Abstract
Passiflora edulis, the yellow passion fruit, is the main crop from the Passiflora genus, which comprises 525 species with its diversity center in South America. Genetic maps and a BAC (bacterial artificial chromosome) genomic library are available, but the nine chromosome pairs of similar size and morphology (2n = 18) hamper chromosome identification, leading to different proposed karyotypes. Thus, the aim of this study was to establish chromosome-specific markers for the yellow passion fruit using single-copy and repetitive sequences as probes in fluorescent in situ hybridizations (FISH) to allow chromosome identification and future integration with whole genome data. Thirty-six BAC clones harboring genes and three retrotransposons (Ty1-copy, Ty3-gypsy, and LINE) were selected. Twelve BACs exhibited a dispersed pattern similar to that revealed by retroelements, and one exhibited subtelomeric distribution. Twelve clones showed unique signals in terminal or subterminal regions of the chromosomes, allowing their genes to be anchored to six chromosome pairs that can be identified with single-copy markers. The markers developed herein will provide an important tool for genomic and evolutionary studies in the Passiflora genus.




Similar content being viewed by others
Change history
19 May 2020
The article “Identification of passion fruit (<Emphasis Type="Italic">Passiflora edulis</Emphasis>) chromosomes using BAC-FISH,” written by Mariela A. Sader, Yhanndra Dias, Zirlane P. da Costa, Carla Munhoz, Helen Penha, Hélène Bergès, Maria Lucia C. Vieira, and Andrea Pedrosa-Harand, was originally published with incorrect values in Figure 4.
Abbreviations
- BACs:
-
Bacterial artificial chromosomes
- FISH:
-
Fluorescent in situ hybridizations
- NORs:
-
Active nucleolar organizer regions
- CMA:
-
Chromomycin A3
- DAPI:
-
4′,6-Diamidino-2-phenlindol
- rDNA:
-
Ribosomal DNA
- TEs:
-
Transposable elements
- SSRs:
-
Microsatellite sequences
- PCR:
-
Polymerase chain reaction
- SSC:
-
Saline–sodium citrate buffer
- SDS:
-
Sodium dodecyl sulfate
- Cy3-dUTP:
-
5-Amino-propargyl-2′-deoxyuridine 5′-triphosphate coupled to red cyanine fluorescent dye
- Alexa:
-
Alexa Fluor 488
- FITC:
-
Fluorescein isothiocyanate
- BSA:
-
Bovine serum albumin
- LINE:
-
Long interspersed nuclear element
References
Araya S, Martins AM, Junqueira NT, Costa AM, Faleiro FG, Ferreira ME (2017) Microsatellite marker development by partial sequencing of the sour passion fruit genome (Passiflora edulis Sims). BMC Genomics 18:549. https://doi.org/10.1186/s12864-019-5576-6
Bennetzen JL, Wang H (2014) The contributions of transposable elements to the structure, function, and evolution of plant genomes. Annu Rev Plant Biol 65:505–530. https://doi.org/10.1146/annurev-arplant-050213-035811
Bernacci LC, Cervi AC, Milward-de-Azevedo MA, Nunes TS, Imig DC, Mezzonato AC (2015) Passifloraceae in Lista de Espécies da Flora do Brasil. Jardim Botânico do Rio de Janeiro. Disponível em: <http://floradobrasil.jbrj.gov.br/jabot/floradobrasil/FB12506>. Accessed August 2017
Cabral JS, Felix LP, Guerra M (2006) Heterochromatin diversity and its co-localization with 5S and 45S rDNA sites in chromosomes of four Maxillaria species (Orchidaceae). Genet Mol Biol 29:659–664. https://doi.org/10.1590/S1415-47572006000400015
Carneiro MS, Camargo LEA, Coelho ASG, Vencovsky R, Júnior RPL, Stenzel NMC, Vieira MLC (2002) RAPD-based genetic linkage maps of yellow passion fruit (Passiflora edulis Sims. f. flavicarpa Deg.). Genome 45:670–678. https://doi.org/10.1139/g02-035
Cheng ZJ, Murata M (2003) A centromeric tandem repeat family originating from a part of Ty3/gypsy-retroelement in wheat and its relatives. Genetics 164:665–672
Cheng Z, Buell CR, Wing RA, Gu M, Jiang J (2001) Toward a cytological characterization of the rice genome. Genome Res 11:2133–2141. https://doi.org/10.1101/gr.194601
Costa ZP (2018) Estudos genômicos em Passiflora edulis (Passifloraceae). Tese, Escola Superior de Agricultura “Luiz de Queiroz”, Universidade de São Paulo
Cuco SM, Vieira MLC, Mondin M, Aguiar-Perecin MLR (2005) Comparative karyotype analysis of three Passiflora L. species and cytogenetic characterization of somatic hybrids. Caryologia 58:220–228. https://doi.org/10.1080/00087114.2005.10589454
De Carvalho CR, Saraiva LS (1993) An air drying technique for maize chromosomes without enzymatic maceration. Biotech Histochem 68:142–145. https://doi.org/10.3109/10520299309104684
Dhar MK, Fuchs J, Houben A (2009) Distribution of eu and heterochromatin in Plantago ovata. Cytogenet Genome Res 125:235–240. https://doi.org/10.1159/000230007
Dong F, Song J, Naess SK, Helgeson JP, Gebhardt C, Jiang J (2000) Development and applications of a set of chromosome-specific cytogenetic DNA markers in potato. Theor Appl Genet 101:1001–1007. https://doi.org/10.1007/s001220051573
Doyle JJ, Doyle JL (1987) A rapid DNA isolation procedure for small quantities of fresh leaf tissue. Phytochem Bull 19:11–15
Feuillet C, MacDougal JM (2003) A new infrageneric classification of Passiflora L. (Passifloraceae). Passiflora 13:34–38
Fonsêca A, Ferreira J, dos Santos TRB, Mosiolek M, Bellucci E, Kami J, Gepts P, Geffroy V, Schweizer D, dos Santos KGB, Pedrosa-Harand A (2010) Cytogenetic map of common bean (Phaseolus vulgaris L.). Chromosom Res 18:487–502. https://doi.org/10.1007/s10577-010-9129-8
Gerlach WL, Bedbrook JR (1979) Cloning and characterization of ribosomal RNA genes from wheat and barley. Nucleic Acids Res 7:1869–1885
Guerra M (1986) Reviewing the chromosome nomenclature of Levan et al. Braz J Genet 9:741–743
Hansen AK, Gilbert LE, Simpson BB, Downie SR, Cervi AC, Jansen RK (2006) Phylogenetic relationships and chromosome number evolution in Passiflora. Syst Bot 31:138–150. https://doi.org/10.1600/036364406775971769
Heslop-Harrison JS, Harrison GE, Leitch IJ (1992) Reprobing of DNA: DNA in situ hybridization preparations. Trends Genet 8:372–373. https://doi.org/10.1016/0168-9525(92)90287-E
Jiang J, Gill BS (2006) Current status and the future of fluorescence in situ hybridization (FISH) in plant genome research. Genome 49:1057–1068. https://doi.org/10.1139/g06-076
Judd WS, Campbell CS, Kellogg EA, Stevens PF, Donoghue MJ (2016) Plant systematics: a phylogenetic approach, 4th ed. Sinauer Associates, Sunderland
Koumbaris GL, Bass HW (2003) A new single-locus cytogenetic mapping system for maize (Zea mays L.): overcoming FISH detection limits with marker-selected sorghum (S. propinquum L.) BAC clones. Plant J 35:647–359. https://doi.org/10.1046/j.1365-313X.2003.01829.x
Krosnick SE, Ford AJ, Freudenstein JV (2009) Taxonomic revision of Passiflora subgenus Tetrapathea including the monotypic genera Hollrungia and Tetrapathea (Passifloraceae), and a new species of Passiflora. Syst Bot 34:375–385. https://doi.org/10.1600/036364409788606343
Lopes R, Lopes MTG, Carneiro M et al (2006) Linkage and mapping of resistance genes to Xanthomonas axonopodis pv. Passiflorae in yellow passion fruit. Genome 49:17–29. https://doi.org/10.1139/g05-081
Lysak MA, Berr A, Pecinka A, Schmidt R, McBreen K, Schubert I (2006) Mechanisms of chromosome number reduction in Arabidopsis thaliana and related Brassicaceae species. Proc Natl Acad Sci 103:5224–5229. https://doi.org/10.1073/pnas.0510791103
Mayeda LY (1997) Estudo citogenético em dez taxons do gênero Passiflora L. (Passifloraceae). Dissertação, Escola Superior de Agricultura “Luiz de Queiroz”, Universidade de São Paulo
Melo NF, Guerra M (2003) Variability of the 5S and 45S rDNA sites in Passiflora L. species with distinct base chromosome numbers. Ann Bot 92:309–316. https://doi.org/10.1093/aob/mcg138
Melo NF, Cervi AC, Guerra M (2001) Karyology and cytotaxonomy of the genus Passiflora L. (Passifloraceae). Plant Syst Evol 226:69–84. https://doi.org/10.1007/s006060170074
Men AE, Meksem K, Kassem MA, Lohar D, Stiller J, Lightfoot D, Gresshoff PM (2001) A bacterial artificial chromosome library of Lotus japonicus constructed in an Agrobacterium tumefaciens-transformable vector. Mol Plant-Microbe Interact 14:422–425. https://doi.org/10.1094/MPMI.2001.14.3.422
Munhoz CF, Santos AA, Arenhart RA, Santini L, Monteiro-Vitorello CB, Vieira MLC (2015) Analysis of plant gene expression during passion fruit–Xanthomonas axonopodis interaction implicates lipoxygenase 2 in host defence. Ann Appl Biol 167:135–155. https://doi.org/10.1111/aab.12215
Munhoz CF, Costa ZP, Cauz-Santos LA, Reátegui ACE, Rodde N, Cauet S, Dornelas MC, Leroy P, Varani AM, Bergès H, Vieira MLC (2018) A gene-rich fraction analysis of the Passiflora edulis genome reveals highly conserved microsyntenic regions with two related Malpighiales species. Sci Rep 8:13024. https://doi.org/10.1038/s41598-018-31330-8
Muschner VC, Zamberlan PM, Bonatto SL, Freitas LB (2012) Phylogeny, biogeography and divergence times in Passiflora (Passifloraceae). Genet Mol Biol 35:1036–1043. https://doi.org/10.1590/S1415-47572012000600019
Nagaki K, Neumann P, Zhang D, Ouyang S, Buell CR, Cheng Z, Jiang J (2004) Structure, divergence, and distribution of the CRR centromeric retrotransposon family in rice. Mol Biol Evol 22:845–855. https://doi.org/10.1093/molbev/msi069
Oliveira AM (1996) Reprodução e citogenética de espécies de Passiflora. Tese, Universidade Estadual Paulista São José do Rio Preto
Oliveira EJ, Vieira MLC, Garcia AAF, Munhoz CF, Margarido GRA, Consoli L, Matta FP, Moraes MC, Zucchi MI, Fungaro MHP (2008) An integrated molecular map of yellow passion fruit based on simultaneous maximum-likelihood estimation of linkage and linkage phases. J Am Soc Hortic Sci 133:35–41. https://doi.org/10.21273/JASHS.133.1.35
Paesold S, Borchardt D, Schmidt T, Dechyeva D (2012) A sugar beet (Beta vulgaris L.) reference FISH karyotype for chromosome and chromosome-arm identification, integration of genetic linkage groups and analysis of major repeat family distribution. Plant J 72:600–611. https://doi.org/10.1111/j.1365-313X.2012.05102.x
Pamponét VCC, Souza MM, Silva GS, Micheli F, de Melo CAF, de Oliveira SG, Costa EA, Corrêa RX (2019) Low coverage sequencing for repetitive DNA analysis in Passiflora edulis Sims: citogenomic characterization of transposable elements and satellite DNA. BMC Genomics 20:262. https://doi.org/10.1186/s12864-019-5576-6
Pedrosa A, Sandal N, Stougaard J, Schweizer D, Bachmair A (2002) Chromosomal map of the model legume Lotus japonicus. Genetics 161:1661–1672
Peterson DG, Tomkins JP, Frisch DA, Wing RA, Paterson AH (2000) Construction of plant bacterial artificial chromosome (BAC) libraries: an illustrated guide. J Agric Genomics 5:1–100
Praça MM, Carvalho CR, Francismar CM, Correa MMA (2008) Morphological aspects of Passiflora edulis f. flavicarpa chromosomes using acridine orange banding and rDNA-FISH tools. Caryologia 61:154–159. https://doi.org/10.1080/00087114.2008.10589623
Rozen S, Skaletsky H (2000) Primer3 on the WWW for general users and for biologist programmers. Methods Mol Biol 132:365–386
Rudnicki M, Silveira MM, Pereira TV, Oliveira MR, Reginatto FH, Dal-Pizzol F, Moreira JCF (2007) Protective effects of Passiflora alata extract pretreatment on carbon tetrachloride induced oxidative damage in rats. Food Chem Toxicol 45:656–661. https://doi.org/10.1016/j.fct.2006.10.022
Santos AA, Penha HA, Bellec A, Munhoz C, Pedrosa-Harand A, Bergès H, Vieira ML (2014) Begin at the beginning: a BAC-end view of the passion fruit (Passiflora) genome. BMC Genomics 15:816. https://doi.org/10.1186/1471-2164-15-816
Soares-Scott, MD (1998) Caracterização citogenetica de algumas especies e hibridos interespecificos de Passiflora Dissertação, Universidade Estadual de Campinas. Disponível em: <http://www.repositorio.unicamp.br/handle/REPOSIP/317970>. Accessed 23 July 2018
Soares-Scott MD, Meletti LMM, Bernacci LC, Passos IRS (2005) Citogenética clássica e molecular em passifloras. In: Maracujá: Germoplasma e Melhoramento Genético; Faleiro, F.G., Junqueira, N.T.V., Braga, M.F., eds.; Embrapa Cerrados: Planaltina, Brazil, pp. 210–240
Souza MM, Palomino G, Pereira TNS, Pereira MG, Viana AP (2004) Flow cytometric analysis of genome size variation in some Passiflora species. Hereditas 141:31–38. https://doi.org/10.1111/j.1601-5223.2004.01739.x
Stack SM, Royer SM, Shearer LA, Chang SB, Giovannoni JJ, Westfall DH, White RA, Anderson LK (2009) Role of fluorescence in situ hybridization in sequencing the tomato genome. Cytogenet Genome Res 124:339–350. https://doi.org/10.1159/000218137
Ulmer T, MacDougal JM (2004) Passiflora: passionflowers of the world. Timber Press, Portland-Cambridge, 430
Viana AJC, Souza MM (2012) Comparative cytogenetics between the species Passiflora edulis and Passiflora cacaoensis. Plant Biol 14:820–827. https://doi.org/10.1111/j.1438-8677.2011.00557.x
Wang K, Guan B, Guo W (2008) Completely distinguishing individual A-genome chromosomes and their karyotyping analysis by multiple bacterial artificial chromosome fluorescence in-situ hybridization. Genetics 178:1117–1122. https://doi.org/10.1534/genetics.107.083576
Wu C, Sun S, Nimmakayala P, Santos FA, Meksem K, Springman R, Ding K, Lightfoot DA, Zhang HB (2004) A BAC- and BIBAC-based physical map of the soybean genome. Genome Res 14:319–326. https://doi.org/10.1101/gr.1405004
Yotoko KSC, Dornelas MC, Togni PD, Fonseca TC, Salzano FM, Bonatto SL, Freitas LB (2011) Does variation in genome sizes reflect adaptive or neutral processes? New clues from Passiflora. PLoS One 6:e18212. https://doi.org/10.1371/journal.pone.0018212
Acknowledgments
We thank Mr. Steve Simmons for proofreading the manuscript.
Funding
The authors received financial support from CNPq (Conselho Nacional de Desenvolvimento Científico e Tecnológico). Scholarships from CNPq (Conselho Nacional de Desenvolvimento Científico e Tecnológico) were awarded to MA Sader, ZP Costa, and A. Pedrosa-Harand. This study was supported in part by the Coordenacão de Aperfeicoamento de Pessoal de Nível Superior - Brasil (CAPES, Finance Code 001), and Fundação de Amparo à Pesquisa do Estado de São Paulo (FAPESP grant no. 2014/25215-2).
Author information
Authors and Affiliations
Contributions
MAS and APH conceived and designed the research. HB, HP, and MLCV contributed to BAC library, plant material, and bioinformatic data. MAS and YD conducted the experiments. CM, MAS, YD, and ZPC analyzed the data. MAS and APH wrote the manuscript. All authors read and approved the manuscript.
Corresponding author
Additional information
Responsible Editor:Jiming Jiang
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Figure S1
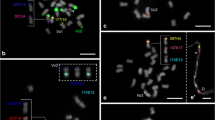
Fluorescent in situ hybridization to P. edulis chromosomes. (a) BAC Pe134H15 (red); (b) Pe164A12 (red) and 35S rDNA (green); (c) BAC Pe173B16 (green) and Pe164M13 (red); (d) BAC Pe195F4 (red) and 35S rDNA (green); (e) BAC Pe173B16 (red) and 35S rDNA (green). Chromosomes were counterstained with DAPI, are visualized in grey and indicated by number. BACs were labeled with Cy3-dUTP and pseudocolored. Bar = 5 μm. (PNG 8848 kb)
ESM 1
(DOCX 22 kb)
Rights and permissions
About this article
Cite this article
Sader, M.A., Dias, Y., Costa, Z.P. et al. Identification of passion fruit (Passiflora edulis) chromosomes using BAC-FISH. Chromosome Res 27, 299–311 (2019). https://doi.org/10.1007/s10577-019-09614-0
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10577-019-09614-0