Abstract
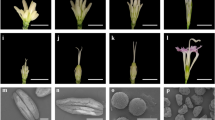
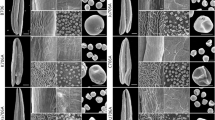
Environment-sensitive genic male sterility is a valid tool for hybrid production and hybrid breeding, but there are no previous reports of the molecular mechanism of fertility conversion. In this study, RNA-seq, phenotypic and cytological observations, and physiological indexes were applied to analyze thermo-sensitive genic male sterility line 4110S under different temperature conditions to explore the fertility transformation mechanism. In total, 3420 differentially expressed genes (DEGs) were identified comprising 2331 upregulated genes and 1089 downregulated genes. The DEGs were apparently distributed among 54 Gene Ontology functional groups. The phenylpropanoid, long-chain fatty acid, and jasmonic acid (JA) biosynthesis pathways were related to male sterility, where their downregulation blocked the synthesis of sporopollenin and JA. Phenotypic and cytological analyses showed that pollen wall defects and anther indehiscence at high temperatures induced sterility. Moreover, enzyme-linked immunosorbent assay results indicated that the abundance of JA was lower in 4110S under restrictive conditions (high temperature) than permissive conditions (low temperature). A possible regulated network of pathways associated with male sterility was suggested. These results provided insights into the molecular mechanism of fertility conversion in the thermosensitive male sterility system.









Similar content being viewed by others
Abbreviations
- DEG:
-
differentially expressed gene
- GO:
-
Gene Ontology
- JA:
-
jasmonic acid
- TGMS:
-
thermo-sensitive genic male sterility
- EGMS:
-
environmental-sensitive genic male sterility
- PAL:
-
phenylalanine ammonia-lyase
- 4CL:
-
4-coumarate:coenzyme A ligase
- LOX:
-
lipoxygenase
- AOS:
-
allene oxide synthase
- AOC:
-
allene oxide cyclase
- OPR:
-
12-oxo-phytodienoic acid reductase
- KOG:
-
Eukaryotic Orthologous Groups
- HCT:
-
O-hydroxycinnamoyltransferase
- AOS:
-
allene oxide synthase
- qRT-PCR:
-
quantitative real-time PCR
- DAPI:
-
4′,6-diamidino-2-phenylindole
- KCS:
-
3-ketoacyl-CoA synthase
- KAR:
-
17-beta-estradiol 17-dehydrogenase
- ELISA:
-
enzyme-linked immunosorbent assay
- FDR:
-
false discovery rate
References
Ai C, Kong L (2018) CGPS: A machine learning-based approach integrating multiple gene set analysis tools for better prioritization of biologically relevant pathways. J Genet Genomics 45(9):489–504. https://doi.org/10.1016/j.jgg.2018.08.002
Altschul SF, Madden TL, Schäffer AA, Zhang J, Zhang Z, Miller W, Lipman DJ (1997) Gapped BLAST and PSI BLAST: a new generation of protein database search programs. Nucleic Acids Res 25(17):3389–3402
An H, Yang Z, Yi B, Wen J, Shen J, Tu J, Ma C, Fu T (2014) Comparative transcript profiling of the fertile and sterile flower buds of pol CMS in B. napus. BMC Genome 15:258. https://doi.org/10.1186/1471-2164-15-258
Anders S, Huber W (2010) Differential expression analysis for sequence count data. Genome Biol 11(10):R106. https://doi.org/10.1186/gb-2010-11-10-r106
Blackmore S, Worrley AH, Skvarla JJ, Rowley JR (2007) Pollen wall development in flowering plants. New Phytol 174:483–498. https://doi.org/10.1111/j.1469-8137.2007.02060.x
Bokszczanin KL, Solanaceae Pollen Thermotolerance Initial Training Network (SPOT-ITN) Consortium, Fragkostefanakis S (2013) Perspectives on deciphering mechanisms underlying plant heat stress response and thermotolerance. Front Plant Sci 4:315. https://doi.org/10.3389/fpls.2013.00315
Chen H, Pan X (2001) Breeding and heterosis utilization of tropical and subtropical hybrid maize. Seed 2:57–58. https://doi.org/10.3969/j.issn.1001-4705.2001.02.028
Chen C, Chen G, Cao B, Lei J (2015) Transcriptional profiling analysis of genic male sterile–fertile Capsicum annuum reveal candidate genes for pollen development and maturation by RNA-Seq technology. Plant Cell Tissue Organ Cult 122:465–476. https://doi.org/10.1007/s11240-015-0784-5
Creelman RA, Mullet JE (1997) Biosynthesis and action of jasmonates in plants. Annu Rev Plant Physiol Mol Biol 48:355–381. https://doi.org/10.1146/annurev.arplant.48.1.355
Deng H, He Q (2017) Research and prospects of hybrid rice with strong heterosis in China. Sci Technol Rev 35:32–37. https://doi.org/10.3981/j.issn.1000-7857.2017.10.004
Dobritsa AA, Shrestha J, Morant M, Pinot F, Matsuno M, Swanson R, Møller BL, Preuss D (2009) CYP704B1 is a long-chain fatty acid omega-hydroxylase essential for sporopollenin synthesis in pollen of Arabidopsis. Plant Physiol 151:574–589. https://doi.org/10.1104/pp.109.144469
Du L, Zhang C, Zhu W, Zhang G (2005) The synthetic way and biological significance of plant secondary metabolism. J Northwest A&F Univ 20:150–155. https://doi.org/10.3969/j.issn.1001-7461.2005.03.041
Feys B, Benedetti CE, Penfold CN, Turner JG (1994) Arabidopsis mutants selected for resistance to the phytotoxin coronatine are male sterile, insensitive to methyl jasmonate, and resistant to a bacterial pathogen. Plant Cell 6:751–759. https://doi.org/10.2307/3869877
Fu Q, Cao Y, Li Y (2010) Advanced on studies and applications of photo-thermo-sensitive male sterility in wheat. J Trit Crops 30:576–580. https://doi.org/10.7606/j.issn.1009-1041.2010.03.038
Fu D, Xiao M, Hayward A, Jiang G, Zhu L, Zhou Q, Li J, Zhang M (2015) What is crop heterosis: new insights into an old topic. J Appl Genet 56:1–13. https://doi.org/10.1007/s13353-014-0231-z
Gan L, Xia K, Zhou X (2004) Involvement of jasmonates in regulation of male fertility in Arabidopsis thaliana. Plant Physiol J 40:269–274
Geng X, Ye J, Yang X, Li S, Zhang L, Song X (2018) Identification of proteins involved in carbohydrate metabolism and energy metabolism pathways and their regulation of cytoplasmic male sterility in wheat. Int J Mol Sci 19:324. https://doi.org/10.3390/ijms19020324
Howe GA, Schilmiller AL (2002) Oxylipin metabolism in response to stress. Curr Opin Plant Biol 5:230–236. https://doi.org/10.1016/S1369-5266(02)00250-9
International Wheat Genome Sequencing Consortium (IWGSC) (2014) A chromosome-based draft sequence of the hexaploid bread wheat (Triticum aestivum) genome. Science 345(6194):1251788. https://doi.org/10.1126/science.1251788
Ishiguro S, Kawai-Oda A, Ueda J, Nishida I, Okada K (2001) The DEFECTIVE IN ANTHER DEHISCIENCE gene encodes a novel phospholipase A1 catalyzing the initial step of jasmonic acid biosynthesis, which synchronizes pollen maturation, anther dehiscence, and flower opening in Arabidopsis. Plant Cell 13:2191–2209. https://doi.org/10.2307/3871502
Jeong HJ, Kang JH, Zhao M, Kwon JK, Choi HS, Bae JH, Lee H, Joung YH, Choi D, Kang BC (2014) Tomato male sterile 1035 is essential for pollen development and meiosis in anthers. J Exp Bot 65:6693–6709. https://doi.org/10.1093/jxb/eru389
Jiang H, Shi G (2009) Advanced search on male sterility in wheat. Biotechnol Bull 10:30–33. https://doi.org/10.13560/j.cnki.biotech.bull.1985.2009.10.027
Jiang K, Pi Y, Hou R, Tang K (2010) Jasmonate biosynthetic pathway: Its physiological role and potential application in plant secondary metabolic engineering. Bull Bot 45:137–148. https://doi.org/10.3969/j.issn.1674-3466.2010.02.001
Ju L, Zhu D, Zhang S, Zhang J, Yu Y, Liu H, Niu N, Wang J (2018) Correlation analysis between aspartic proteinase and tapetum metabolism in physiological male sterile line wheat. J Nuclear Agric Sci 32:0430–0437. https://doi.org/10.3864/j.issn.0578-1752.2017.21.002
Kim D, Langmead B, Salzberg SL (2015) HISAT: a fast spliced aligner with low memory requirements. Nat Methods 12(4):357–360. https://doi.org/10.1038/nmeth.3317
León J, Sánchez-Serrano JJ (1999) Molecular biology of jasmonic acid biosynthesis in plants. Plant Physiol Biochem 37:373–380. https://doi.org/10.1016/s0981-9428(99)80043-6
Li C, Williams MM, Loh YT, Lee GI, Howe GA (2002) Resistance of cultivated tomato to cell content-feeding herbivores is regulated by the octadecanoid-signaling pathway. Plant Physiol 130:494–503. https://doi.org/10.1104/pp.005314
Li J, Han S, Ding X, He T, Dai J, Yang S, Gai J (2015) Comparative transcriptome analysis between the cytoplasmic male sterile line NJCMS1A and its maintainer NJCMS1B in soybean (Glycine max (L.) Merr.). PLoS One 10:e0126771. https://doi.org/10.1371/journal.pone.0126771
Liu G, Xu S, Liu X (2006) Characteristics of “NEWFREE” CMS line and utilization in ornamental sunflower. Acta Agron Sin 32:1752–1755. https://doi.org/10.3321/j.issn:0496-3490.2006.11.026
Livak KJ, Schmittgen TD (2001) Analysis of relative gene expression data using real-time quantitative PCR and the 2(-Delta Delta C(T)) Method. Methods 25:402–408. https://doi.org/10.1006/meth.2001.1262
Mayfield JA, Preuss D (2000) Rapid initiation of Arabidopsis pollination requires the oleosin-domain protein GRP17. Nat Cell Biol 2:128–130. https://doi.org/10.1038/35000084
Mcconn M, Browse J (1996) The critical requirement for linolenic acid is pollen development, not photosynthesis, in an Arabidopsis mutant. Plant Cell 8:403–416. https://doi.org/10.1105/tpc.8.3.403
Meng L, Liu Z, Zhang L, Hu G, Song X (2016) Cytological characterization of a thermo-sensitive cytoplasmic male-sterile wheat line having K-type cytoplasm of Aegilops kotschyi. Breed Sci 66:752–761. https://doi.org/10.1270/jsbbs.16039
Meyer RC, Törjék O, Becher M, Altmann T (2004) Heterosis of biomass production in Arabidopsis establishment during early development. Plant Physiol 134:1813–1823. https://doi.org/10.1104/pp.103.033001
Pertea M, Pertea GM, Antonescu CM, Chang TC, Mendell JT, Salzberg SL (2015) Stringtie enables improved reconstruction of a transcriptome from RNA-seq reads. Nat Biotechnol 33:290–295. https://doi.org/10.1038/nbt.3122
Piffanelli P, Ross JHE, Murphy DJ (1998) Biogenesis and function of the lipidic structures of pollen grains. Sex Plant Reprod 11:65–80. https://doi.org/10.1007/s004970050122
Preuss D, Lemieux B, Yen G, Davis RW (1993) A conditional sterile mutation eliminates surface components from Arabidopsis pollen and disrupts cell signaling during fertilization. Genes Dev 7:974–985. https://doi.org/10.1101/gad.7.6.974
Qi T, Song S, Ren Q, Wu D, Huang H, Chen Y, Fan M, Peng W, Ren C, Xie D (2011) The jasmonate-ZIM-domain proteins interact with the WD-Repeat/bHLH/MYB complexes to regulate jasmonate-mediated anthocyanin accumulation and trichome initiation in Arabidopsis thaliana. Plant Cell 23:1795–1814. https://doi.org/10.1105/tpc.111.083261
Sanders PM, Lee PY, Biesgen C, Boone JD, Beals TP, Weiler EW, Goldberg RB (2000) The Arabidopsis delayed dehiscence1 gene encodes an enzyme in the jasmonic acid synthesis pathway. Plant Cell 12:1041–1061. https://doi.org/10.1105/tpc.12.7.1041
Sharma M, Laxmi A (2016) Jasmonates: emerging players in controlling temperature stress tolerance. Front Plant Sci 6:1129. https://doi.org/10.3389/fpls.2015.01129
Shi J, Liang WQ, Zhang DB (2007) Pollen wall development in plant. Plant Physiol J 43:588–592
Stintzi A, Browse J (2000) The Arabidopsis male-sterile mutant, opr3, lacks the 12-oxophytodienoic acid reductase required for jasmonate synthesis. Proc Natl Acad Sci U S A 9:10625–10630. https://doi.org/10.1073/pnas.190264497
Tang Z, Zhang L, Xu C, Yuan S, Zhang F, Zheng Y, Zhao C (2012) Uncovering small RNA-mediated responses to cold stress in a wheat thermosensitive genic male-sterile line by deep sequencing. Plant Physiol 159:721–738. https://doi.org/10.1104/pp.112.196048
Wang S, Lu J, Song XF, Ren SC, You C, Xu J, Liu CM, Ma H, Chang F (2017) Cytological and transcriptomic analyses reveal important roles of CLE19 in pollen exine formation. Plant Physiol 175:1186–1202. https://doi.org/10.1104/pp.17.00439
Wang K, Guo ZL, Zhou WT, Zhang C, Zhang ZY, Lou Y, Xiong SX, Yao XZ, Fan JJ, Zhu J, Yang ZN (2018) The regulation of sporopollenin genes for rapid pollen wall formation. Plant Physiol 178:283–294. https://doi.org/10.1104/pp.18.00219
Wu J, Mao X, Cai T, Luo J, Wei L (2006) KOBAS server: a web-based platform for automated annotation and pathway identification. Nucleic Acids Res 34:W720–W724
Xie DX, Feys BF, James S, Nieto-Rostro M, Turner JG (1998) COl1: an Arabidopsis gene required for jasmonate-regulated defense and fertility. Science 280:1091–1094. https://doi.org/10.1016/1369-5266(88)80029-3
Xie C, Mao X, Huang J, Ding Y, Wu J, Dong S, Kong L, Gao G, Li C, Wei L (2011) KOBAS 2.0: a web server for annotation and identification of enriched pathways and diseases. Nucleic Acids Res 39:W316–W322. https://doi.org/10.1093/nar/gkr483
Xu Y, Kang H (2001) Research progress on the two-lines hybrid wheat of photo-thermo-sensitivity. Shananxi J Agric Sci 01:28–29. https://doi.org/10.3969/j.issn.0488-5368.2001.09.012
Yang X, Wu D, Shi J, He Y, Pinot F, Grausem B, Yin C, Zhu L, Chen M, Luo Z, Liang W, Zhang D (2014) Rice CYP703A3, a cytochrome P450 hydroxylase, is essential for development of anther cuticle and pollen exine. J Integr Plant Biol 56:979–994. https://doi.org/10.1111/jipb.12212
Yang X, Geng X, Liu Z, Ye J, Zhang L, Song X (2018) A sterility induction trait in the genic male sterility wheat line 4110S induced by high temperature and its cytological response. Crop Sci 58:1–11. https://doi.org/10.2135/cropsci2017.12.0714
Ye J, Duan Y, Hu G, Geng X, Zhang G, Yan P, Liu Z, Zhang L, Song X (2017) Identification of candidate genes and biosynthesis pathways related to fertility conversion by wheat KTM3315A transcriptome profiling. Front Plant Sci 8:449. https://doi.org/10.3389/fpls.2017.00449
Zhang W (2000) Improving rape yield by heterosis. Gansu Agric 2:39–40. https://doi.org/10.15979/j.cnki.cn62-1104/f.2000.02.016
Zheng Y, Jiao C, Sun H, Rosli HG, Pombo MA, Zhang P, Banf M, Dai X, Martin GB, Giovannoni JJ, Zhao PX, Rhee SY, Fei Z (2016) iTAK: a program for genome-wide prediction and classification of plant transcription factors, transcriptional regulators, and protein kinases. Mol Plant 9:1667–1670. https://doi.org/10.1016/j.molp.2016.09.014
Acknowledgments
We thank Beijing BioMarker Company for their help with high-throughput sequencing. The data from this study was deposited in NCBI Sequence Read Archive under accession SRA: SRP211924. The line 4110S was presented by Dr. P.H. Dong from the College of Agronomy, Henan University of Science and Technology, China.
Funding
This study was supported by the National Natural Science Foundation of China (31771874) and the Program in Science and Technology of Yangling State Demonstration Zone of Agricultural High-tech Industries (2018NY-19).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
All experiments were carried out with the approval of Northwest A&F University.
Competing interest
The authors declare that they have no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary materials
Supplementary Table S1
Sequence-specific primers used for qRT-PCR (PDF 117 kb)
Supplementary Table S2
Gene Ontology (GO) classifications of differentially expressed genes (PDF 174 kb)
Supplementary Table S3
Eukaryotic Orthologous Groups (KOG) functional classifications of annotated differentially expressed genes (PDF 158 kb)
Supplementary Table S4
Transcript facror IDs and corresponding families (PDF 198 kb)
Supplementary Table S5
KEGG pathways and DEGs number (PDF 133 kb)
Supplementary Table S6
Genes and KEGG terms in networks (PDF 116 kb)
Supplementary Fig. S1
The GO enrichment of biological processes (PDF 578 kb)
Supplementary Fig. S2
The GO enrichment of cellular components (PDF 486 kb)
Supplementary Fig. S3
The GO enrichment of molecular functions (PDF 356 kb)
Supplementary File S1
The detailed experimental methods. (PDF 135 kb)
Rights and permissions
About this article
Cite this article
Yang, X., Ye, J., Zhang, L. et al. Blocked synthesis of sporopollenin and jasmonic acid leads to pollen wall defects and anther indehiscence in genic male sterile wheat line 4110S at high temperatures. Funct Integr Genomics 20, 383–396 (2020). https://doi.org/10.1007/s10142-019-00722-y
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10142-019-00722-y