Abstract
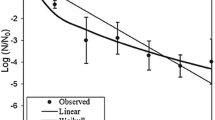
Male-specific coliphages (MSCs) are currently used to assess the virologic quality of shellfish-growing waters and to assess the impact of sewage release or adverse weather events on bivalve shellfish. Since MSC can have either DNA or RNA genomes, and most research has been performed exclusively on RNA MSCs, persistence of M13, a DNA MSC, was evaluated for its persistence as a function of time and temperature within Eastern oysters (Crassostrea virginica). Oysters were individually exposed to seawater containing a total of 1010 to 1012 pfu of M13 for 24 h at 15 °C followed by maintenance in tanks with as many as 21 oysters in continuously UV-sterilized water for up to 6 weeks at either 7, 15, or 22 °C. Two trials for each temperature were performed combining three shucked oysters per time point which were assayed by tenfold serial dilution in triplicate. Initial contamination levels averaged 106.9 and ranged from 106.0 to 107.0 of M13. For oysters held for 3 weeks, log10 reductions were 1.7, 3.8, and 4.2 log10 at 7, 15, and 22 °C, respectively. Oysters held at 7 and 15 °C for 6 weeks showed average reductions of 3.6 and 5.1 log10, respectively, but still retained infectious M13. In total, this work shows that DNA MSC may decline within shellfish in a manner analogous to RNA MSCs.

Similar content being viewed by others
References
Allwood, P. B., Malik, Y. S., Hedberg, C. W., &. Goyal, S. M. (2003). Survival of F-specific RNA coliphage, feline calicivirus, and Esherichia coli in water: A comparative study. Applied and Environmental Microbiology, 69, 5707–5710.
Anon. (2009). Modified double agar overlay method for determining male-specific coliphage in soft shelled clams and American oysters. ISSC Summary of Actions 2009. Proposal 05-114.
Burkhardt, W., & Calci, K. R. (2000). Selective accumulation may account for shellfish-associated viral illness. Applied and Environmental Microbiology, 66, 1375–1378.
Burkhardt, W., Watkins, W. D., & Rippey, S. R. (1992a). Survival and replication of male-specific bacteriophages in molluscan shellfish. Applied and Environmental Microbiology, 58, 1371–1373.
Burkhardt, W., Watkins, W. D., & Rippey, S. R. (1992b). Seasonal effects on accumulation of microbial indicator organisms by Mercenaria mercenaria. Applied and Environmental Microbiology, 58, 826–831.
Choi, C., & Kingsley, D. H. (2016). Temperature-dependent persistence of human norovirus within oysters (Crassostrea virginica). Food and Environmental Virology, 8, 141–147.
Cole, D., Long, S. C., & Sobsey, M. D. (2003). Evaluation of F + RNA and DNA coliphages as source-specific indicators of fecal contamination of surface waters. Applied and Environmental Microbiology, 69, 6507–6514.
Cornax, R., Moriñigo, M. A., Gonzalez-Jaen, F., Alonso, M. C., & Borrego, J. J. (1994). Bacteriophages presence in human faeces of healthy subjects and patients with gastrointestinal disturbances. Zentralblatt für Bakteriologie, 281, 214–224.
Croci, L., De Medici, D., Scalfaro, C., Fiore, A., Divizia, M., Donia, D., Cosentino, A. M., Moretti, P., & Constantini, G. (2000). Determination of enteroviruses, hepatitis A virus, bacteriophages, and Escherichia coli in Adriatic Sea mussels. Journal of Applied Microbiology, 88, 293–298.
Debartolomeis, J., & Cabelli, V. J. (1991). Evaluation of an Escherichia coli host strain for enumeration of F male-specific bacteriophages. Applied and Environmental Microbiology, 5, 1301–1305.
Doré, W. J., Henshilwood, K., & Lees, D. N. (2000). Evaluation of F-specific RNA bacteriophage as a candidate human enteric virus indicator for bivalve molluscan shellfish. Applied and Environmental Microbiology, 66, 1280–1285.
Doré, W. J., & Lees, D. L. (1995). Behavior of Escherichia coli and male-specific bacteriophage in environmentally contaminated bivalve molluscs before and after depuration. Applied and Environmental Microbiology, 61, 2830–2834.
Ettayebi, K., Crawford, S. E., Murakami, K., Broughman, J. R., Karandikar, U., Tenge, V. R., Neill, F. H., Blutt, S. E., Zeng, X. L., Qu, L., Kou, B., Opekun, A. R., Burrin, D., Graham, D. Y., Ramani, S., Atmar, R. L., & Estes, M. K. (2016). Replication of human noroviruses in stem cell-derived human enteroids. Science, 353, 1387–1393.
Flannery, J., Keaveney, S., & Doré, W. (2009). Use of FRNA bacteriophages to indicate the risk of norovirus contamination in Irish oysters. Journal of Food Protection, 72, 2358–2362.
Hartard, C., Banas, S., Loutreul, J., Rincé, A., Benoit, F., Boudaud, N., & Gantzer, C. (2016). Relevance of F-specific RNA bacteriophages in assessing human norovirus risk in shellfish and environmental waters. Applied and Environmental Microbiology, 82, 5709–5719.
Hartard, C., Banas, S., Rivet, R., Boudaud, N., & Gantzer, C. (2017). Rapid and sensitive method to assess human viral pollution in shellfish using infectious F-specific RNA bacteriophages: Application to marketed products. Food Microbiology, 63, 248–254.
Hartard, C., Leclerc, M., Rivet, R., Maul, A., Loutreul, J., Banas, S., Boudaud, N., & Gantzer, C. (2018). F-specific RNA bacteriophages, especially members of subgroup II, should be reconsidered as good indicators of viral pollution of oysters. Applied and Environmental Microbiology, 84, e01866–e01817.
Hartard, C., Rivet, R., Banas, S., & Gantzer, C. (2015). Occurrence of and sequence variation among F-specific RNA bacteriophage subgroups in feces and wastewater of urban and animal origins. Applied and Environmental Microbiology, 81, 6505–6515.
Hata, A., Hanamoto, S., Shirasaka, Y., Yamashita, N., & Tanaka, H. (2016). Quantitative distribution of infectious F-specific RNA phage genotypes in surface waters. Applied and Environmental Microbiology, 82, 4244–4252.
Havelaar, A. H., & Pot-Hogeboom, W. M. (1988). F-specific RNA-bacteriophages as model viruses in water hygiene: Ecological aspects. Water Science and Technology, 20, 399–407.
Havelaar, A. H., Pot-Hogeboom, W. M., Furuse, K., Pot, R., & Hormann, M. P. (1990). F-specific RNA bacteriophages and sensitive host strains in faeces and wastewater of human and animal origin. Journal of Applied Microbiology, 69, 30–37.
Jones, M. K., Grau, K. R., Costantini, V., Kolawole, A. O., de Graaf, M., Freiden, P., Graves, C. L., Koopmans, M., Wallet, S. M., Tibbetts, S. A., Schultz-Cherry, S., Wobus, C. E., Vinjé, J., & Karst, S. M. (2015). Human norovirus culture in B cells. Nature Protocols, 10, 1939–1947.
Kingsley, D. H., Chen, H., & Meade, G. K. (2018). Persistence of MS-2 Bacteriophage within Eastern Oysters. Food and Environmental Virology, 10, 83–88.
Kingsley, D. H., Fay, J., Calci, K., Pouillot, R., Woods, J., Chen, H., Niemira, B. A., & Van Doren, J. (2017). Evaluation of chlorine treatment levels on inactivation of human norovirus and MS2 during sewage treatment. Applied and Environmental Microbiology, 83, e01270–e01217.
Kingsley, D. H., & Richards, G. P. (2003). Persistence of hepatitis A virus within oysters. Journal of Food Protection, 66, 331–334.
Kitajima, M., Iker, B. C., Pepper, I. L., & Gerba, C. P. (2014). Relative abundance and treatment reduction of viruses during wastewater treatment processes-Identification of potential viral indicators. Science of the Total Environment. 488–489, 290 – 296.
LeClerc, H., Edberg, S., Pierzo, V., & Delattre, J. M. (2000). Bacteriophages as indicators of enteric viruses and public health risk in groundwaters. Journal of Applied Microbiology, 88, 5–21.
Long, S. C., El-Khoury, S. S., Oudejans, S. J. G., Sobsey, M. D., & Vinjé, J. (2005). Assessment of sources and diversity of male-specific coliphages for source tracking. Environmental Engineering Science, 22, 367–377.
Love, D. C., Lovelace, G. L., & Sobsey, M. D. (2010). Removal of Escherichia coli, Enterococcus fecalis, coliphage MS2, poliovirus, and hepatitis A virus from oysters (Crassostrea virginica) and hard shell clams (Mercinaria mercinaria) by depuration. International Journal of Food Microbiology, 143, 211–217.
Luther, K., & Fujioka, R. (2004). Usefulness of monitoring tropical streams for male-specific RNA coliphages. Journal of Water Health, 2, 171–181.
Nappier, S. P., Graczyk, T. K., & Schwab, K. J. (2008). Bioaccumulation, retention, and depuration of enteric viruses by Crassostrea virginica and Crassostrea ariakensis oysters. Applied and Environmental Microbiology, 74, 6825–6831.
Neter, J., & Kutner, M. H., Nachtsheim, C. J., & Wasserman, W. (1996). Applied linear regression models (pp. 531–563). Chicago: McGraw-Hill Co., Inc.
NSSP Guide for the Control of Molluscan Shellfish: 2017 revision; Retrieved November 1 2018 https://www.fda.gov/food/guidanceregulation/federalstatefoodprograms/ucm2006754.htm.
Peleg, M., & Cole, M. B. (1998). Reinterpretation of microbial survival curves. Critical Reviews in Food Science and Nutrition, 38, 353–380.
Pouillot, R., Van Doren, J. M., Woods, J., Plante, D., Smith, M., Goblick, G., Roberts, C., Locas, A., Hajen, W., Stobo, J., White, J., Holtzman, J., Buenaventura, E., Burkhardt, W. I. I. I., Catford, A., Edwards, R., DePaola, A., & Calci, K. (2015). Meta-analysis of the reduction of norovirus and male-specific coliphage concentrations in wastewater treatment plants. Applied and Environmental. Microbiology, 81, 4669–4681.
Primrose, S. B., Seeley, N. D., Logan, K. B., & Nicolson, J. W. (1982). Methods for studying aquatic bacteriophage ecology. Applied and Environmental Microbiology, 43, 694–701.
Provost, K., Dancho, B. A., Ozbay, G., Anderson, R. S., Richards, G. P., & Kingsley, D. H. (2011). Hemocytes are sites of enteric virus persistence within oysters. Applied and Environmental Microbiology, 77, 8360–8369.
Purnell, S., Ebdon, J., Buck, A., Tupper, M., & Taylor, H. (2016). Removal of phages and viral pathogens in a full-scale MBR: Implications for wastewater reuse and potable water. Water Research, 100, 20–27.
Richards, G. P. (1985). Outbreaks of shellfish-associated enteric virus illness in the United States: Requisite for development of viral guidelines. Journal of Food Protection, 48, 815–823.
Richards, G. P. (1988). Microbial purification of shellfish: A review of depuration and relaying. Journal of Food Protection, 51, 218–251.
Rippey, S. R. (1994). Infectious diseases associated with molluscan shellfish consumption. Clinical Microbiology Reviews, 7, 419–425.
Schaffner, D. W., & Labuza, T. P. (1997). Predictive microbiology: Where are we, and where are we going? Food Technology, 51, 95–99.
Schwab, K. J., Neill, F. H., Estes, M. K., Metcalf, T. G., & Atmar, R. L. (1998). Distribution of norwalk virus within shellfish following bioaccumulation and subsequent depuration by detection using RT-PCR. Journal of Food Protection, 61, 1674–1680.
Shaw, R., & Cabelli, V. J. (1981). R plasmid transfer frequencies from environmental Escherichia coli to laboratory and fecal strains. Applied and Environmental Microbiology, 40, 756–764.
Simmons, F. J., & Xagoraraki, I. (2011). Release of infectious human enteric viruses by full scale wastewater utilities. Water Research, 45, 3590–3598.
Wang, D., Wu, Q., Yao, L., Wei, M., Kou, X., & Zhang, J. (2008). New target tissue for food-borne virus detection in oysters. Letters in Applied Microbiology, 47, 405–409.
Acknowledgements
We wish to thank Gary Richards (USDA, Dover, DE) and Brendan Niemira (USDA, Wyndmoor, PA) for critical reading of the manuscript.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
U.S. Department of Agriculture is an equal opportunity provider and employer. Mention of trade names or commercial products is solely for the purpose of providing specific information and does not imply recommendation or endorsement by the U.S. Department of Agriculture.
Rights and permissions
About this article
Cite this article
Kingsley, D.H., Chen, H., Annous, B.A. et al. Evaluation of a Male-Specific DNA Coliphage Persistence Within Eastern Oysters (Crassostrea virginica). Food Environ Virol 11, 120–125 (2019). https://doi.org/10.1007/s12560-019-09376-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12560-019-09376-2