Abstract
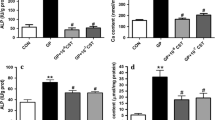
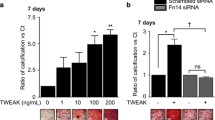
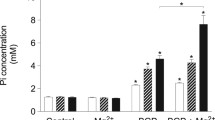
Arterial medial calcification (AMC) has been associated with phenotypic changes in vascular smooth muscle cells (VSMCs) that reportedly makes them more osteoblast-like. Previous work has shown that ATP/UTP can inhibit AMC directly via P2 receptors and indirectly by NPP1-mediated hydrolysis to produce the mineralisation inhibitor, pyrophosphate (PPi). This study investigated the role of P2X receptors in the inhibitory effects of extracellular nucleotides on VSMC calcification. We found that Bz-ATP, α,β-meATP and β,γ-meATP inhibited calcification by up to 100%. Culture in a high-phosphate medium (2 mM) was associated with increased VSMC death and apoptosis; treatment with Bz-ATP, α,β-meATP and β,γ-meATP reduced apoptosis to levels seen in non-calcifying cells. Calcification was also associated with alterations in the protein levels of VSMC (e.g. SM22α and SMA) and osteoblast-associated (e.g. Runx2 and osteopontin) markers; Bz-ATP, α,β-meATP and β,γ-meATP attenuated these changes in protein expression. Long-term culture with Bz-ATP, α,β-meATP and β,γ-meATP resulted in lower extracellular ATP levels and an increased rate of ATP breakdown. P2X receptor antagonists failed to prevent the inhibitory effects of these analogues suggesting that they act via P2X receptor-independent mechanisms. In agreement, the breakdown products of α,β-meATP and β,γ-meATP (α,β-meADP and methylene diphosphonate, respectively) also dose-dependently inhibited VSMC calcification. Furthermore, the actions of Bz-ATP, α,β-meATP and β,γ-meATP were unchanged in VSMCs isolated from NPP1-knockout mice, suggesting that the functional effects of these compounds do not involve NPP1-mediated generation of PPi. Together, these results indicate that the inhibitory effects of ATP analogues on VSMC calcification and apoptosis in vitro may be mediated, at least in part, by mechanisms that are independent of purinergic signalling and PPi.








Similar content being viewed by others
References
Zhu D, Mackenzie NC, Farquharson C, Macrae VE (2012) Mechanisms and clinical consequences of vascular calcification. Front Endocrinol (Lausanne) 3:95
Proudfoot D, Skepper JN, Hegyi L, Bennett MR, Shanahan CM et al (2000) Apoptosis regulates human vascular calcification in vitro: evidence for initiation of vascular calcification by apoptotic bodies. Circ Res 87:1055–1062
Narisawa S, Harmey D, Yadav MC, O’Neill WC, Hoylaerts MF et al (2007) Novel inhibitors of alkaline phosphatase suppress vascular smooth muscle cell calcification. J Bone Miner Res 22:1700–1710
Zhu D, Mackenzie NC, Millan JL, Farquharson C, Macrae VE (2011) The appearance and modulation of osteocyte marker expression during calcification of vascular smooth muscle cells. PLoS ONE 6:e19595
Shroff RC, Shanahan CM (2007) The vascular biology of calcification. Semin Dial 20:103–109
Patel JJ, Zhu D, Opdebeeck B, D’Haese P, Millan JL et al (2018) Inhibition of arterial medial calcification and bone mineralization by extracellular nucleotides: the same functional effect mediated by different cellular mechanisms. J Cell Physiol 233:3230–3243
Kapustin AN, Davies JD, Reynolds JL, McNair R, Jones GT et al (2011) Calcium regulates key components of vascular smooth muscle cell-derived matrix vesicles to enhance mineralization. Circ Res 109:e1–e12
Burnstock G (2007) Physiology and pathophysiology of purinergic neurotransmission. Physiol Rev 87:659–797
Burnstock G, Kennedy C (1985) Is there a basis for distinguishing two types of P2-purinoceptor? Gen Pharmacol 16:433–440
Abbracchio MP, Burnstock G (1994) Purinoceptors: are there families of P2X and P2Y purinoceptors? Pharmacol Ther 64:445–475
North RA, Surprenant A (2000) Pharmacology of cloned P2X receptors. Annu Rev Pharmacol Toxicol 40:563–580
Burnstock G, Ralevic V (2014) Purinergic signaling and blood vessels in health and disease. Pharmacol Rev 66:102–192
Fish RS, Klootwijk E, Tam FW, Kleta R, Wheeler DC et al (2013) ATP and arterial calcification. Eur J Clin Investig 43:405–412
Cote N, El HD, Pepin A, Guauque-Olarte S, Ducharme V et al (2012) ATP acts as a survival signal and prevents the mineralization of aortic valve. J Mol Cell Cardiol 52:1191–1202
Qian S, Regan JN, Shelton MT, Hoggatt A, Mohammad KS et al (2017) The P2Y2 nucleotide receptor is an inhibitor of vascular calcification. Atherosclerosis 257:38–46
Villa-Bellosta R, Sorribas V (2013) Prevention of vascular calcification by polyphosphates and nucleotides-role of ATP. Circ J 77:2145–2151
Prosdocimo DA, Douglas DC, Romani AM, O’Neill WC, Dubyak GR (2009) Autocrine ATP release coupled to extracellular pyrophosphate accumulation in vascular smooth muscle cells. Am J Phys Cell Physiol 296:C828–C839
Lohman AW, Billaud M, Isakson BE (2012) Mechanisms of ATP release and signalling in the blood vessel wall. Cardiovasc Res 95:269–280
Fleisch H, Bisaz S (1962) Mechanism of calcification: inhibitory role of pyrophosphate. Nature 195:911
Orriss IR, Arnett TR, Russell RG (2016) Pyrophosphate: a key inhibitor of mineralisation. Curr Opin Pharmacol 28:57–68
Sali A, Favaloro JM, Terkeltaub R, Goding JW (1999) Germline deletion of the nucleoside triphosphate pyrophosphohydrolase (NTPPPH) plasma cell membrane glycoprotein (PC-1) produces abnormal calcification of the periarticular tissues. In: Vanduffe L, Lemmens R (eds) Ecto-ATPases and related ectonucleotides. Shaker Publishing BV, Mastricht, pp 267–282
Orriss IR, Key ML, Brandao-Burch A, Patel JJ, Burnstock G et al (2012) The regulation of osteoblast function and bone mineralisation by extracellular nucleotides: the role of P2X receptors. Bone 51:389–400
Taylor SE, Shah M, Orriss IR (2014) Generation of rodent and human osteoblasts. BoneKey Rep 3:585
Orriss IR, Knight GE, Utting JC, Taylor SE, Burnstock G et al (2009) Hypoxia stimulates vesicular ATP release from rat osteoblasts. J Cell Physiol 220:155–162
Joseph SM, Pifer MA, Przybylski RJ, Dubyak GR (2004) Methylene ATP analogs as modulators of extracellular ATP metabolism and accumulation. Br J Pharmacol 142:1002–1014
Zimmermann H, Zebisch M, Strater N (2012) Cellular function and molecular structure of ecto-nucleotidases. Purinergic Signal 8:437–502
Villa-Bellosta R, Wang X, Millan JL, Dubyak GR, O’Neill WC (2011) Extracellular pyrophosphate metabolism and calcification in vascular smooth muscle. Am J Physiol Heart Circ Physiol 301:H61–H68
Mackenzie NC, Huesa C, Rutsch F, Macrae VE (2012) New insights into NPP1 function: lessons from clinical and animal studies. Bone 51:961–968
Sheen CR, Kuss P, Narisawa S, Yadav MC, Nigro J et al (2015) Pathophysiological role of vascular smooth muscle alkaline phosphatase in medial artery calcification. J Bone Miner Res 30:824–836
Orriss IR, Utting JC, Brandao-Burch A, Colston K, Grubb BR et al (2007) Extracellular nucleotides block bone mineralization in vitro: evidence for dual inhibitory mechanisms involving both P2Y2 receptors and pyrophosphate. Endocrinology 148:4208–4216
Kukley M, Stausberg P, Adelmann G, Chessell IP, Dietrich D (2004) Ecto-nucleotidases and nucleoside transporters mediate activation of adenosine receptors on hippocampal mossy fibers by P2X7 receptor agonist 2'-3'-O-(4-benzoylbenzoyl)-ATP. J Neurosci 24:7128–7139
Sugitani H, Wachi H, Murata H, Sato F, Mecham RP et al (2003) Characterization of an in vitro model of calcification in retinal pigmented epithelial cells. J Atheroscler Thromb 10:48–56
Bauer C, le Saux O, Pomozi V, Aherrahrou R, Kriesen R et al (2018) Etidronate prevents dystrophic cardiac calcification by inhibiting macrophage aggregation. Sci Rep 8:5812
Miyai K, Ariyasu D, Numakura C, Yoneda K, Nakazato H et al (2015) Hypophosphatemic rickets developed after treatment with etidronate disodium in a patient with generalized arterial calcification in infancy. Bone Rep 3:57–60
Orriss IR, Key ML, Colston KW, Arnett TR (2009) Inhibition of osteoblast function in vitro by aminobisphosphonates. J Cell Biochem 106:109–118
Idris AI, Rojas J, Greig IR, van’t Hof RJ, Ralston SH (2008) Aminobisphosphonates cause osteoblast apoptosis and inhibit bone nodule formation in vitro. Calcif Tissue Int 82:191–201
Jacobson KA, Ivanov AA, de Castro S, Harden TK, Ko H (2009) Development of selective agonists and antagonists of P2Y receptors. Purinergic Signal 5:75–89
Funding
The authors are grateful for the funding from the British Heart Foundation (grant number PG/15/13/31296) and the Biotechnology and Biological Sciences Research Council (BBSRC) in the form of Institute Strategic Programme grants (BB/J004316/1 and BB/P013732/1).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
This article does not contain any studies with human participants or animals performed by any of the authors.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Patel, J.J., Bourne, L.E., Millán, J.L. et al. Inhibition of vascular smooth muscle cell calcification by ATP analogues. Purinergic Signalling 15, 315–326 (2019). https://doi.org/10.1007/s11302-019-09672-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11302-019-09672-3