Abstract
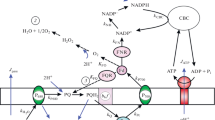
The regulation by adenylates of activities of various partial electron transport systems in spinach chloroplasts was studied using systems from H2O to 2,5-dimethyl-p-benzoquinone, H2O to 2,6-dichlorophenolindophenol, reduced 2,6-dichlorophenolindophenol to methyl viologen, and H2O to methyl viologen or ferricyanide. Adenylates regulated all of them. The ratio of the amount of esterified Pi (P) to that of electrons transported (Δe) in coupling with phosphorylation manifested that there are two phosphorylation sites: one between H2O and 2,5-dimethyl-p-benzoquinone or 2,6-dichlorophenolindophenol and another between reduced 2,6-dichlorophenolindophenol and methyl viologen, under the proposed stoichiometries,i.e., P/ΔH+=0.5 and ΔH+/Δe=1, where ΔH+ is the amount of protons pumped by electron transport (= those translocated during phosphorylation), when the basal electron transport (the part not regulated by adenylates) was excluded. The effects of pH, phlorizin, and methylamine on the adenylate regulation of electron transport, and the stimulation profile of electron transport coupled with quasiarsenylation suggested no distinction between the two phosphorylation sites.
Similar content being viewed by others
References
Y. Mukohata, T. Yagi, Y. Sugiyama, A. Matsuno, and M. Higashida,J. Bioenerg.,7 (1975) 91.
Y. Mukohata and T. Yagi,J. Bioenerg.,7 (1975) 111.
Y. Mukohata, T. Yagi, A. Matsuno, M. Higashida, and Y. Sugiyama,Plant Cell Physiol.,15 (1974) 163.
Y. Mukohata, T. Yagi, M. Higashida, A. Matsuno, and Y. Sugiyama,Plant Cell Physiol.,15 (1974) 619.
D. I. Arnon,Plant Physiol.,24 (1949) 1.
K. Asada, M. Takahashi, and M. Urano,Anal. Biochem.,48 (1972) 311.
R. Hill and F. Bendall,Nature,186 (1960) 136.
R. Ouitrakul and S. Izawa,Biochim. Biophys. Acta,305 (1973) 105.
A. Trebst, inMethods in Enzymology, A. San Pietro, ed., Academic Press, New York (1972), Vol. 24, pp. 146–165.
L.P. Vernon and W.S. Zaugg,J. Biol. Chem.,235 (1960) 2728.
C.C. Black Jr.,Biochim. Biophys. Acta,120 (1966) 332.
G. Zweig and M. Avron,Biochem. Biophys. Res. Commun.,19 (1965) 397.
B. Kok, H.J. Rurainski, and O.V.H. Owens,Biochim. Biophys. Acta,109 (1965) 347.
S. Saha, R. Ouitrakul, S. Izawa, and N.E. Good,J. Biol. Chem.,246 (1971) 3204.
Y. Mukohata, T. Yagi, and Y. Sugiyama,J. Bioenerg.,7 (1975) 103.
P. Mitchell,Biol. Rev.,41 (1966) 445.
R.E. McCarty, J.S. Fuhrman and Y. Tsuchiya,Proc. Natl. Acad. Sci. USA,68 (1971) 2522.
A. Telfer and M.C.W. Evans,Biochim. Biophys. Acta,256 (1972) 625.
H. Roy and E.N. Moudrianakis,Proc. Natl. Acad. Sci. USA,68 (1971) 2720.
G. Girault, J-M. Galmiche, M. Michel-Villaz, and J. Thiery,Eur. J. Biochem.,38 (1973) 473.
V. Vambutas and W. Bertsch,Biochim. Biophys. Acta,376 (1975) 169.
S.G. Reeves and D.O. Hall,Biochim. Biophys. Acta,314 (1973) 66.
J.M. Gould and S. Izawa,Biochim. Biophys. Acta,314 (1973) 211.
M. Schwartz,Nature,219 (1968) 915.
S. Izawa and G. Hind,Biochim. Biophys. Acta,143 (1967), 377.
H. Böhme and A. Trebst,Biochim. Biophys. Acta,180 (1969) 137.
K.R. West and J.T. Wiskich,Biochim. Biophys. Acta,292 (1973) 197.
W. Junge and W. Ausländer,Biochim. Biophys. Acta,333 (1974) 59.
S. Izawa, G.D. Winget, and N.E. Good,Biochem. Biophys. Res. Commun.,22 (1966) 223.
S. Izawa and N.E. Good,Biochim. Biophys. Acta,162 (1968) 380.
G. Forti, L. Rosa, and F. Garlaschi,FEBS Lett.,27 (1972) 23.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Yagi, T., Makohata, Y. Adenylate regulation of photosynthetic electron transport and the coupling sites of phosphorylation in spinach chloroplasts. J Bioenerg Biomembr 8, 247–255 (1976). https://doi.org/10.1007/BF00761450
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00761450