Abstract
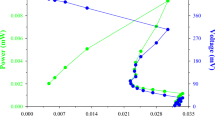
The production of biosurfactant by Pseudomonas aeruginosa TGC01 using crude glycerol and sodium nitrate as the sole substrate and nitrogen source, respectively, was investigated using two mineral culture media. Two inoculum sizes (5 and 10% v/v) and two volumes of the culture medium (50 and 100 mL) in 500 mL Erlenmeyer flask also were used. Enzymatic hydrolyses of waste office paper (WOP), newspaper (NP) and eucalyptus wood chips (EWC) were carried out using the biosurfactant from P. aeruginosa TGC01. The decrease in volume of the culture medium increased the production of rhamnolipid by 500% in relation to concentration obtained when higher volume of culture medium was used. High quantity biosurfactant was recovered (11 g/L) with desired surface active properties after extraction using chloroform:methanol (v/v). The biosurfactant was able to reduce the water surface tension from 72 to 27 mN/m with a critical micelle concentration (CMC) of 100 mg/L and a stable emulsion index (above 60%) in the enzymatic hydrolysis (pH 4.8 and 50 °C for 4 h). Biosurfactant increased the glucose released in the enzymatic hydrolysis in relation to control (without tensoactive) when WOP (19% increase) and NP (113% increase) were used. The process for NP (18% lignin) was economical, given that the biosurfactant present made a delignification process unnecessary.





Similar content being viewed by others
References
Abalos A, Pinazo A, Infante M (2001) Physicochemical and antimicrobial properties of new rhamnolipids produced by Pseudomonas aeruginosa AT10 from soybean oil refinery wastes. Langmuir 17:1367–1371. https://doi.org/10.1021/la0011735
Abdel-Mawgoud AM, Hausmann R, Lépine F et al (2011) Rhamnolipids: detection, analysis, biosynthesis, genetic regulation, and bioengineering of production. Biosurfactants Genes Appl 20:13–56. https://doi.org/10.1007/978-3-642-14490-5
Almeida DG, Da Silva RDS, Luna JM, Rufino RD, Santos VA, Banat IM, Sarubbo LA (2016) Biosurfactants: promising molecules for petroleum biotechnology advances. Front Microbiol 7:1–14. https://doi.org/10.3389/fmicb.2016.01718
Araújo L, Guimarães C, Silva R, Santiago V, Souza M, Nitschke M, Freire D (2016) Rhamnolipid and surfactin: anti-adhesion/antibiofilm and antimicrobial effects. Food Control 61:171–178. https://doi.org/10.1016/j.foodcont.2015.11.036
Banat I, Makkar R, Cameotra S (2000) Potential commercial applications of microbial surfactants. Appl Microbiol Biotechnol 53:495–508. https://doi.org/10.1007/s002530051648
Barber W, Stuckey D (2000) Nitrogen removal in a modified anaerobic baffled reactor (ABR): 1, denitrification. Water Res 10:2413–2422. https://doi.org/10.1016/S0043-1354(99)00425-X
Benincasa M, Contiero J, Manresa M (2000) Rhamnolipid production by Pseudomonas aeruginosa LBI growing on soapstock as the sole carbon source. J Food Eng 54:283–288. https://doi.org/10.1016/S0260-8774(01)00214-X
Carvalho D, Queiroz J, Colodette J (2016) Assessment of alkaline pretreatment for the production of bioethanol from eucalyptus, sugarcane bagasse and sugarcane straw. Ind Crop Prod 94:932–941. https://doi.org/10.1016/j.indcrop.2016.09.069
Chandra RP, Bura R, Mabee BA, Pan X, Saddler JN (2007) Substrate pretreatment the key to effective enzymatic hydrolysis of lignocellulosics? Adv Biochem Eng Biotechnol 108:67–93. https://doi.org/10.1007/10_2007_064
Chrzanowski Ł, Ławniczak Ł, Czaczyk K (2012) Why do microorganisms produce rhamnolipids? World J Microb Biotechnol 28:401–419. https://doi.org/10.1007/s11274-011-0854-8
Chu K, Feng X (2013) Enzymatic conversion of newspaper and office paper to fermentable sugars. Process Saf Environ 91:123–130. https://doi.org/10.1016/j.psep.2011.11.006
Cooper D, Goldenberg B (1987) Surface-active agents from two Bacillus species. Appl Environ Microb 53:224–229
de Cássia FR, Almeida DG, Meira HM, Silva EJ, Farias CBB, Rufino RD, Luna JM, Sarubbo LA (2017) Production and characterization of a new biosurfactant from Pseudomonas cepacia grown in low-cost fermentative médium and its application in the oil industry. Biocatal Agric Biotechnol 12:206–215. https://doi.org/10.1016/j.bcab.2017.09.004
Eriksson T, Borjesson J, Tjerneld F (2002) Mechanism of surfactant effect in enzymatic hydrolysis of lignocellulose. Enzyme Microb Technol 31:353–364. https://doi.org/10.1016/S0141-0229(02)00134-5
Gudina E, Teixeira J, Rodrigues L (2010) Isolation and functional characterization of a biosurfactant produced by Lactobacillus paracasei. Colloid Surf 76:298–304. https://doi.org/10.1016/j.colsurfb.2009.11.008
Jiang Y, Li Y, Kang X, Zhou Q, Zhou X, Zhu Y (2012) Characteristics of leakage pollution of longpan road petrol station and its enlightenment. J Environ Protect 3:49–54. https://doi.org/10.4236/jep.2012.31006
Kaar W, Holtzapple M (1998) Benefits from tween during enzymic hydrolysis of corn stove. Biotechnol Bioeng 59:419–427
Kaya T, Aslim B, Kariptaş E (2014) Production of biosurfactant by Pseudomonas spp. isolated from industrial waste in Turkey. Turk J Biol 38:307–317. https://doi.org/10.3906/biy-1303-18
Lima D, Luna R, Martín C, Gouveia E (2015) Comparison of bioethanol production from acid hydrolyzates of waste office paper using Saccharomyces cerevisiae and Spathaspora passalidarum. Cell Chem Technol 49:463–469. https://doi.org/10.5151/chemeng-cobeq2014-0438-25490-177845
Liu J, Wu G, Yang S, Mu B (2014) Structural characterization of rhamnolipid produced by Pseudomonas aeruginosa strain FIN2 isolated from oil reservoir water. World J Microb Biotechnol 30:1473–1484. https://doi.org/10.1007/s11274-013-1565-0
Mercadé M, Manresa M, Robert M (1993) Olive oil mill effluent (OOME): new substrates for biosurfactant production. Bioresour Technol 43:1–6
Nonaka H, Kobayashi A, Funaoka M (2013) Lignin isolated from steam-exploded eucalyptus wood chips by phase separation and its affinity to Trichoderma reesei celulase. Bioresour Technol 140:431–434. https://doi.org/10.1016/j.biortech.2013.04.109
Palittapongarnpim M, Pokethitiyook P, Upatham E, Tangbanluekal L (1988) Biodegradation of crude oil by soil microorganisms in the tropic. Biodegradation 9:83–90. https://doi.org/10.1023/A:1008272303740
Patel RM, Desai AJ (2003) Biosurfactant production by GS3 from molasses. Lett Appl Microbiol 25(2):91–94
Rahman K, Rahman T, Mcclean S, Marchant R, Banat I (2002) Rhamnolipid biosurfactant production by strains of Pseudomonas aeruginosa using low cost raw materials. Biotechnol Prog 18:1277–1281. https://doi.org/10.1021/bp020071x
Rikalović M, Gojgić-Cvijović G, Vrvić M, Karadžić I (2012) Production and characterization of rhamnolipids from Pseudomonas aeruginosa san-ai. J Serb Chem Soc 77:27–42. https://doi.org/10.2298/JSC110211156R
Rocha J, Alencar B, Mota H, Gouveia E (2016) Enzymatic hydrolysis of waste office paper for ethanol production by Spathaspora passalidarum. Cell Chem Technol 50:243–246
Santos DKF, Rufino RD, Luna JM, Santos VA, Sarubbo LA (2016) Biosurfactants: multifunctional biomolecules of the 21st century. Int J Mol Sci 17:401. https://doi.org/10.3390/ijms17030401
Schmitt E, Bura R, Gustafson R, Cooper J, Vajzovi A (2012) Converting lignocellulosic solid waste into ethanol for the State of Washington: an investigation of treatment technologies and environmental impacts. Bioresour Technol 104:400–409. https://doi.org/10.1016/j.biortech.2011.10.094
Silva SNRL, Farias CBB, Rufino RD, Luna JM, Sarubbo LA (2010) Glycerol as substrate for the production of biosurfactant by Pseudomonas aeruginosa UCP0992. Colloids Surf B Biointerfaces 79:174–183. https://doi.org/10.1016/j.colsurfb.2010.03.050
Silva EJ, Silva NMPR, Rufino RD, Luna JM, Silva RO, Sarubbo LA (2014) Characterization of a biosurfactant produced by Pseudomonas cepacia CCT6659 in the presence of industrial wastes and its application in the biodegradation of hydrophobic compounds in soil. Colloids Surf B 117:36–41. https://doi.org/10.1016/j.colsurfb.2014.02.012
Sousa J, Correia J, Melo V, Gonçalves L, Cruz A (2014) Cinética e caracterização de ramnolipídeos produzidos por Pseudomonas aeruginosa MSIC02 utilizando glicerol como fonte de carbono. Quim Nova 37:431–441. https://doi.org/10.5935/0100-4042.20140064
Vasconcelos U, Oliveira F, França F (2013) Raw glycerol as cosubstrate on the PHAs biodegradation in soil. Can J Pure Appl Sci 7:2203–2209
Wang W, Fan B, Li C, Liu S, Li M (2011) Effects of rhamnolipid on the cellulase and xylanase in hydrolysis of wheat straw. Bioresour Technol 102:6515–6521. https://doi.org/10.1016/j.biortech.2011.02.102
Wang L, Sharifzadeh M, Templer R, Murphy R (2013) Bioethanol production from various waste papers: economic feasibility. Appl Energy 111:1172–1182. https://doi.org/10.1016/j.apenergy.2012.08.048
Wei Y, Chou C, Chang J (2005) Rhamnolipid production by indigeneous Pseudomonas aeruginosa J4 originating from petrochemical wastewater. Biochem Eng J 27:146–154. https://doi.org/10.1016/j.bej.2005.08.028
Wu J, Yeh K, Lu W, Lin C, Chang J (2008) Rhamnolipid production with indigenous Pseudomonas aeruginosa EM1 isolated from oil contaminated site. Bioresour Technol 99:1157–1164. https://doi.org/10.1016/j.biortech.2007.02.026
Zhang Q, He G, Wang J, Cai W, Xu Y (2009) Mechanisms of the stimulatory effects of rhamnolipid biosurfactant on rice straw hydrolysis. Appl Energy 86:S233–S237. https://doi.org/10.1016/j.apenergy.2009.04.030
Acknowledgements
The authors acknowledge financial support from the Fundação de Amparo à Ciência e Tecnologia do Estado de Pernambuco (FACEPE), Recife, PE, Brazil and the America Biomass Technologies (Dr. Henrique Baudel), São Paulo, Brazil for donation of the eucalyptus wood chips. The English text of this paper was revised by Sidney Pratt, Canadian, MAT (The Johns Hopkins University), RSAdip—TESL (Cambridge University).
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Bezerra, K.G.O., Gomes, U.V.R., Silva, R.O. et al. The potential application of biosurfactant produced by Pseudomonas aeruginosa TGC01 using crude glycerol on the enzymatic hydrolysis of lignocellulosic material. Biodegradation 30, 351–361 (2019). https://doi.org/10.1007/s10532-019-09883-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10532-019-09883-w