Abstract
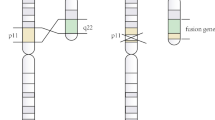
A 67-year-old female was referred to our hospital with a sternal fracture in March 2008. She received a diagnosis of multiple myeloma (MM) BJP-κ type (ISS stage III). G-banding karyotype revealed 46, XX, t(11;22)(q23.3;q11.2) (Hubacek, Gene 592:193–9, 2016), which was later confirmed to be congenital. After repeated rounds of chemotherapy with bortezomib and lenalidomide, she obtained a very good partial response in August 2014, and she was followed up with no treatment. However, she relapsed in February 2016. At that time, fluorescence in situ hybridization identified del(13q) and t(4;14)(p16;q32), which are associated with a poor prognosis. Furthermore, PCR analysis showed that the chromosome 11 breakpoint was at the APOA5/APOA4 locus at 11q23.3, which is associated with malignancy, and that the chromosome 22 breakpoint was at the SEPT5 intron 1 locus, which also plays a role in leukemogenesis through formation of a fusion gene with MLL. Although she was treated with three further lines of therapy, she died from disease progression in August 2017. Synergism between t(11;22) and t(4;14) may have induced the double-refractory phenotype to proteasome inhibitor and lenalidomide, at least during the chemorefractory phase. We present a biological analysis of this case and a review of the literature.




Similar content being viewed by others
References
Palumbo A, Anderson K. Multiple myeloma. N Engl J Med. 2011;364:1046–60.
Raab MS, Podar K, Breitkreutz I, Richardson PG, Anderson KC. Multiple myeloma. Lancet. 2009;374:324–39.
Taylor TD, Noguchi H, Totoki Y, Toyoda A, Kuroki Y, et al. Human chromosome 11 DNA sequence and analysis including novel gene identification. Nature. 2006;440:497–500.
Yokoyama A. Transcriptional activation by MLL fusion proteins in leukemogenesis. Exp Hematol. 2017;46:21–30.
Slany RK. The molecular mechanics of mixed lineage leukemia. Oncogene. 2016;35:5215–23.
Zuo W, Wang SA, DiNardo C, Yabe M, Li S, et al. Acute leukaemia and myelodysplastic syndromes with chromosomal rearrangement involving 11q23 locus, but not MLL gene. J Clin Pathol. 2017;70:244–9.
Leiba M, Duek A, Amariglio N, Avigdor A, Benyamini N, et al. Translocation t(11;14) in newly diagnosed patients with multiple myeloma: Is it always favorable? Genes Chromosomes Cancer. 2016;55:710–8.
Cremer FW, Kartal M, Hose D, Bila J, Buck I, et al. High incidence and intraclonal heterogeneity of chromosome 11 aberrations in patients with newly diagnosed multiple myeloma detected by multiprobe interphase FISH. Cancer Genet Cytogenet. 2005;161:116–24.
Delihas N. A family of long intergenic non-coding RNA genes in human chromosomal region 22q11.2 carry a DNA translocation breakpoint/AT-rich sequence. PLoS One. 2018;13:e0195702.
Kurahashi H, Inagaki H, Hosoba E, Kato T, Ohye T, et al. Molecular cloning of a translocation breakpoint hotspot in 22q11. Genome Res. 2007;17:461–9.
McDonald-McGinn DM, Sullivan KE, Marino B, Philip N, Swillen A, et al. 22q11.2 deletion syndrome. Nat Rev Dis Primers. 2015;1:15071.
Hiroi N, Hiramoto T, Harper KM, Suzuki G, Boku S. Mouse models of 22q11.2-associated autism spectrum disorder. Autism Open Access Suppl. 2012;1:001.
Launay E, Henry C, Meyer C, Chappe C, Taque S, et al. MLL-SEPT5 fusion transcript in infant acute myeloid leukemia with t(11;22)(q23;q11). Leuk Lymphoma. 2014;55:662–7.
Kurahashi H, Shaikh TH, Zackai EH, Celle L, Driscoll DA, et al. Tightly clustered 11q23 and 22q11 breakpoints permit PCR-based detection of the recurrent constitutional t(11;22). Am J Hum Genet. 2000;67:763–8.
Meyer C, Hofmann J, Burmeister T, Groger D, Park TS, et al. The MLL recombinome of acute leukemias in 2013. Leukemia. 2013;27:2165–76.
Cerveira N, Bizarro S, Teixeira MR. MLL-SEPTIN gene fusions in hematological malignancies. Biol Chem. 2011;392:713–24.
Hall PA, Russell SE. The pathobiology of the septin gene family. J Pathol. 2004;204:489–505.
Ono R, Ihara M, Nakajima H, Ozaki K, Kataoka-Fujiwara Y, et al. Disruption of Sept6, a fusion partner gene of MLL, does not affect ontogeny, leukemogenesis induced by MLL-SEPT6, or phenotype induced by the loss of Sept4. Mol Cell Biol. 2005;25:10965–78.
Guardiola M, Ribalta J. Update on APOA5 Genetics: toward a better understanding of its physiological impact. Curr Atheroscler Rep. 2017;19:30.
Hubacek JA. Apolipoprotein A5 fifteen years anniversary: lessons from genetic epidemiology. Gene. 2016;592:193–9.
Au A, Griffiths LR, Irene L, Kooi CW, Wei LK. The impact of APOA5, APOB, APOC3 and ABCA1 gene polymorphisms on ischemic stroke: evidence from a meta-analysis. Atherosclerosis. 2017;265:60–70.
Major JM, Yu K, Weinstein SJ, Berndt SI, Hyland PL, et al. Genetic variants reflecting higher vitamin e status in men are associated with reduced risk of prostate cancer. J Nutr. 2014;144:729–33.
Chang SH, Luo S, Thomas TS, O’Brian KK, Colditz GA, et al. (2017) Obesity and the transformation of monoclonal gammopathy of undetermined significance to multiple myeloma: a population-based cohort study. J Natl Cancer Inst 109.
Lauby-Secretan B, Scoccianti C, Loomis D, Grosse Y, Bianchini F, et al. Body fatness and cancer—viewpoint of the IARC working group. N Engl J Med. 2016;375:794–8.
Morris EV, Edwards CM. (2018) Adipokines, adiposity, and bone marrow adipocytes: Dangerous accomplices in multiple myeloma. J Cell Physiol
Shah MY, Martinez-Garcia E, Phillip JM, Chambliss AB, Popovic R, et al. MMSET/WHSC1 enhances DNA damage repair leading to an increase in resistance to chemotherapeutic agents. Oncogene. 2016;35:5905–15.
Kaseb HO, Lewis DW, Saunders WS, Gollin SM. Cell division patterns and chromosomal segregation defects in oral cancer stem cells. Genes Chromosomes Cancer. 2016;55:694–709.
Li F, Zhai YP, Lai T, Zhao Q, Zhang H, et al. MB4-2/MB4-3 transcripts of IGH-MMSET fusion gene in t(4;14)(pos) multiple myeloma indicate poor prognosis. Oncotarget. 2017;8:51608–20.
Marango J, Shimoyama M, Nishio H, Meyer JA, Min DJ, et al. The MMSET protein is a histone methyltransferase with characteristics of a transcriptional corepressor. Blood. 2008;111:3145–54.
Pei H, Zhang L, Luo K, Qin Y, Chesi M, et al. MMSET regulates histone H4K20 methylation and 53BP1 accumulation at DNA damage sites. Nature. 2011;470:124–8.
Author information
Authors and Affiliations
Contributions
R.S. and K.A. analyzed the clinical status of the patient, analyzed the data, and prepared the manuscript. T.W., Y.N., Y.K., Y.A., Y.O., and H.K. analyzed the data.
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare no competing financial interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
About this article
Cite this article
Suzuki, R., Warita, T., Nakamura, Y. et al. A case of double-refractory multiple myeloma with both the IgH-MMSET fusion protein and the congenital abnormality t(11;22). Int J Hematol 109, 731–736 (2019). https://doi.org/10.1007/s12185-019-02603-3
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12185-019-02603-3