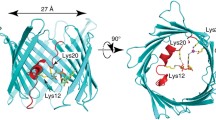
Abstract
It was recently asserted that the voltage-dependent anion channel (VDAC) serves as a global regulator, or governor, of mitochondrial function (Lemasters and Holmuhamedov, Biochim Biophys Acta 1762:181–190, 2006). Indeed, VDAC, positioned on the interface between mitochondria and the cytosol (Colombini, Mol Cell Biochem 256:107–115, 2004), is at the control point of mitochondria life and death. This large channel plays the role of a “switch” that defines in which direction mitochondria will go: to normal respiration or to suppression of mitochondria metabolism that leads to apoptosis and cell death. As the most abundant protein in the mitochondrial outer membrane (MOM), VDAC is known to be responsible for ATP/ADP exchange and for the fluxes of other metabolites across MOM. It controls them by switching between the open and “closed” states that are virtually impermeable to ATP and ADP. This control has dual importance: in maintaining normal mitochondria respiration and in triggering apoptosis when cytochrome c and other apoptogenic factors are released from the intermembrane space into the cytosol. Emerging evidence indicates that VDAC closure promotes apoptotic signals without direct involvement of VDAC in the permeability transition pore or hypothetical Bax-containing cytochrome c permeable pores. VDAC gating has been studied extensively for the last 30 years on reconstituted VDAC channels. In this review we focus exclusively on physiologically relevant regulators of VDAC gating such as endogenous cytosolic proteins and mitochondrial lipids. Closure of VDAC induced by such dissimilar cytosolic proteins as pro-apoptotic tBid and dimeric tubulin is compared to show that the involved mechanisms are rather distinct. While tBid mostly modulates VDAC voltage gating, tubulin blocks the channel with the efficiency of blockage controlled by voltage. We also discuss how characteristic mitochondrial lipids, phospatidylethanolamine and cardiolipin, could regulate VDAC gating. Overall, we demonstrate that VDAC gating is not just an observation made under artificial conditions of channel reconstitution but is a major mechanism of MOM permeability control.
Similar content being viewed by others
References
Andre N, Carre M, Brasseur G, Pourroy B, Kovacic H, Briand C, Braguer D (2002) FEBS Lett 532:256–260
Appaix F, Kuznetsov AV, Usson Y, Kay L, Andrienko T, Olivares J, Kaambre T, Sikk P, Margreiter R, Saks V (2003) Exp Physiol 88:175–190
Ardail D, Privat JP, Egretcharlier M, Levrat C, Lerme F, Louisot P (1990) J Biol Chem 265:18797–18802
Azoulay-Zohar H, Israelson A, Abu-Hamad S, Shoshan-Barmatz V (2004) Biochem J 377:347–355
Baines CP, Kaiser RA, Sheiko T, Craigen WJ, Molkentin JD (2007) Nat Cell Biol 9:550–555
Basanez G, Sharpe JC, Galanis J, Brandt TB, Hardwick JM, Zimmerberg J (2002) J Biol Chem 277:49360–49365
Bernardi P, Krauskopf A, Basso E, Petronilli V, Blalchy-Dyson E, Di Lisa F, Forte MA (2006) FEBS J 273:2077–2099
Bernier-Valentin F, Rousset B (1982) J Biol Chem 257:7092–7099
Beutner G, Ruck A, Riede B, Welte W, Brdiczka D (1996) FEBS Lett 396:189–195
Bezrukov SM (2000) Curr Opin Colloid Interface Sci 5:237–243
Bezrukov SM, Rand RP, Vodyanoy I, Parsegian VA (1998) Faraday Discuss 111:173–183
Brink-van der Laan EV, Killian JA, de Kruijff B (2004) Biochim Biophys Acta 1666:275–288
Cantor RS (1999) Biophys J 76:2625–2639
Carre M, Andre N, Carles G, Borghi H, Brichese L, Briand C, Braguer D (2002) J Biol Chem 277:33664–33669
Colombini M (1980) J Membr Biol 53:79–84
Colombini M (1989) J Membr Biol 111:103–111
Colombini M (2004) Mol Cell Biochem 256:107–115
Colombini M, Yeung CL, Tung J, König T (1987) Biochim Biophys Acta 905:279–286
Colombini M, Blachly-Dyson E, Forte M (1996) In: Narahashi T (ed) Ion channels, vol. 4. Plenum, New York, pp 169–202
Cortese JD, Voglino AL, Hackenbrock CR (1992) Biochim Biophys Acta 1100:189–197
Crompton M (1999) Biochem J 341:233–249
Doring C, Colombini M (1985) J Membr Biol 83:81–86
Epand RF, Martinou JC, Fornallaz-Mulhauser M, Hughes DW, Epand RM (2002) J Biol Chem 277:32632–32639
Esteve MA, Carre M, Braguer D (2007) Curr Cancer Drug Targets 7:713–729
Galluzzi L, Kroemer G (2007) Nat Cell Biol 9:487–489
Goforth RL, Chi AK, Greathouse DV, Providence LL, Koeppe RE, Andersen OS (2003) J Gen Physiol 121:477–493
Gruner SM (1985) Proc Natl Acad Sci USA 82:3665–3669
Gullingsrud J, Schulten K (2004) Biophys J 86:3496–3509
Hodge T, Colombini M (1997) J Membr Biol 157:271–279
Keller SL, Bezrukov SM, Gruner SM, Tate MW, Vodyanoy I, Parsegian VA (1993) Biophys J 65:23–27
Krasilnikov OV, Sabirov RZ, Ternovsky VI, Merzliak PG, Muratkhodjaev JN (1992) FEMS Microbiol Immunol 105:93–100
Krauskopf A, Eriksson O, Craigen WJ, Forte MA, Bernardi P (2006) Biochim Biophys Acta 1757:590–595
Kuwana T, Mackey MR, Perkins G, Ellisman MH, Latterich M, Schneiter R, Green DR, Newmeyer DD (2002) Cell 111:331–342
Lai JC, Tan WZ, Benimetskaya L, Miller P, Colombini M, Stein CA (2006) Proc Natl Acad Sci USA 103:7494–7499
Lee AC, Zizi M, Colombini M (1994) J Biol Chemi 269:30974–30980
Lee AC, Xu XF, Colombini M (1996) J Biol Chemi 271:26724–26731
Lemasters JJ, Holmuhamedov E (2006) Biochim Biophys Acta 1762:181–190
Lemeshko VV (2006) Eur Biophys J 36:57–66
Li XX, Vander Heiden MG, Thompson CB, Colombini M (2001) Biophysi J 80:239A–239A
Lutter M, Fang M, Luo X, Nishijima M, Xie XS, Wang XD (2000) Nat Cell Biol 2:754–756
Majewski N, Nogueira V, Bhaskar P, Coy PE, Skeen JE, Gottlob K, Chandel NS, Thompson CB, Robey RB, Hay N (2004) Mol Cell 16:819–830
Mannella CA, Guo XW, Cognon B (1989) FEBS Lett 253:231–234
Mikhailov V, Mikhailova M, Pulkrabek DJ, Dong Z, Venkatachalam MA, Saikumar P (2001) J Biol Chem 276:18361–18374
Minn AJ, Velez P, Schendel SL, Liang H, Muchmore SW, Fesik SW, Fill M, Thompson CB (1997) Nature 385:353–357
Monge C, Beraud N, Rostovtseva T, Sackett D, Vendelin M, Saks V (2008) Biophys J 94:1562
Pavlov EV, Priault M, Pietkiewicz D, Cheng EHY, Antonsson B, Manon S, Korsmeyer SJ, Mannella CA, Kinnally KW (2001) J Cell Biol 155:725–731
Peng S, Blachly-Dyson E, Forte M, Colombini M (1992) Biophys J 62:123–135
Polcic P, Forte M (2003) Biochem J 374:393–402
Porcelli AM, Ghelli A, Zanna C, Pinton P, Rizzuto R, Rugolo M (2005) Biochem Biophys Res Commun 326:799–804
Priault M, Chaudhuri B, Clow A, Camougrand N, Manon S (1999) Eur J Biochem 260:684–691
Robey RB, Hay N (2006) Oncogene 25:4683–4696
Rostovtseva T, Colombini M (1996) J Biol Chem 271:28006–28008
Rostovtseva T, Colombini M (1997) Biophys J 72:1954–1962
Rostovtseva TK, Komarov A, Bezrukov SM, Colombini M (2002a) Biophys J 82:193–205
Rostovtseva TK, Komarov A, Bezrukov SM, Colombini M (2002b) J Membr Biol 187:147–156
Rostovtseva TK, Antonsson B, Suzuki M, Youle RJ, Colombini M, Bezrukov SM (2004) J Biol Chem 279:13575–13583
Rostovtseva TK, Tan WZ, Colombini M (2005) J Bioenerg Biomembranes 37:129–142
Rostovtseva TK, Kazemi N, Weinrich M, Bezrukov SM (2006) J Biol Chem 281:37496–37506
Rostovtseva TK, Sackett DL, Sheldon K, Monge C, Saks V, Bezrukov SM (2008) Biophys J 94:1760
Saks VA, Kuznetsov AV, Khuchua ZA, Vasilyeva EV, Belikova JO, Kesvatera T, Tiivel T (1995) J Mol Cell Cardiol 27:625–645
Saks V, Dzeja P, Schlattner U, Vendelin M, Terzic A, Wallimann T (2006) J Physiol-London 571:253–273
Saks V, Vendelin M, Aliev MK, Kekelidze T, Engelbrecht J (2007) In: Gibson GF, Dienel G (eds) Brain energetics: integration of molecular and cellular processes. Springer, Berlin, pp 815–860
Shimizu S, Narita M, Tsujimoto Y (1999) Nature 399:483–487
Shimizu S, Ide T, Yanagida T, Tsujimoto Y (2000a) J Biol Chem 275:12321–12325
Shimizu S, Shinohara Y, Tsujimoto Y (2000b) Oncogene 19:4309–4318
Simbeni R, Pon L, Zinser E, Paltauf F, Daum G (1991) J Biol Chem 266:10047–10049
Song JM, Midson C, Blachly-Dyson E, Forte M, Colombini M (1998a) Biophys J 74:2926–2944
Song JM, Midson C, Blachly-Dyson E, Forte M, Colombini M (1998b) J Biol Chem 273:24406–24413
Suchyna TM, Tape SE, Koeppe RE, Andersen OS, Sachs F, Gottlieb PA (2004) Nature 430:235–240
Tan WZ, Colombini M (2007) Biochim Biophys Acta 1768:2510–2515
Tan WZ, Lai JC, Miller P, Stein CA, Colombini M (2007a) Am J Physiol Cell Physiol 292:C1388–C1397
Tan WZ, Loke YH, Stein CA, Miller P, Colombini M (2007b) Biophys J 93:1184–1191
Thomas L, Blachly-Dyson E, Colombini M, Forte M (1993) Proc Natl Acad Sci USA 90:5446–5449
Vander Heiden MG, Chandel NS, Li XX, Schumacker PT, Colombini M, Thompson CB (2000) Proc Natl Acad Sci USA 97:4666–4671
Vander Heiden MG, Li XX, Gottleib E, Hill RB, Thompson CB, Colombini M (2001) J Biol Chem 276:19414–19419
Xu X, Forbes JG, Colombini M (2001) J Membr Biol 180:73–81
Zimmerberg J, Parsegian VA (1986) Nature 323:36–39
Zizi M, Thomas L, Blachly-Dyson E, Forte M, Colombini M (1995) J Membr Biol 144:121–129
Zizi M, Byrd C, Boxus R, Colombini M (1998) Biophys J 75:704–713
Zoratti M, Szabo D, De Marchi U (2005) Biochim Biophys Acta 1706:40–52
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Rostovtseva, T.K., Bezrukov, S.M. VDAC regulation: role of cytosolic proteins and mitochondrial lipids. J Bioenerg Biomembr 40, 163–170 (2008). https://doi.org/10.1007/s10863-008-9145-y
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10863-008-9145-y