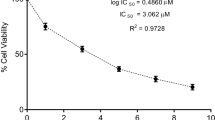
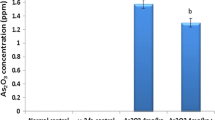
Abstract
Arsenic toxicity is one of the major environmental problems causing various diseases, cardiovascular disorders is one of them. Several epidemiological studies have shown that arsenic causes cardiac hypertrophy but the detailed molecular mechanism is to be studied yet. This study is designed to determine the molecules involved in the augmentation of arsenic-induced cardiac hypertrophy. Furthermore, the effects of oleic acid on arsenic-induced hypertrophy and cardiac injury have also been investigated. Our results show that arsenic induces cardiac hypertrophy both in vivo in mice and in vitro in rat H9c2 cardiomyocytes. Moreover, arsenic results in decreased activity of AMPK and FoxO1 along with increased NFATc3 expression, a known cardiac hypertrophy inducer. In addition, activation of AMPK and FoxO1 results in reduced NFATc3 expression causing attenuation of arsenic-induced cardiac hypertrophy in H9c2 cells. Interestingly, we have observed that oleic acid helps in ameliorating cardiac hypertrophy in arsenic-exposed mice. Our studies on protection from arsenic-induced cardiac hypertrophy by oleic acid in H9c2 cells shows that oleic acid activates AMPK along with increased nuclear FoxO1 localization, thereby reducing NFATc3 expression and attenuating cardiomyocyte hypertrophy. This study will help in finding out new avenues in treating arsenic-induced cardiac hypertrophy.










Similar content being viewed by others
Abbreviations
- AMPK:
-
Adenosine monophosphate-activated protein kinase
- FoxO:
-
Forkhead box transcription factor
- CVDs:
-
Cardiovascular-associated diseases
- ROS:
-
Reactive oxygen species
- OA:
-
Oleic acid
- i.p.:
-
Intraperitoneal
- ppm:
-
Parts per million
- IAEC:
-
Institutional Animal Ethics Committee
- DMEM:
-
Dulbecco’s modified Eagle’s medium
- FBS:
-
Fetal Bovine serum
- PBS:
-
Phosphate-buffered saline
- PCR:
-
Polymerase chain reaction
- PFA:
-
Paraformaldehyde
- WGA:
-
Wheat germ agglutinin
- BSA:
-
Bovine serum albumin
- ANP:
-
Atrial natriuretic peptide
- BNP:
-
Brain natriuretic peptide
- S.E.:
-
Standard error
References
Al-Shudiefat, A. A. R., Sharma, A. K., Bagchi, A. K., Dhingra, S., & Singal, P. K. (2013). Oleic acid mitigates TNF-a-induced oxidative stress in rat cardiomyocytes. Molecular and Cellular Biochemistry,372, 75–82.
Bairwa, S. C., Parajuli, N., & Dyck, J. R. B. (2016). The role of AMPK in cardiomyocyte health and survival. Biochimica et Biophysica Acta (BBA)-Molecular Basis of Disease,1862(12), 2199–2210.
Beauchamp, E. M., Kosciuczuk, E. M., Serrano, R., Nanavati, D., Swindell, E. P., Viollet, B., et al. (2015). Direct binding of arsenic trioxide to AMPK and generation of inhibitory effects on acute myeloid leukemia precursors. Molecular Cancer Therapeutics,14(1), 202–212.
Bhattacharya, S. (2016). Medicinal plants and natural products in amelioration of arsenic toxicity: A short review. Pharmaceutical Biology,55(1), 349–354.
Chan, A. Y. M., Dolinsky, V. W., Soltys, C. L. M., Viollet, B., Baksh, S., Light, P. E., et al. (2008). Resveratrol inhibits cardiac hypertrophy via AMP-activated protein kinase and Akt. The Journal of Biological Chemistry,283, 24194–24201.
Chan, A. Y., Soltys, C. L., Young, M. E., Proud, C. G., & Dyck, J. R. (2004). Activation of AMP-activated protein kinase inhibits protein synthesis associated with hypertrophy in the cardiac myocyte. Journal of Biological Chemistry,279, 32771–32779.
Divya, S. P., Pratheeshkumar, P., Son, Y. O., Roy, R. V., Hitron, J. A., Kim, D., et al. (2015). Arsenic induces insulin resistance in mouse adipocytes and myotubes via oxidative stress-regulated mitochondrial Sirt3-FOXO3a signaling pathway. Toxicological Sciences,146(2), 290–300.
Ellinsworth, D. C. (2015). Arsenic, reactive oxygen, and endothelial dysfunction. Journal of Pharmacology and Experimental Therapeutics,353(3), 458–464.
Fan, Y., Wang, C., Zhang, Y., Hang, P., Liu, Y., Pan, Z., et al. (2013). Genistein ameliorates adverse cardiac effects induced by arsenic trioxide through preventing cardiomyocytes apoptosis. Cellular Physiology and Biochemistry,31, 80–91.
Firdaus, F., Zafeer, M. F., Ahmad, M., & Afzal, M. (2018). Anxiolytic and anti-inflammatory role of thymoquinone in arsenic-induced hippocampal toxicity in Wistar rats. Heliyon,4(6), e00650.
Gonçalves-de-Albuquerque, C. F., Medeiros-de-Moraes, I. M., Oliveira, F. M. J., Burth, P., Bozza, P. T., Faria, M. V. C., et al. (2016). Omega-9 oleic acid induces fatty acid oxidation and decreases organ dysfunction and mortality in experimental sepsis. PLos ONE,11(4), 153607.
Hamann, I., Petroll, K., Hou, X., Anwar-Mohamed, A., El-Kadi, A. O., & Klotz, L. O. (2014). Acute and long-term effects of arsenite in HepG2 cells: Modulation of insulin signaling. BioMetals,27(2), 317–332.
Herum, K. M., Lunde, I. G., Skrbic, B., Florholmen, G., Behmen, D., Sjaastad, I., et al. (2013). Syndecan-4 signaling via NFAT regulates extracellular matrix production and cardiac myofibroblast differentiation in response to mechanical stress. Journal of Molecular and Cellular Cardiology,54, 73–81.
Davis, Jennifer, Burr, Adam R., Davis, Gregory F., Birnbaumer, Lutz, & Molkentin, Jeffery D. (2012). A TRPC6-dependent pathway for myofibroblast transdifferentiation and wound healing in vivo. Developmental Cell,23(4), 705–715.
Kris-Etherton, P. M. (1999). Monounsaturated fatty acids and risk of cardiovascular disease. Circulation,100, 1253–1258.
Li, H. L., Yin, R., Chen, D., Liu, D., Wang, D., Yang, Q., et al. (2007). Long-term activation of adenosine monophosphate-activated protein kinase attenuates pressure-overload-induced cardiac hypertrophy. Journal of Cellular Biochemistry,100, 1086–1099.
Liu, B., Pan, S., Dong, X., Qiao, H., Jiang, H., Krissansen, G. W., et al. (2006). Opposing effects of arsenic trioxide on hepatocellular carcinomas in mice. Cancer Science,97, 675–681.
Livingstone, K. M., Lovegrove, J. A., & Givens, D. I. (2012). The impact of substituting SFA in dairy products with MUFA or PUFA on CVD risk: Evidence from human intervention studies. Nutrition Research Reviews,25(2), 193–206.
Manna, P., Sinha, M., & Sil, P. C. (2008). Arsenic-induced oxidative myocardial injury: Protective role of arjunolic acid. Archives of Toxicology,82, 137–149.
Medeiros-de-Moraes, I. M., Gonçalves-de-Albuquerque, C. F., Kurz, R. M. A., Oliveira, F. M. J., Pereira de Abreu, V. H., Torres, R. C., et al. (2018). Omega-9 oleic acid, the main compound of olive oil, mitigates inflammation during experimental sepsis. Oxidative Medicine and Cellular Longevity. https://doi.org/10.1155/2018/6053492.
Miao, X., Tang, Z., Wang, Y., Su, G., Sun, W., Wei, W., et al. (2013). Metallothionein prevention of arsenic trioxide-induced cardiac cell death is associated with its inhibition of mitogen-activated protein kinases activation in vitro and in vivo. Toxicology Letters,220, 277–285.
Moon, K., Guallar, E., & Navas-Acien, A. (2012). Arsenic exposure and cardiovascular disease: An updated systematic review. Current Atherosclerosis Reports,14(6), 542–555.
Ni, Y. G., Berenji, K., Wang, N., Oh, M., Sachan, N., Dey, A., et al. (2006). Foxo transcription factors blunt cardiac hypertrophy by inhibiting calcineurin signaling. Circulation,114(11), 1159–1168.
Pace, C., Dagda, R., & Angermann, J. (2017). Antioxidants protect against arsenic induced mitochondrial cardio-toxicity. Toxics,5(4), 38.
Phan, N. N., Wang, C. Y., & Lin, Y. C. (2014). The novel regulations of MEF2A, CAMKK2, CALM3, and TNNI3 in ventricular hypertrophy induced by arsenic exposure in rats. Toxicology,324, 123–135.
Li, Qing, Dong, Qiu-Ting, Yang, Yue-Jin, Tian, Xia-Qiu, Jin, Chen, Huang, Pei-Sen, et al. (2016). AMPK-mediated cardioprotection of atorvastatin relates to the reduction of apoptosis and activation of autophagy in infarcted rat hearts. American Journal of Translational Research,8(10), 4160–4171.
Roehrl, M. H. A., Kang, S., Aramburu, J., Wagner, G., Rao, A., & Hogan, P. G. (2004). Selective inhibition of calcineurin-NFAT signaling by blocking protein–protein interaction with small organic molecules. PNAS,101(20), 7554–7559.
Rooij, E. V., Doevendans, P. A., Theije, C. C., Babiker, F. A., Molkentin, J. D., & Windt, L. J. D. (2002). Requirement of nuclear factor of activated T-cells in calcineurin-mediated cardiomyocyte hypertrophy. Journal of Biological Chemistry,277(50), 48617–48626.
Salvadó, L., Coll, T., Gómez-Foix, A. M., Salmerón, E., Barroso, E., Palomer, X., et al. (2013). Oleate prevents saturated-fatty-acid-induced ER stress, inflammation and insulin resistance in skeletal muscle cells through an AMPK-dependent mechanism. Diabetologia,56, 1372–1382.
Sanchez-Soria, P., Broka, D., Monks, S. L., & Camenisch, T. D. (2012). Chronic low-level arsenite exposure through drinking water increases blood pressure and promotes concentric left ventricular hypertrophy in female mice. Toxicologic Pathology,40(3), 504–512.
Sengupta, A., Kalinichenko, V. V., & Yutzey, K. E. (2013). FoxO and FoxM1 transcription factors have antagonistic functions in neonatal cardiomyocyte cell cycle withdrawal and IGF1 gene regulation. Circulation Research,112(2), 267–277.
Sengupta, A., Molkentin, J. D., Paik, J. H., Pinho, R. A. D., & Yutzey, K. E. (2011). FoxO transcription factors promote cardiomyocyte survival upon induction of oxidative stress. The Journal of Biological Chemistry,286(9), 7468–7478.
States, J. C., Srivastava, S., Chen, Y., & Barchowsky, A. (2009). Arsenic and cardiovascular disease. Toxicological Sciences,107(2), 312–323.
Tseng, C. H., Chong, C. K., Tseng, C. P., Hsueh, Y. M., Chiou, H. Y., Tseng, C. C., et al. (2003). Long-term arsenic exposure and ischemic heart disease in arseniasis-hyperendemic villages in Taiwan. Toxicology Letters,137(1–2), 15–21.
Wang, K., Long, B., Zhou, J., & Li, P. F. (2010). miR-9 and NFATc3 regulate myocardin in cardiac hypertrophy. The Journal of Biological Chemistry,285, 11903–11912.
Wilkins, B. J., De Windt, L. J., Bueno, O. F., Braz, J. C., Glascock, B. J., Kimball, T. F., et al. (2002). Targeted disruption of NFATc3, but not NFATc4, reveals an intrinsic defect in calcineurin-mediated cardiac hypertrophic growth. Molecular and Cellular Biology,22(21), 7603–7613.
Yu, H., Berkel, T. J. C., & Biessen, E. A. L. (2007). Therapeutic potential of VIVIT, a selective peptide inhibitor of nuclear factor of activated T cells, in cardiovascular disorders. Cardiovascular Drug Reviews,25(2), 175–187.
Yun, H., Park, S., Kim, M. J., Yang, W. K., Im, D. U., Yang, K. R., et al. (2014). AMP-activated protein kinase mediates the antioxidant effects of resveratrol through regulation of the transcription factor FoxO1. The FEBS Journal,281, 4421–4438.
Zhao, X. Y., Li, G. Y., Liu, Y., Chai, L. M., Chen, J. X., Zhang, Y., et al. (2008). Reseveratrol protects against arsenic trioxide-induced cardiotoxicity in vitro and in vivo. British Journal of Pharmacology,154, 105–113.
Acknowledgements
We thank SC lab members for their kind support in the initial cell culture studies. We thank Dr. Amalesh Samanta, Department of Pharmaceutical Technology, Jadavpur University for his assistance in the animal study. All the authors read and approved the manuscript.
Funding
This work was supported by Department of Biotechnology sponsored Bio-CARe (BT/Bio-CARe/01/9686/2013-14) grant and Central University Grant Commission-Start Up Grant [No. F.4.5(192-FRP)/2015(BSR)] to AS and Central University Grant Commission-Major Research Project Grant (MRP-MAJOR-BIOT-2013-12885); Central Govt. of India, Ministry of Science and Technology, Department of Biotechnology sponsored project Grant (BT/PR11785/BRB/10/1324/2014) and Department of Science & technology (DST-SERB, No.: EMR/2017/001382) to SC.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflict of interests.
Additional information
Handling editor: Y. James Kang.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Samanta, J., Mondal, A., Saha, S. et al. Oleic Acid Protects from Arsenic-Induced Cardiac Hypertrophy via AMPK/FoxO/NFATc3 Pathway. Cardiovasc Toxicol 20, 261–280 (2020). https://doi.org/10.1007/s12012-019-09550-9
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12012-019-09550-9