation gradient. Ecology 85(8):2094–2099. https://doi.org/10.1890/04-0156
Sih A (1992) Prey uncertainty and the balancing of antipredator and feeding needs. Am Nat 139(5):1052–1069. https://doi.org/10.1086/285372
Sih A, Crowley P, McPeek M, Petranka J, Strohmeier K (1985) Predation competition, and prey communities: a review of field experiments. Ann Rev Ecol Syst 16:269–311. https://doi.org/10.1146/annurev.es.16.110185.001413
Silberbush A, Blaustein L (2011) Mosquito females quantify risk of predation to their progeny when selecting an oviposition site. Func Ecol 25(5):1091–1095. https://doi.org/10.1111/j.1365-2435.2011.01873.x
Southwood TRE, Murdie G, Yasuno M, Tonn RJ, Reader PM (1972) Studies on the life budget of Aedes aegypti in Wat Samphaya, Bangkok, Thailand. Bull World Health Organ 46:211–226
Sparks JT, Bohbot JD, Dickens JC (2014) The genetics of chemoreception in the labella and tarsi of Aedes aegypti. Insect Biochem Mol Bio 48:8–16. https://doi.org/10.1016/j.ibmb.2014.02.004
Spencer M, Blaustein L, Cohen JE (2002) Oviposition habitat selection by mosquitoes (Culiseta longiareolata) and consequences for population size. Ecology 83(3):669–679. https://doi.org/10.1890/0012-9658(2002)083%5b0669:ohsbmc%5d2.0.co;2
Tun-Lin W, Burkot TR, Kay BH (2000) Effects of temperature and larval diet on development rates and survival of the dengue vector Aedes aegypti in north Queensland, Australia. Med Vet Entomol 14:31–37. https://doi.org/10.1046/j.1365-2915.2000.00207.x
Verdolin JL (2006) Meta-analysis of foraging and predation risk trade-offs in terrestrial systems. Behav Ecol Sociobiol 60(4):457–464. https://doi.org/10.1007/s00265-006-0172-6
Vonesh JR, Blaustein L (2010) Predator-induced shifts in mosquito oviposition site selection: a meta-analysis and implications for vector control. Isr J Ecol Evol 56(3–4):123–139. https://doi.org/10.1560/IJEE.56.3-4.263
Walsh MR, Reznick DN (2008) Interactions between the direct and indirect effects of predators determine life history evolution in a killifish. PNAS 105(2):594–599. https://doi.org/10.1073/pnas.0710051105
Walsh RK, Facchinelli L, Ramsey JM, Bond JG, Gould F (2011) Assessing the impact of density dependence in field populations of Aedes aegypti. J Vector Ecol 36(2):300–307. https://doi.org/10.1111/j.1948-7134.2011.00170.x
Winnie JJ, Creel S (2017) The many effects of carnivores on their prey and their implications for trophic cascades, and ecosystem structure and function. Food Webs 12:88–94. https://doi.org/10.1016/j.fooweb.2016.09.002
Wong J, Stoddard ST, Astete H, Morrison AC, Scott TW (2011) Oviposition site selection by the dengue vector Aedes aegypti and its implications for dengue control. PLoS Negl Trop Dis 5:4. https://doi.org/10.1371/journal.pntd.0001015
Zandona E, Auer SK, Kilham SS, Howard JL, Lopez-Sepulcre A, Connor MPO et al (2011) Diet quality and prey selectivity correlate with life histories and predation regime in Trinidadian guppies. Func Ecol 25(5):964–973. https://doi.org/10.1111/j.1365-2435.2011.01865.x
Acknowledgements
We thank Mahalakshmi and Rajashree for maintenance of mosquito colony; Karthikeyan Chandrasegaran for discussions; Rishi Valley School and Indian Institute of Science for providing infrastructure. The work was supported by Department of Biotechnology—Indian Institute of Science (DBT-IISc) partnership Grant, Department of Science and Technology—Fund for Improvement of S&T infrastructure (DST-FIST) Grant and Ministry of Human Resource Development—Indian Institute of Science for graduate scholarship. Funding was provided by Department of Biotechnology, Ministry of Science and Technology.
Author information
Authors and Affiliations
Contributions
MS and KI designed the study. MS collected the data and wrote the first draft of the manuscript. MS, SQ, VG, and KI contributed substantially to revisions.
Corresponding author
Additional information
Communicated by Aaron J. Wirsing.
We wish to submit this work for the Highlighted student research category. This work was done as part of the Ph.D. thesis of the lead author. In this study, we take a rare approach of measuring fitness pay-offs in multiple currencies to explain the puzzle of how predation risk can induce variability in anti-predator responses—from attraction to aversion towards predators.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Sharma, M., Quader, S., Guttal, V. et al. The enemy of my enemy: multiple interacting selection pressures lead to unexpected anti-predator responses. Oecologia 192, 1–12 (2020). https://doi.org/10.1007/s00442-019-04552-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00442-019-04552-4