Abstract
Introduction
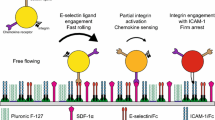
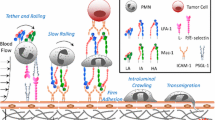
The arrest of rolling T lymphocytes at specific locations is crucial to proper immune response function. We previously developed a model of chemokine-driven integrin activation, termed integrative signaling adhesive dynamics (ISAD). In addition, we have shown that loss of diacylglycerol kinase (DGK) leads to a gain of function regarding adhesion under shear flow. We undertook this study to understand the sensitivity of adhesion to perturbations in other signaling molecules.
Methods
We adapted multi-parametric sensitivity analysis (MPSA) for use in our ISAD model to identify important parameters, including initial protein concentrations and kinetic rate constants, for T lymphocyte arrest. We also compared MPSA results to those obtained from a single parametric sensitivity analysis.
Results
In addition to the previously shown importance of DGK in lymphocyte arrest, PIP2 cleavage and Rap1 activation are crucial in determining T cell arrest dynamics, which agree with previous experimental findings. The l-selectin density on the T lymphocyte surface also plays a large role in determining the distance rolled before arrest. Both the MPSA and single-parametric method returned similar results regarding the most sensitive kinetic rate constants.
Conclusion
We show here that the regulation of the amount of second messengers are, in general, more critical for determining T lymphocyte arrest over the initial signaling proteins, highlighting the importance of amplification of signaling in cell adhesion responses. Overall, this work provides a mechanistic insight of the contribution of key pathways and components, thus may help to identify potential therapeutic targets for drug development against immune disorders.





Similar content being viewed by others
Abbreviations
- DAG:
-
Diacylglycerol
- DGK:
-
Diacylglycerol kinase
- ICAM-1:
-
Intracellular adhesion molecule-1
- ISAD:
-
Integrative-signaling adhesive dynamics
- LFA-1:
-
Lymphocyte function-associated protein-1
- MPSA:
-
Multi-parametric sensitivity analysis
- GPCR:
-
G protein coupled receptor
- IP3:
-
Inositol triphosphate
- PIP2 :
-
Phosphatidylinositol 4,5-bisphosphate
- PLC:
-
Phospholipase C
- GEF:
-
Guanine nucleotide exchange factor
References
Aldridge, B. B., J. M. Burke, D. A. Lauffenburger, and P. K. Sorger. Physicochemical modelling of cell signalling pathways. Nat. Cell Biol. 8:1195–1203, 2006.
Beste, M. T., D. Lee, M. R. King, G. A. Koretzky, and D. A. Hammer. An integrated stochastic model of “inside-out” integrin activation and selective T-lymphocyte recruitment. Langmuir 28:2225–2237, 2012.
Bialkowska, K., et al. The integrin co-activator kindlin-3 is expressed and functional in a non-hematopoietic cell, the endothelial cell. J. Biol. Chem. 285:18640–18649, 2010.
Caputo, K. E., and D. A. Hammer. Adhesive dynamics simulation of G-protein-mediated chemokine-activated neutrophil adhesion. Biophys. J. Biophys. Soc. 96:2989–3004, 2009.
Chen, W., and C. Zhu. Mechanical regulation of T-cell functions. Immunol. Rev. 256:160–176, 2013.
Cho, K.-H., S.-Y. Shin, W. Kolch, and O. Wolkenhauer. Experimental design in systems biology, based on parameter sensitivity analysis using a monte carlo method: a case study for the TNFα-mediated NF-κ B signal transduction pathway. Simulation 79:726–739, 2003.
DeNucci, C. C., J. S. Mitchell, and Y. Shimizu. Integrin function in T-cell homing to lymphoid and nonlymphoid sites: getting there and staying there. Crit. Rev. Immunol. 29:87–109, 2009.
Elf, J., A. Doncic, and M. Ehrenberg. Mesoscopic Reaction-Diffusion in Intracellular Signaling. Bellingham: International Society for Optics and Photonics, p. 114, 2003.
Galkina, E., et al. T lymphocyte rolling and recruitment into peripheral lymph nodes is regulated by a saturable density of L-selectin (CD62L). Eur. J. Immunol. 37:1243–1253, 2007.
Gallatin, W. M., I. L. Weissman, and E. C. Butcher. A cell-surface molecule involved in organ-specific homing of lymphocytes. Nature 304:30–34, 1983.
Ghandour, H., X. Cullere, A. Alvarez, F. W. Luscinskas, and T. N. Mayadas. Essential role for Rap1 GTPase and its guanine exchange factor CalDAG-GEFI in LFA-1 but not VLA-4 integrin mediated human T-cell adhesion essential role for Rap1 GTPase and its guanine exchange factor CalDAG-GEFI in LFA-1 but not VLA-4 integrin—mediated. Regulation 110:3682–3690, 2009.
Herter, J., and A. Zarbock. Integrin regulation during leukocyte recruitment. J. Immunol. 190:4451–4457, 2013.
Hogg, N., I. Patzak, and F. Willenbrock. The insider’s guide to leukocyte integrin signalling and function. Nat. Rev. Immunol. 11:416–426, 2011.
Katagiri, K., A. Maeda, M. Shimonaka, and T. Kinashi. RAPL, a Rap1-binding molecule that mediates Rap1-induced adhesion through spatial regulation of LFA-1. Nat. Immunol. 4:741–748, 2003.
Kim, M., C. V. Carman, W. Yang, A. Salas, and T. A. Springer. The primacy of affinity over clustering in regulation of adhesiveness of the integrin αLβ2. J. Cell Biol. 167:1241–1253, 2004.
Lee, D., J. Kim, M. T. Beste, G. A. Koretzky, and D. A. Hammer. Diacylglycerol kinase zeta negatively regulates CXCR16-stimulated T lymphocyte firm arrest to ICAM-1 under shear flow. Integr. Biol. 4:606, 2012.
Ley, K., and G. S. Kansas. Selectins in T-cell recruitment to non-lymphoid tissues and sites of inflammation. Nat. Rev. Immunol. 4:325–336, 2004.
Ley, K., C. Laudanna, M. I. Cybulsky, and S. Nourshargh. Getting to the site of inflammation: the leukocyte adhesion cascade updated. Nat. Rev. Immunol. 7:678–689, 2007.
Luo, B.-H., C. V. Carman, and T. A. Springer. Structural basis of integrin regulation and signaling. Annu. Rev. Immunol. 25:619–647, 2007.
McKay, M. D., R. J. Beckman, and W. J. Canover. A comparison of three methods for selecting values of input variables in the analysis of output from a computer code. Technometrics 21:239–245, 1979.
Montresor, A., L. Toffali, G. Constantin, and C. Laudanna. Chemokines and the signaling modules regulating integrin affinity. Front. Immunol. 3:1–10, 2012.
Shamri, R., et al. Lymphocyte arrest requires instantaneous induction of an extended LFA-1 conformation mediated by endothelium-bound chemokines. Nat. Immunol. 6:497–506, 2005.
Shimonaka, M., et al. Rap1 translates chemokine signals to integrin activation, cell polarization, and motility across vascular endothelium under flow. J. Cell Biol. 161:417–427, 2003.
Tiemann, C. A., J. Vanlier, M. H. Oosterveer, A. K. Groen, P. A. J. Hilbers, and N. A. W. van Riel. Parameter trajectory analysis to identify treatment effects of pharmacological interventions. PLoS Comput. Biol. 9(8):e1003166, 2013.
von Andrian, U. H., and C. R. Mackay. T-cell function and migration. Two sides of the same coin. N. Engl. J. Med. 343:1020–1034, 2000.
Weiss, A., and J. B. Imboden. Cell surface molecules and early events involved in human T lymphocyte activation. Cambridge: Academic Press, pp. 1–38, 1987.
Yoon, J., and T. S. Deisboeck. Investigating differential dynamics of the MAPK signaling cascade using a multi-parametric global sensitivity analysis. PLoS ONE 4(2):e4560, 2009.
Zi, Z. Sensitivity analysis approaches applied to systems biology models. IET Syst. Biol. 5:336–346, 2011.
Zi, Z., K. H. Cho, M. H. Sung, X. Xia, J. Zheng, and Z. Sun. In silico identification of the key components and steps in IFN-γ induced JAK-STAT signaling pathway. FEBS Lett. 579:1101–1108, 2005.
Acknowledgments
This work was supported by NIH 1R01AI082292 (D.A.H. and G.A.K.) and 5R01GM123019 (D.A.H.).
Conflict of interest
Dooyoung Lee, Michael T. Beste, Nicholas R. Anderson, Gary A. Koretzky and Daniel A. Hammer declare that they have no conflicts of interest.
Data Availability
The codes used to perform the calculations in this paper are available upon request to the corresponding author, Daniel A. Hammer, hammer@seas.upenn.edu, in accordance with the data sharing policy of the National Institutes of Health which funded this work.
Ethical Standards
No human studies were carried out by the authors for this article. No animal studies were carried out by the authors for this article.
Author information
Authors and Affiliations
Contributions
DL designed the study, performed simulations, analyzed the data and wrote the paper. MTB designed the study, performed simulations and analyzed the data. NRA wrote the paper. GAK designed the study. DAH designed the study and wrote the paper.
Corresponding author
Additional information
Associate Editor Michael R. King oversaw the review of this article.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic Supplementary Material
Below is the link to the electronic supplementary material.
12195_2019_575_MOESM1_ESM.eps
Supplemental Fig. 1. Cumulative frequency of MPSA with respect to kinetic parameters in chemokine-triggered integrin LFA-1 activation signaling during T lymphocyte trafficking under flow using distance to stop as metric of interest. Red curves represent unacceptable cases and blue curves denote acceptable cases. The K–S statistic is calculated with the maximal vertical difference between the two curves.. Supplementary material 1 (EPS 1570 kb)
Rights and permissions
About this article
Cite this article
Lee, D., Beste, M.T., Anderson, N.R. et al. Identifying Key Pathways and Components in Chemokine-Triggered T Lymphocyte Arrest Dynamics Using a Multi-Parametric Global Sensitivity Analysis. Cel. Mol. Bioeng. 12, 193–202 (2019). https://doi.org/10.1007/s12195-019-00575-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12195-019-00575-2