Abstract
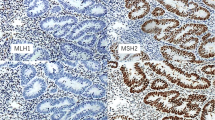
Lynch syndrome accounts for 3–5% of colorectal cancers and is due to a germline mutation in one of the mismatch repair genes MLH1, MSH2, MSH6, and PMS2. Somatic hypermethylation of the MLH1 promoter is commonly associated to sporadic cases. Strategies have been developed to identify patients with Lynch Syndrome based on clinical findings, tumoral phenotype, family history and immunohistochemistry analysis. However, there still are some pitfalls in this strategy, possibly responsible for an underdiagnosis of Lynch syndrome. Here we report the case of a 37 years-old man presenting with two concomitant tumors located in the rectosigmoid and in the ileocecal angle. Both tumors were microsatellites instability-high (MSI-H) and showed a loss of MLH1 and PMS2 protein expression, but only one had MLH1 promoter hypermethylation. Constitutional analysis of mismatch repair genes could not be performed from a blood sample, because of the early death of the patient. However, tumoral tissue analyses revealed in both tumors a pathogenic variant in the MLH1 gene. Further analysis of the surrounding tumor-free tissue also showed the presence of this alteration of the MHL1 gene. Finally, the same pathogenic variant was present constitutionally in one of the siblings of the patient, confirming its hereditary nature. This new case of concomitant presence of MLH1 promoter hypermethylation and MLH1 germline mutation demonstrates that the presence of MLH1 promoter hypermethylation should not rule out the diagnosis of Lynch Syndrome.
Similar content being viewed by others
References
Bonadona V, Bonaïti B, Olschwang S, Grandjouan S, Huiart L, Longy M, Guimbaud R, Buecher B, Bignon YJ, Caron O, Colas C, Noguès C, Lejeune-Dumoulin S, Olivier-Faivre L, Polycarpe-Osaer F, Nguyen TD, Desseigne F, Saurin JC, Berthet P, Leroux D, Duffour J, Manouvrier S, Frébourg T, Sobol H, Lasset C, Bonaïti-Pellié C (2011) Cancer risks associated with germline mutations in MLH1, MSH2, and MSH6 genes in Lynch syndrome. French Cancer Genet Netw 305:2304–2310. https://doi.org/10.1001/jama.2011.743
Ligtenberg MJL, Kuiper RP, Chan TL, Goossens M, Hebeda KM, Voorendt M, Lee TYH, Bodmer D, Hoenselaar E, Hendriks-Cornelissen SJB, Tsui WY, Kong CK, Brunner HG, van Kessel AG, Yuen ST, van Krieken JHJM, Leung SY, Hoogerbrugge N (2009) Heritable somatic methylation and inactivation of MSH2 in families with Lynch syndrome due to deletion of the 3’ exons of TACSTD1. Nat Genet 41:112–117. https://doi.org/10.1038/ng.283
Lynch HT, de la Chapelle A (2003) Hereditary colorectal cancer. N Engl J Med 348:919–932. https://doi.org/10.1056/NEJMra012242
Watson P, Vasen HFA, Mecklin JP, Bernstein I, Aarnio M, Järvinen HJ, Myrhøj T, Sunde L, Wijnen JT, Lynch HT (2008) The risk of extra-colonic, extra-endometrial cancer in the Lynch syndrome. Int J Cancer 123:444–449. https://doi.org/10.1002/ijc.23508
Cancer Genome Atlas N (2012) Comprehensive molecular characterization of human colon and rectal cancer. Nature 487:330–337. https://doi.org/10.1038/nature11252
Crépin M, Dieu MC, Lejeune S, Escande F, BoIDin D, Porchet N, Morin G, Manouvrier S, Mathieu M, Buisine MP (2012) EvIDence of constitutional MLH1 epimutation associated to transgenerational inheritance of cancer susceptibility. Hum Mutat 33:180–188. https://doi.org/10.1002/humu.21617
Peltomäki P (2014) Epigenetic mechanisms in the pathogenesis of Lynch syndrome. Clin Genet 85:403–412. https://doi.org/10.1111/cge.12349
Rahner N, Friedrichs N, Steinke V, Aretz S, Friedl W, Buettner R, Mangold E, Propping P, Walldorf C (2008) Coexisting somatic promoter hypermethylation and pathogenic MLH1 germline mutation in Lynch syndrome. J Pathol 214:10–16. https://doi.org/10.1002/path.2263
Hagen CE, Lefferts J, Hornick JL, Srivastava A (2011) “Null pattern” of immunoreactivity in a Lynch syndrome-associated colon cancer due to germline MSH2 mutation and somatic MLH1 hypermethylation. Am J Surg Pathol 35:1902–1905. https://doi.org/10.1097/PAS.0b013e318237c6ab
Raymond VM, Morris AM, Hafez KS, Greenson JK (2015) MLH1 promotor hypermethylation does not rule out a diagnosis of Lynch syndrome: a case report. Fam Cancer 14:77–80. https://doi.org/10.1007/s10689-014-9753-0
Cini G, Carnevali I, Quaia M, Chiaravalli AM, Sala P, Giacomini E, Maestro R, Tibiletti MG, Viel A (2015) Concomitant mutation and epimutation of the MLH1 gene in a Lynch syndrome family. Carcinogenesis 36:452–458. https://doi.org/10.1093/carcin/bgv015
Maliaka YK, Chudina AP, Belev NF, Alday P, Bochkov NP, Buerstedde JM (1996) CpG dinucleotides in the hMSH2 and hMLH1 genes are hotspots for HNPCC mutations. Hum Genet 97:251–255
Kurzawski G, Suchy J, Kładny J, Safranow K, Jakubowska A, Elsakov P, Kucinskas V, Gardovski J, Irmejs A, Sibul H, Huzarski T, Byrski T, Debniak T, Cybulski C, Gronwald J, Oszurek O, Clark J, Góźdź S, Niepsuj S, Słomski R, Pławski A, Łacka-Wojciechowska A, Rozmiarek A, Fiszer-Maliszewska Ł, Bebenek M, Sorokin D, Stawicka M, Godlewski D, Richter P, Brozek I, Wysocka B, Jawień A, Banaszkiewicz Z, Kowalczyk J, Czudowska D, Goretzki PE, Moeslein G, Lubiński J (2002) Germline MSH2 and MLH1 mutational spectrum in HNPCC families from Poland and the Baltic States. J Med Genet 39:E65
Kurzawski G, Suchy J, Lener M, Kłujszo-Grabowska E, Kładny J, Safranow K, Jakubowska K, Jakubowska A, Huzarski T, Byrski T, Debniak T, Cybulski C, Gronwald J, Oszurek O, Oszutowska D, Kowalska E, Góźdź S, Niepsuj S, Słomski R, Pławski A, Łacka-Wojciechowska A, Rozmiarek A, Fiszer-Maliszewska Ł, Bebenek M, Sorokin D, Sasiadek MM, Stembalska A, Grzebieniak Z, Kilar E, Stawicka M, Godlewski D, Richter P, Brozek I, Wysocka B, Limon J, Jawień A, Banaszkiewicz Z, Janiszewska H, Kowalczyk J, Czudowska D, Scott RJ, Lubiński J (2006) Germline MSH2 and MLH1 mutational spectrum including large rearrangements in HNPCC families from Poland (update study). Clin Genet 69:40–47. https://doi.org/10.1111/j.1399-0004.2006.00550.x
Bartosova Z, Fridrichova I, Bujalkova M, Wolf B, Ilencikova D, Krizan P, Hlavcak P, Palaj J, Lukac L, Lukacova M, Böör A, Haider R, Jiricny J, Nyström-Lahti M, Marra G (2003) Novel MLH1 and MSH2 germline mutations in the first HNPCC families identified in Slovakia. Hum Mutat 21:449. https://doi.org/10.1002/humu.9127
Hampel H, de la Chapelle A (2013) How do we approach the goal of Identifying everybody with Lynch syndrome? Fam Cancer 12:313–317. https://doi.org/10.1007/s10689-013-9611-5
Moreira L, Muñoz J, Cuatrecasas M, Quintanilla I, Leoz ML, Carballal S, Ocaña T, López-Cerón M, Pellise M, Castellví-Bel S, Jover R, Andreu M, Carracedo A, Xicola RM, Llor X, Boland CR, Goel A, Castells A, Balaguer F, Gastrointestinal Oncology Group of the Spanish Gastroenterological Association (2015) Prevalence of somatic mutl homolog 1 promoter hypermethylation in Lynch syndrome colorectal cancer. Cancer 121:1395–1404. https://doi.org/10.1002/cncr.29190
Herman JG, Umar A, Polyak K, Graff JR, Ahuja N, Issa JP, Markowitz S, Willson JK, Hamilton SR, Kinzler KW, Kane MF, Kolodner RD, Vogelstein B, Kunkel TA, Baylin SB (1998) IncIDence and functional consequences of hMLH1 promoter hypermethylation in colorectal carcinoma. Proc Natl Acad Sci USA 95:6870–6875
Poynter JN, Siegmund KD, Weisenberger DJ, Long TI, Thibodeau SN, Lindor N, Young J, Jenkins MA, Hopper JL, Baron JA, Buchanan D, Casey G, Levine AJ, Le Marchand L, Gallinger S, Bapat B, Potter JD, Newcomb PA, Haile RW, Laird PW, Colon Cancer Family Registry Investigators (2008) Molecular characterization of MSI-H colorectal cancer by MLHI promoter methylation, immunohistochemistry, and mismatch repair germline mutation screening. Cancer Epidemiol Biomark Prev 17:3208–3215. https://doi.org/10.1158/1055-9965.EPI-08-0512
Bouzourene H, Hutter P, Losi L, Martin P, Benhattar J (2010) Selection of patients with germline MLH1 mutated Lynch syndrome by determination of MLH1 methylation and BRAF mutation. Fam Cancer 9:167–172. https://doi.org/10.1007/s10689-009-9302-4
Kaz A, Kim YH, Dzieciatkowski S, Lynch H, Watson P, Kay Washington M, Lin L, Grady WM (2007) EvIDence for the role of aberrant DNA methylation in the pathogenesis of Lynch syndrome adenomas. Int J Cancer 120:1922–1929. https://doi.org/10.1002/ijc.22544
Racek AR, Rabe KG, Wick MJ, Psychogios A, Lindor NM (2011) Primary appendiceal mucinous adenocarcinoma in two first-degree relatives: case report and review. Hered Cancer Clin Pract 9:1. https://doi.org/10.1186/1897-4287-9-1
Misdraji J, Burgart LJ, Lauwers GY (2004) Defective mismatch repair in the pathogenesis of low-grade appendiceal mucinous neoplasms and adenocarcinomas. Mod Pathol 17:1447–1454. https://doi.org/10.1038/modpathol.3800212
Taggart MW, Galbincea J, Mansfield PF, Fournier KF, Royal RE, Overman MJ, RashID A, Abraham SC (2013) High-level microsatellite instability in appendiceal carcinomas. Am J Surg Pathol 37:1192–1200. https://doi.org/10.1097/PAS.0b013e318282649b
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors have declared no conflict of interest.
Rights and permissions
About this article
Cite this article
Kientz, C., Prieur, F., Clemenson, A. et al. MLH1 promoter hypermethylation: are you absolutely sure about the absence of MLH1 germline mutation? About a new case. Familial Cancer 19, 11–14 (2020). https://doi.org/10.1007/s10689-019-00151-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10689-019-00151-7