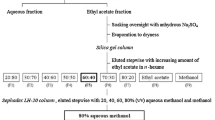
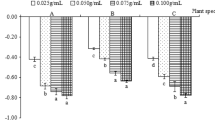
Abstract
Erythrina fusca is a dominant species in the Brazilian Pantanal. We hypothesized that E. fusca possess allelopathic potential and we evaluated effects of extracts on germination and development of Lactuca sativa, a bioindicator species. We tested the effect of leaves, bark, roots, and seeds extracts of E. fusca on germination and speed index, using high, moderate and low concentration (0.2, 1 and 5 mg mL–1). To evaluate effects on development, we subjected seedlings of L. sativa to the same treatments and measured root and aerial part length. High concentration of extracts reduced L. sativa germination; leaves extract caused the maximum reduction on germination of L. sativa, similar to 2,4-Dichlorophenoxyacetic acid (2,4-D); this extract has flavonoids and saponins as main compounds, classes that also occur in the bark and roots extracts in lower concentrations; bark and roots (5 mg mL–1), leaves and roots (1 mg mL–1) decreased these traits as well, but in lower magnitude. A significant reduction in root length was induced by highest concentration of all extracts (5 mg mL–1); the results suggest that erythrinic alkaloids should interfere in the root length once the seeds accumulate almost exclusively this class of compounds. Our results showed that all parts of E. fusca had adverse effects on germination or development of L. sativa, showing that different class of compounds secondary metabolites is involved in this activity. Possibly, this phytotoxicity influences monodominance of E. fusca in Pantanal, but studies are essential to evaluate effects of it on other native species.




Similar content being viewed by others
References
Aguilera N, Guedes LM, Becerra J, Baeza C, Hernández V (2015) Morphological effects at radicle level by direct contact of invasive Acacia dealbata Link. Flora 215:54–59. https://doi.org/10.1016/j.flora.2015.07.007
Alvares CA, Stape JL, Sentelhas PC, Gonçalves JLM, Sparovek G (2013) Köppen’s climate classification map for Brazil. Meteorol Z 22(6):711–728. https://doi.org/10.1127/0941-2948/2013/0507
Bridson DM, Forman L (2004) The herbarium handbook. Royal Botanic Gardens, Kew
Brunetti AE, Carnevale Neto F, Vera MC, Taboada C, Pavarini DP, Bauermeister A, Lopes NP (2018) An integrative omics perspective for the analysis of chemical signals in ecological interactions. Chem Soc Rev 47:1574–1591. https://doi.org/10.1039/c7cs00368d
Cândido ACS, Schmidt V, Laura VA, Faccenda O, Hess SC, Simionatto E, Peres MTLP (2010) Potencial alelopático da parte aérea de Senna occidentalis (L.) Link (Fabaceae, Caesalpinioideae): bioensaios em laboratório. Acta Bot Bras 24:235–242. https://doi.org/10.1590/S0102-33062010000100025
Carvalho MSS, Andrade-Vieira LF, Santos FE, Correa FF, Cardoso MG, Vilela LR (2019) Allelopathic potential and phytochemical screening of ethanolic extracts from five species of Amaranthus spp. in the plant model Lactuca sativa. Sci Hortic 245:90–98. https://doi.org/10.1016/j.scienta.2018.10.001
Centenaro C, Corrêa LGP, Karas MJ, Virtuoso S, Dias FG, Miguel OG, Miguel MD (2009) Contribuição ao estudo alelopático de Erythrina velutina Willd. Fabaceae. Rev Bras Farmacogn 19:304–308. https://doi.org/10.1590/S0102-695X2009000200021
Connell JH, Lowman MD (1989) Low-diversity tropical rain forests: some possible mechanisms for their existence. Am Nat 134:88–119. https://doi.org/10.1086/284967
Dao TT, Nguyen PH, Thuong PT, Kang KW, Na W, Ndinteh DT, Mbafor JT, Oh WK (2009) Pterocarpans with inhibitory effects on protein tyrosine phosphatase 1B from Erythrina lysistemon Hutch. Phytochemistry 70:2053–2057. https://doi.org/10.1016/j.phytochem.2009.09.012
de Bertoldi C, de Leo M, Braca A, Ercoli L (2009) Bioassay-guided isolation of allelochemicals from Avena sativa L.: allelopathic potential of flavone C-glycosides. Chemoecology 19(3):169–176. https://doi.org/10.1007/s00049-009-0019-5
Fernandes PEN, Silva SJ, Assis PLF, Alves BCH, Nascimento FTR (2018) Allelopathic effects of aqueous extract of leaves and roots of Luetzelburgia auriculata (Allemo) Ducke on seeds germination and initial growth of lettuce (Lactuca sativa L.). Afr J Agric Res 13(13):635–643. https://doi.org/10.5897/AJAR2016.11364
Ferreira AG, Aquila MEA (2000) Alelopatia: uma área emergente da ecofisiologia. Rev Bras Fisiol Veg 12:175–204
Frota AVB, Ikeda-Castrillon SK, Kantek DLZ, Silva CJ (2017) Macrohabitats da Estação Ecológica de Taiamã, no contexto da Área Úmida Pantanal mato-grossense, Brasil. Bol Mus Para Emílio Goeldi Cien Nat 12:239–254
García-Mateos R, Peña-Valdivia CB, Soto-Hernández M (2002) Phytotoxicity of crude alkaloid fractions from Erythrina americana. Rev Soc QuíM Méx 46(1):04–09
Harborne JB (1996) Plant secondary metabolism. In: Crawley MJ (ed) Plant ecology, 2nd ed. Blackwell Publishing, Oxford, p 132–155
Hart TB, Hart JA, Murphy PG (1989) Monodominant and species-rich forests of the humid tropics: causes for their co-occurrence. Am Nat 133:613–633. https://doi.org/10.1086/284941
Hart TB (1990) Monospecific dominance in tropical rain forests. Trends Ecol Evol 5:6–11. https://doi.org/10.1016/0169-5347(90)90005-X
Hebets EA, Papaj DR (2005) Complex signal function: developing a framework of testable hypotheses. Behav Ecol Sociobiol 57:197–214. https://doi.org/10.1007/s00265-004-0865-7
Hooper AM, Tsanuo MK, Chamberlain K, Tittcomb K, Scholes J, Hassanali A, Khan ZR, Pickett JA (2010) Isoschaftoside, a C-glycosylflavonoid from Desmodium uncinatum root exudate, is an allelochemical against the development of Striga. Phytochemistry 71(8-9):904–908. https://doi.org/10.1016/j.phytochem.2010.02.015
Hooper AM, Caulfield JC, Hao B, Pickett JA, Midega CAO, Khan ZR (2015) Isolation and identification of Desmodium root exudates from drought tolerant species used as intercrops against Striga hermonthica. Phytochemistry 117:380–387. https://doi.org/10.1016/j.phytochem.2015.06.026
Id VLE, Costa BV, Mignoni DSB, Veronesi MB, Simões K, Braga MR, Santos Junior NA (2015) Phytotoxic effect of Sesbania virgata (Cav.) Pers. on seeds of agronomic and forestry species. J Res 26:339–346. https://doi.org/10.1007/s11676-015-0026-z
Innok P, Rukachaisirikul T, Suksamrarn A (2009) Flavanoids and pterocarpans from the bark of Erythrina fusca. Chem Pharm Bull 57:993–996. https://doi.org/10.1248/cpb.57.993
Innok P, Rukachaisirikul T, Souwalak P, Suksamrarn A (2010) Fuscacarpans A–C, new pterocarpans from the stems of Erythrina fusca. Fitoterapia 81:518–523. https://doi.org/10.1016/j.fitote.2010.01.009
INMET-Instituto Nacional de Meteorologia. Estações Meteorológica de Observação de Superfície Automática. http://www.inmet.gov.br/portal/index.php?r=estacoes/estacoesAutomaticas. Accessed 15 March 2019
Juma BF, Majinda RRT (2004) Erythrinaline alkaloids from the flowers and pods of Erythrina lysistemon and their DPPH radical scavenging properties. Phytochemistry 65:1397–1404. https://doi.org/10.1016/j.phytochem.2004.04.029
Kato-Noguchi H (2003) Isolation and identification of an allelopathic substance in Pisum sativum. Phytochemistry 62(7):1141–1144. https://doi.org/10.1016/S0031-9422(02)00673-8
Labouriau LG, Valadares MEB (1976) On the germination of seeds Calotropis procera (Ait.) Ait.f. Acad Bras Ciênc 48:263–284. https://doi.org/10.1590/S0001-37652013000300013
Lorenzi H (1998) Árvores Brasileiras: manual de identificação e cultivo de plantas arbóreas do Brasil. Vol. 2. Editora Plantarum, Nova Odessa
Macías FA, Oliveros-Bastidas A, Marín D, Carrera C, Chinchilla N, Molinillo JMG (2008) Plant biocommunicators: their phytotoxicity, degradation studies and potential use as herbicide models. Phytochem Rev 7:179–194. https://doi.org/10.1007/s11101-007-9062-4
Maguire JD (1962) Speed of germination-aid in selection and evaluation for seedling emergence and vigor. Crop Sci 2:176–177. https://doi.org/10.2135/cropsci1962.0011183X000200020033x
Mierziak J, Kostyn K, Kulma A (2014) Flavonoids as important molecules of plant interactions with the environment. Molecules 19(10):16240–16265. https://doi.org/10.3390/molecules191016240
Mori SA, Mattos-Silva LA, Lisboa, G, Coradin L (1989) Manual de manejo do herbário fanerogâmico. CEPLAC, Ilhéus
Oliveira AK, Coelho MFB, Maia SSS, Diogenes FEP, Medeiros Filho S (2012) Alelopatia de extratos de diferentes órgãos de mulungu na germinação de alface. Hortic Bras 30:480–483. https://doi.org/10.1590/S0102-05362012000300020
Oliveira AK, Coelho MFB, Maia SSS, Diogenes FEP, Medeiros Filho S (2013) Potencial alelopático do extrato aquoso de sementes de mulungu (Erythrina velutina Willd.). Rev Bras Ciênc Agrar 8:252–257. https://doi.org/10.5039/agraria.v8i2a1899
Oliveira DC, Soares GLG, Isaias RMS (2008) Phytotoxicity of the extracts of Lonchocarpus muehlbergianus Hassl. (Fabaceae) leaflets and galls on seed germination and early development of lettuce. Acta Bot Bras 22:1095–1100. https://doi.org/10.1590/S0102-33062008000400020
Ooka JK, Owens DK (2018) Allelopathy in tropical and subtropical species. Phytochem Rev 17:1225–1237. https://doi.org/10.1007/s11101-018-9596-7
Palm CA, Sanchez PA (1990) Decomposition and nutrient release patterns of the leaves of three tropical legumes. Biotropica 22:330–338. https://doi.org/10.2307/2388550
Pérez AJ, Hassan EM, Pecio L, Omer EA, Kucinska M, Murias M, Stochmal A (2015) Triterpenoid saponins and C-glycosyl flavones from stem bark of Erythrina abyssinica Lam and their cytotoxic effects. Phytochem Lett 13:59–67. https://doi.org/10.1016/j.phytol.2015.05.013
Pott A, Oliveira AKM, Damasceno-Junior GA, Silva JSV (2011) Plant diversity of the Pantanal wetland. Braz J Biol 71:265–273. https://doi.org/10.1590/S1519-69842011000200005
R Development Core Team. R: a language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria ISBN 3-900051-07-0. http://www.R-project.org/. Accessed 18 Feb 2019
Rice EL (1984) Allelopathy. Academic Press, New York
Russo RO, Baguinon NT (1997) Erythrina fusca Loureiro. In: Faridah Hanum I, Van der Maesen LJG (eds) Plant resources of South-East Asia No. 11: auxiliary plants. Backhuys Publisher, Leiden, pp 121–123
Scavo A, Restuccia A, Mauromicale G (2018) Allelopathy: principles and basic aspects for agroecosystem control. In: Gaba S, Smith B, Lichtfouse E (eds) Sustainable agriculture reviews 28. Springer, Cham, pp 47–101
Scrivanti LR, Anton AM (2019) Allelopathic effect of endemic South American Bothriochloa species (Poaceae: Andropogonaceae). J Essent Oil Res 31(3):247–254. https://doi.org/10.1080/10412905.2018.1563569
Silva, MC, Araujo ECG, Silva TC, Araújo AB, Lins TRS, Leão SLM, Lima TV (2019) Alelopatic effects of Tectona grandis LF in the germination and initial development of lettuce (Lactuca sativa L.). J Agric Sci 11(1). https://doi.org/10.5539/jas.v11n1p382
Soares GLG, Scalon VR, Pereira TO, Vieira DA (2002) Potencial alelopático do extrato aquoso de folhas de algumas leguminosas arbóreas brasileiras. Floresta Ambient 9:119–126
Stevenson PC, Nicolson SW, Wright GA (2017) Plant secondary metabolites in nectar: impacts on pollinators and ecological functions. Funct Ecol 31:65–75. https://doi.org/10.1111/1365-2435.12761
Tanaka H, Oh-Uchi T, Etoh H, Shimizu H, Tateishi Y (2002) Isoflavonoids from the roots of Erythrina poeppigiana. Phytochem 60:789–794. https://doi.org/10.1016/S0031-9422(02)00164-4
Tsanuo MK, Hassanali A, Hooper AM, Khan Z, Kaberia F, Pickett JA, Wadhams LJ (2003) Isoflavanones from the allelopathic aqueous root exudate of Desmodium uncinatum. Phytochemistry 64(1):265–273. https://doi.org/10.1016/S0031-9422(03)00324-8
Wang C, Xiao H, Zhao L, Liu J, W L, Zhang F, Shi Y, Du D (2016) The allelopathic effects of invasive plant Solidago canadensis on seed germination and growth of Lactuca sativa enhanced by different types of acid deposition. Ecotoxicology 25:555–562. https://doi.org/10.1007/s10646-016-1614-1
Wang C, Wu B, Jiang K (2019) Allelopathic effects of Canada goldenrod leaf extracts on the seed germination and seedling growth of lettuce reinforced under salt stress. Ecotoxicology 28(1):103–116. https://doi.org/10.1007/s10646-018-2004-7
Weston LA, Mathesius U (2013) Flavonoids: their structure, biosynthesis and role in the rhizosphere, including allelopathy. J Chem Ecol 39(2):283–297. https://doi.org/10.1007/s10886-013-0248-5
Wink M, Latz-Brüning B (1995) Allelopathic properties of alkaloids and other natural products: possible modes of action. In: Inderjit Dakshini KMM, Einhellig FA (eds) Allelopathy: organisms, processes and applications. American Chemical Society Symposium Series, Washington, pp 117–126
Wu AP, Yu H, Gao SQ, Huang ZY, He WM, Miao SL, Dong M (2009) Differential belowground allelopathic effects of leaf and root of Mikania micrantha. Trees 23(1):11–17. https://doi.org/10.1007/s00468-008-0249-0
Yenesew A, Irungu B, Derese S, Midiwo JO, Heydenreich M, Peter MG (2003) Two prenylated flavonoids from the stem bark of Erythrina burttii. Phytochem 63:445–448. https://doi.org/10.1016/S0031-9422(03)00209-7
Acknowledgements
We thank Instituto Chico Mendes de Conservação da Biodiversidade (ICMBio) for support. Universidade Federal de Mato Grosso do Sul (UFMS) and Programa de Pós-Graduação em Ecologia e Conservação (PPGEC) for financial support. We thank Coordenação de Aperfeiçoamento de Pessoal de Nível Superior (CAPES), which provided scholarships to DG. This work was supported by Instituto Nacional de Ciência e Tecnologia em Áreas Úmidas (INAU) funded by Conselho Nacional de Desenvolvimento Científico e Tecnológico and Ministério da Ciência, Tecnologia e Inovação (CNPq no. 573990/2008-5). We thank Êmelli LF de Souza for the help during the experiments. We also thank the anonymous reviewers for their important contributions during the paper submission process.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Informed consent
Informed consent was obtained from all individuals included in the study.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
About this article
Cite this article
Gris, D., Boaretto, A.G., Marques, M.R. et al. Secondary metabolites that could contribute to the monodominance of Erythrina fusca in the Brazilian Pantanal. Ecotoxicology 28, 1232–1240 (2019). https://doi.org/10.1007/s10646-019-02133-y
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10646-019-02133-y