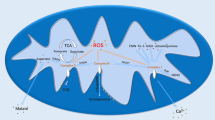
Abstract
Many natural products could induce apoptosis through mitochondrial pathways. However, direct interactions between natural products and mitochondria have rarely been reported. In this work, the effects and regulatory mechanisms of Jaceosidin on the isolated rat liver mitochondria have been studied. The results of the experiments which by introducing exogenous Ca2+ illustrated that Jaceosidin has the protective effects on the structure and function of the isolated mitochondria. These protective effects were related to the chelation of Ca2+ with Jaceosidin. Besides, Jaceosidin could scavenge reactive oxygen species produced during electron transport, and weaken the mitochondrial lipid peroxidation rate, which may be attributed to the antioxidant effect of phenolic hydroxyl groups of Jaceosidin. In addition, Jaceosidin has some damage effects on mitochondrial function, such as the inhibition of mitochondrial respiration and the increase of mitochondrial membrane fluidity. These results of this work provided comprehensive information to clarify the mechanisms of Jaceosidin on mitochondria, which may be the bidirectional regulatory mechanisms.









Similar content being viewed by others
References
Andreyev AY, Kushnareva YE, Starkov AA (2005) Mitochondrial metabolism of reactive oxygen species. Biochemistry (Moscow) 70(2):200–214. https://doi.org/10.1007/s10541-005-0102-7
Aranda A, Sequedo L, Tolosa L, Quintas G, Burello E, Castell JV, Gombau L (2013) Dichloro-dihydro-fluorescein diacetate (DCFH-DA) assay: a quantitative method for oxidative stress assessment of nanoparticle-treated cells. Toxicol In Vitro 27(2):954–963. https://doi.org/10.1016/j.tiv.2013.01.016
Aruoma OI, Halliwell B, Laughton MJ, Quinlan GJ, Gutteridge JMC (1989) The mechanism of initiation of lipid peroxidation. Evidence against a requirement for an iron(II)-iron(III) complex. Biochem J 258(2):617–620. https://doi.org/10.1042/bj2580617
Azzi A, Montecucco C (1976) Probes for energy transduction in membranes. J Bioenergy Biomembr 8(5):257–269. https://doi.org/10.1007/bf00761451
Brookes PS, Yoon Y, Robotham JL, Anders MW, Sheu SS (2004) Calcium, ATP, and ROS: a mitochondrial love-hate triangle. Am J Physiol Cell Physiol 287(4):C817–C833. https://doi.org/10.1152/ajpcell.00139.2004
Dong J-X, Zhao G-Y, Yu Q-L-Y, Li R, Yuan L, Chen J, Liu Y (2013) Mitochondrial dysfunction induced by honokiol. J Membr Biol 246(5):375–381. https://doi.org/10.1007/s00232-013-9543-x
Dong P, Li JH, Xu SP, Wu XJ, Xiang X, Yang QQ, Jin JC, Liu Y, Jiang FL (2016) Mitochondrial dysfunction induced by ultra-small silver nanoclusters with a distinct toxic mechanism. J Hazard Mater 308:139–148. https://doi.org/10.1016/j.jhazmat.2016.01.017
Dorta DJ, Pigoso AA, Mingatto FE, Rodrigues T, Pestana CR, Uyemura SA, Santos AC, Curti C (2008) Antioxidant activity of flavonoids in isolated mitochondria. Phytother Res 22(9):1213–1218. https://doi.org/10.1002/ptr.2441
Feissner RF, Skalska J, Gaum WE, Sheu SS (2009) Crosstalk signaling between mitochondrial Ca2+ and ROS. Front Biosci-Landmark 14:1197–1218. https://doi.org/10.2741/3303
Gao JL, Wu M, Liu W, Feng ZJ, Zhang YZ, Jiang FL, Liu Y, Dai J (2015) Dysfunction of rice mitochondrial membrane induced by Yb3+. J Membr Biol 248(6):1159–1165. https://doi.org/10.1007/s00232-015-9833-6
Ge YB, Wang ZG, Xiong Y, Huang XJ, Mei ZN, Hong ZG (2016) Anti-inflammatory and blood stasis activities of essential oil extracted from Artemisia argyi leaf in animals. J Nat Med 70(3):531–538. https://doi.org/10.1007/s11418-016-0972-6
Giorgio V, Guo L, Bassot C, Petronilli V, Bernardi P (2018) Calcium and regulation of the mitochondrial permeability transition. Cell Calcium 70:56–63. https://doi.org/10.1016/j.ceca.2017.05.004
Halestrap AP, Connern CP, Griffiths EJ, Kerr PM (1997) Cyclosporin A binding in mitochondrial cyclophilin inhibits the permeability transition pore and protects hearts from ischaemia/reperfusion injury. Mol Cell Biochem 174(1–2):167–172. https://doi.org/10.1023/a:1006879618176
Harvey AE, Manning DL (1950) Spectrophotometric methods of establishing empirical formulas of colored complexes in solution. J Am Chem Soc 72(10):4488–4493. https://doi.org/10.1021/ja01166a044
Horvath SE, Daum G (2013) Lipids of mitochondria. Prog Lipid Res 52(4):590–614. https://doi.org/10.1016/j.plipres.2013.07.002
Hurst S, Hoek J, Sheu SS (2017) Mitochondrial Ca2+ and regulation of the permeability transition pore. J Bioenergy Biomembr 49(1):27–47. https://doi.org/10.1007/s10863-016-9672-x
Jiao YH, Zhang Q, Pan LL, Chen XY, Lei KL, Zhao J, Jiang FL, Liu Y (2015) Rat liver mitochondrial dysfunction induced by an organic arsenical compound 4-(2-nitrobenzaliminyl) phenyl arsenoxide. J Membr Biol 248(6):1071–1078. https://doi.org/10.1007/s00232-015-9818-5
Kim MJ, Kim DH, Lee KW, Yoon DY, Surh YJ (2007) Jaceosidin induces apoptosis in ras-transformed human breast epithelial cells through generation of reactive oxygen species. Ann N Y Acad Sci 1095:483–495. https://doi.org/10.1196/annals.1397.052
Lee SH, Bae EA, Park EK, Shin YW, Baek NI, Han EJ, Chung HG, Kim DH (2007) Inhibitory effect of eupatilin and jaceosidin isolated from Artemisia princeps in IgE-induced hypersensitivity. Int Immunopharmacol 7(13):1678–1684. https://doi.org/10.1016/j.intimp.2007.08.028
Li Y, Tan Y (2013) Jaceosidin inhibits proliferation of human bladder cancer T24 cells through induction of cell cycle arrest and apoptosis. Bangl J Pharmacol 8(3):349–356. https://doi.org/10.3329/bjp.v8i3.15620
Li X, Liu Y, Deng F, Wang C, Qu S (2000) Microcalorimetric study of the toxic effect of sodium selenite on the mitochondria metabolism of Carassius auratus liver. Biol Trace Elem Res 77(3):261–272. https://doi.org/10.1385/bter:77:3:261
Li H-R, Qin C-Q, Zhang Z-H, Zhu J-C, Liu Y (2006) Stimulation effect of lithium on the metabolic activity of liver tissue mitochondria measured by microcalorimetry. Biol Trace Elem Res 114(1–3):163–174. https://doi.org/10.1385/bter:114:1:163
Li J, Zhang Y, Xiao Q, Tian F, Liu X, Li R, Zhao G, Jiang F, Liu Y (2011) Mitochondria as target of quantum dots toxicity. J Hazard Mater 194:440–444. https://doi.org/10.1016/j.jhazmat.2011.07.113
Lv W, Sheng X, Chen T, Xu Q, Xie X (2008) Jaceosidin induces apoptosis in human ovary cancer cells through mitochondrial pathway. J Biomed Biotechnol 2008:394802. https://doi.org/10.1155/2008/394802
Ma L, Dong JX, Fu WR, Li XY, Chen J, Liu Y (2018) Mitochondrial morphology and function impaired by dimethyl sulfoxide and dimethyl Formamide. J Bioenergy Biomembr 50(4):297–305. https://doi.org/10.1007/s10863-018-9759-7
Marchesi E, Rota C, Fann YC, Chignell CF, Mason RP (1999) Photoreduction of the fluorescent dye 2′-7′-dichlorofluorescein: a spin trapping and direct electron spin resonance study with implications for oxidative stress measurements. Free Radic Biol Med 26(1–2):148–161. https://doi.org/10.1016/s0891-5849(98)00174-9
Mignotte B, Vayssiere J-L (1998) Mitochondria and apoptosis. Eur J Biochem 252(1):1–15. https://doi.org/10.1046/j.1432-1327.1998.2520001.x
Narita M, Shimizu S, Ito T, Chittenden T, Lutz RJ, Matsuda H, Tsujimoto Y (1998) Bax interacts with the permeability transition pore to induce permeability transition and cytochrome c release in isolated mitochondria. Proc Natl Acad Sci USA 95(25):14681–14686. https://doi.org/10.1073/pnas.95.25.14681
Ouyang Z, Li W, Meng Q, Zhang Q, Wang X, Elgehama A, Wu X, Shen Y, Sun Y, Wu X, Xu Q (2017) A natural compound jaceosidin ameliorates endoplasmic reticulum stress and insulin resistance via upregulation of SERCA2b. Biomed Pharmacother 89:1286–1296. https://doi.org/10.1016/j.biopha.2017.03.023
Park E, Kwon B-M, Jung I-K, Kim J-H (2014) Hypoglycemic and antioxidant effects of jaceosidin in streptozotocin-induced diabetic mice. J Nutr Health 47(5):313–320. https://doi.org/10.4163/jnh.2014.47.5.313
Peng L, Yi L, Jia H, Fengjiao D, Daiwen P, Songsheng Q (2003) Microcalorimetric investigation of the effect of La3+ on mitochondria isolated from avian chicken liver tissue cells. J Therm Anal Calorim 73(3):843–849. https://doi.org/10.1023/a:1025842800225
Perez-Hernandez IH, Dominguez-Fuentes JM, Palomar-Morales M, Zazueta-Mendizabal AC, Baiza-Gutman A, Mejia-Zepeda R (2017) Liver mitochondrial membrane fluidity at early development of diabetes and its correlation with the respiration. J Bioenergy Biomembr 49(3):231–239. https://doi.org/10.1007/s10863-017-9700-5
Sueishi Y, Ishikawa M, Yoshioka D, Endoh N, Oowada S, Shimmei M, Fujii H, Kotake Y (2012) Oxygen radical absorbance capacity (ORAC) of cyclodextrin-solubilized flavonoids, resveratrol and astaxanthin as measured with the ORAC-EPR method. J Clin Biochem Nutr 50(2):127–132. https://doi.org/10.3164/jcbn.11-21
Tueller DJ, Harley JS, Hancock CR (2017) Effects of curcumin and ursolic acid on the mitochondrial coupling efficiency and hydrogen peroxide emission of intact skeletal myoblasts. Biochem Biophys Res Commun 492(3):368–372. https://doi.org/10.1016/j.bbrc.2017.08.101
Vakifahmetoglu-Norberg H, Ouchida AT, Norberg E (2017) The role of mitochondria in metabolism and cell death. Biochem Biophys Res Commun 482(3):426–431. https://doi.org/10.1016/j.bbrc.2016.11.088
Woo SM, Kwon TK (2016) Jaceosidin induces apoptosis through Bax activation and down-regulation of Mcl-1 and c-FLIP expression in human renal carcinoma Caki cells. Chem-Biol Interact 260:168–175. https://doi.org/10.1016/j.cbi.2016.10.011
Wright JS (2002) Predicting the antioxidant activity of curcumin and curcuminoids. J Mol Struc-Theochem 591(1–3):207–217. https://doi.org/10.1016/s0166-1280(02)00242-7
Yang LY, Gao JL, Gao T, Dong P, Ma L, Jiang FL, Liu Y (2016) Toxicity of polyhydroxylated fullerene to mitochondria. J Hazard Mater 301:119–126. https://doi.org/10.1016/j.jhazmat.2015.08.046
Yi L, Peng L, Fengjiao D, Zhong C, Cunxin W, Songsheng Q (2001) A microcalorimetric study of the effect of La3+ on mitochondria isolated from Star-Cross 288 chicken liver tissue cells. Biol Trace Elem Res 84(1–3):103–112. https://doi.org/10.1385/bter:84:1-3:103
Yuan L, Gao T, He H, Jiang FL, Liu Y (2017) Silver ion-induced mitochondrial dysfunction via a nonspecific pathway. Toxicol Res (Camb) 6(5):621–630. https://doi.org/10.1039/c7tx00079k
Zhang S, Fu J, Zhou Z (2004) In vitro effect of manganese chloride exposure on reactive oxygen species generation and respiratory chain complexes activities of mitochondria isolated from rat brain. Toxicol In Vitro 18(1):71–77. https://doi.org/10.1016/j.tiv.2003.09.002
Acknowledgements
This work was supported by the National Natural Science Foundation of China (Grant No. 21463008) and Bagui Scholar Program of Guangxi Province (2016).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical Approval
The Wistar rats used in this work were handled according to the Guidelines of the China Animal Welfare Legislation, as approved by the Committee on Ethics in the Care and Use of Laboratory Animals of the College of Life Sciences, Wuhan University.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Fu, WR., Chen, JL., Li, XY. et al. Bidirectional Regulatory Mechanisms of Jaceosidin on Mitochondria Function: Protective Effects of the Permeability Transition and Damage of Membrane Functions. J Membrane Biol 253, 25–35 (2020). https://doi.org/10.1007/s00232-019-00102-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00232-019-00102-4